Chemistry:Barium peroxide

| |
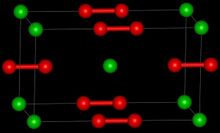 Barium cations Ba2+ Peroxide anions O2− 2 | |
| Names | |
|---|---|
| IUPAC name
barium peroxide
| |
| Other names
Barium binoxide,
Barium dioxide | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1449 |
| |
| |
| Properties | |
| BaO 2 | |
| Molar mass |
|
| Appearance | Grey-white crystalline solid (anhydrous) Colorless solid (octahydrate) |
| Odor | Odorless |
| Density |
|
| Melting point | 450 °C (842 °F; 723 K) |
| Boiling point | 800 °C (1,470 °F; 1,070 K) (decomposes to BaO & O 2.[1]) |
| |
| Solubility | dissolves with decomposition in acid |
| −40.6×10−6 cm3/mol | |
| Structure | |
| Tetragonal[2] | |
| D174h, I4/mmm, tI6 | |
| 6 | |
| Hazards | |
| GHS pictograms |  
|
| GHS Signal word | Warning |
| H272, H302, H332 | |
| P210, P220, P221, P261, P264, P270, P271, P280, P301+312, P304+312, P304+340, P312, P330, P370+378, P501 | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Barium peroxide is an inorganic compound with the formula BaO
2. This white solid (gray when impure) is one of the most common inorganic peroxides, and it was the first peroxide compound discovered. Being an oxidizer and giving a vivid green colour upon ignition (as do all barium compounds), it finds some use in fireworks; historically, it was also used as a precursor for hydrogen peroxide.[3]
Structure
Barium peroxide consists of barium cations Ba2+ and peroxide anions O2−
2. The solid is isomorphous to calcium carbide, CaC
2.
Preparation and use
Barium peroxide arises by the reversible reaction of O
2 with barium oxide. The peroxide forms around 500 °C (932 °F) and oxygen is released above 820 °C (1,510 °F).[1]
- 2 BaO + O
2 ⇌ 2 BaO
2
This reaction is the basis for the now-obsolete Brin process for separating oxygen from the atmosphere. Other oxides, e.g. Na
2O and SrO, behave similarly.[4]
In another obsolete application, barium peroxide was once used to produce hydrogen peroxide via its reaction with sulfuric acid:[3]
- BaO
2 + H
2SO
4 → H
2O
2 + BaSO
4
The insoluble barium sulfate is filtered from the mixture.
Footnotes
- ↑ 1.0 1.1 Middleburgh, Simon C.; Lagerlof, Karl Peter D.; Grimes, Robin W. (January 2013). "Accommodation of Excess Oxygen in Group II Monoxides". Journal of the American Ceramic Society 96 (1): 308–311. doi:10.1111/j.1551-2916.2012.05452.x.
- ↑ Massalimov, I. A.; Kireeva, M. S.; Sangalov, Yu. A. (2002). "Structure and Properties of Mechanically Activated Barium Peroxide". Inorganic Materials 38 (4): 363–366. doi:10.1023/A:1015105922260.
- ↑ 3.0 3.1 Harald Jakob; Stefan Leininger; Thomas Lehmann; Sylvia Jacobi; Sven Gutewort. "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a19_177.pub2.
- ↑ Wiberg, Egon; Wiberg, Nils; Holleman, A. F. (2001). Inorganic chemistry (1st English ed.). San Diego : Berlin ; New York: Academic Press ; De Gruyter. ISBN 0-12-352651-5.
See also
External links
 |

