Chemistry:Barium orthotitanate
Barium orthotitanate is the inorganic compound with the chemical formula Ba2TiO4. It is a colourless solid that is of interest because of its relationship to barium titanate, a useful electroceramic.
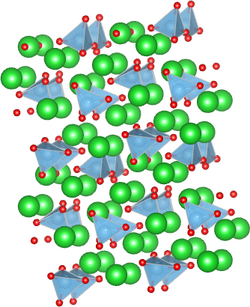
Structure
The solid has two known phases: a low-temperature (β) phase with P21/n symmetry[1] and a high-temperature (α′) phase with P21nb symmetry.[2] The structure of Ba2TiO4 is unusual among the titanates because its titanium atoms sit in a four-oxygen tetrahedron rather than a six-oxygen octahedron.[1][3]
Production
It forms as white crystals from a melt of BaCl2, BaCO3 and TiO2[4] or from just sintering BaCO3 and TiO2.[5] Another method of preparation is heating pellets of Ba(OH)2 and TiO2.[6] Additionally, there are polymer precursor, sol-gel and reverse micellar routes to Ba2TiO4 synthesis.[3] Ba2TiO4 has also been successfully grown as a thin film with chemical vapor deposition.[7]
Properties
- Room temperature entropy – 47.0 cal/deg. mol[6]
- Dielectric constant – 20 (at 100 kHz)[8]
- It is hygroscopic and decomposes with swelling in moist air.[9]
Barium orthotitanate can remove up to 99.9% of CO2 from a high-temperature gas stream[10] by the reaction:
- Ba2TiO4 + CO2 → BaTiO3 + BaCO3
References
- ↑ 1.0 1.1 Wu, Kang Kun; Brown, I. D. (10 April 1973). "The Crystal Structure of β-Barium Orthotitanate, β-Ba2TiO4, and the Bond Strength-Bond Length Curve of Ti-O". Acta Crystallographica B29 (9): 2009–2012. doi:10.1107/S0567740873005959.
- ↑ Günter, John R.; Jameson, Geoffrey B. (14 September 1983). "Orthorhombic Barium Orthotitanate, α′-Ba2TiO4". Acta Crystallographica C40: 207–210. doi:10.1107/S0108270184003619.
- ↑ 3.0 3.1 Shanker, Vishnu; Ahmad, Tokeer; Ganguli, Ashok K. (October 2004). "Investigation of Ba2–xSrxTiO4: Structural aspects and dielectric properties". Bulletin of Materials Science 27 (5): 421–427. doi:10.1007/BF02708558.
- ↑ Bland, J. A. (August 1961). "The Crystal Structure of Barium Orthotitanate, Ba2TiO4". Acta Crystallographica 14 (8): 875–881. doi:10.1107/S0365110X61002527.
- ↑ Bobovich, Ya. S.. "The Raman Scattering of Barium Orthotitanate". Optics and Spectroscopy 13: 254–255.
- ↑ 6.0 6.1 Todd, S. S.; Lorenson, R.E. (August 1952). "Heat Capacities at Low Temperatures and Entropies at 298.16K. of Orthotitanates of Barium and Strontium". Journal of the American Chemical Society 74 (15): 2043–2045. doi:10.1021/ja01135a014.
- ↑ Guo, Dongyun; Goto, Takashi; Wang, Chuanbin; Shen, Qiang; Zhang, Lianmeng (2012). "High-speed growth of (103)-oriented Ba2TiO4 film by laser chemical vapor deposition". Materials Letters 70: 135–137. doi:10.1016/j.matlet.2011.11.094.
- ↑ Ahmad, Tokeer; Ganguli, Ashok K. (1 October 2004). "Synthesis of nanometer-sized particles of barium orthotitanate prepared through a modified reverse micellar route: Structural characterization, phase stability and dielectric properties". Journal of Materials Research 19 (10): 2905–2912. doi:10.1557/JMR.2004.0406. Bibcode: 2004JMatR..19.2905A.
- ↑ Jaffe, Bernard; Cook Jr., William R.; Jaffe, Hans (1971). "5. Barium Titanate". Piezoelectric Ceramics. London and New York: Academic Press. p. 62. ISBN 0123795508.
- ↑ Saito, Yoshinori; Sato, Hideto; Sakabe, Yukio (20 May 2008). "CO2 Recovery at High Temperatures Using Ba2TiO4". Journal of Chemical Engineering of Japan 41 (5): 441–446. doi:10.1252/jcej.07WE236.
 |