Chemistry:Indaziflam
| |
| Names | |
|---|---|
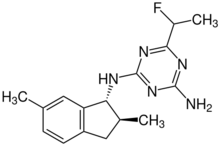
| IUPAC name
2-N-[(1R,2S)-2,6-dimethyl-2,3-dihydro-1H-inden-1-yl]-6-(1-fluoroethyl)-1,3,5-triazine-2,4-diamine[1]
| |
| Identifiers | |
3D model (JSmol)
|
|
| 20920435 | |
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties[2] | |
| C16H20FN5 | |
| Molar mass | 301.369 g·mol−1 |
| Density | 1.23 g/mL |
| Melting point | 183 °C (361 °F; 456 K) |
| 2.8 mg/L (20 °C) | |
| log P | 2.8 |
| Hazards | |
| GHS pictograms |  
|
| GHS Signal word | Warning |
| H373, H410 | |
| P260, P273, P314, P391, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Indaziflam is a preemergent herbicide especially for grass control in tree and bush crops.
History
In 1991, the Japanese company Idemitsu Kosan filed a patent to 2-amino 6-fluoroalkyl triazine derivatives as herbicides.[3] One of these compounds was subsequently given the ISO common name triaziflam but had limited success as a commercial herbicide.[4][5] Bayer scientists subsequently investigated this area of chemistry and identified indaziflam as having superior properties, which they patented and developed under the code number BCS-AA10717.[6][7] The compound was first registered for use in the USA in 2010.[8][9]
Mechanism of action
Indaziflam is an inhibitor of cellulose biosynthesis. This mechanism of action was theorized to be responsible for indaziflam's effect in 2009[7] and proven in 2014.[10] The cellulose biosynthesis inhibitors (CBIs) are identified as Class 29 by the Weed Science Society of America/Herbicide Resistance Action Committee.[11][12]
Resistance
As of March 2021[update] there are no resistant populations known[13] and none for the broader CBI class (discounting quinclorac).[11][14][15][16][17]
Brand names
Indaziflam composes all or part of the a.i. of several herbicides from Bayer Environmental Science (now owned by Cinven, aka Envu, per Bayer's and Envu's websites), [18] [19] including Rejuvra,[20] the Esplanade[21] line (sometimes mixed with diquat dibromide and glyphosate isopropylamine),[22] Marengo,[23][24] Specticle,[25][24] and Bayer CropScience (the inventor of the ingredient), like Alion.[26]
Uses
Indaziflam is approved in the United States for hops, Rubus spp., Coffea spp., bushberries, tropical crops, drupes/stone fruit, and tree nuts.[27] It is used as a preemergent.[28][27]
References
- ↑ Weed Science Society of America. "Common and chemical names approved by WSSA". http://wssa.net/wp-content/uploads/WSSA-Approved-Chem-Names.pdf.
- ↑ Pesticide Properties Database. "Indaziflam". University of Hertfordshire. http://sitem.herts.ac.uk/aeru/ppdb/en/Reports/1663.htm.
- ↑ ; Hirata T. & Kobayashi I. et al."Herbicidal compositions comprising 2-Amino-4-Arylalkylamino-6-Haloalkyl-1,3,5-Triazines and Chlorophenoxy Acids and, optionally, substituted ureas" US patent 5169425, issued 1992-12-08, assigned to Idemitsu Kosan Company Limited
- ↑ Grossmann, Klaus; Tresch, Stefan; Plath, Peter (2001). "Triaziflam and Diaminotriazine Derivatives Affect Enantioselectively Multiple Herbicide Target Sites". Zeitschrift für Naturforschung C 56 (7–8): 559–569. doi:10.1515/znc-2001-7-814. PMID 11531090.
- ↑ "Triaziflam (ISO)". https://chem.nlm.nih.gov/chemidplus/name/triaziflam%20%5Biso%5D.
- ↑ ; Dietrich H. & Minn K. et al."Amino 1,3,5-Triazines N-substituted with chiral bicyclic radicals" WO patent 2004069814, issued 2004-08-19, assigned to Bayer Cropscience GMBH
- ↑ 7.0 7.1 Meyer, F.; Hanrahan, R.; Michel, J.; Monke, B.; Mudge, L.; Norton, L.; Olsen, C.; Parker, A. et al. (2009). "Indaziflam/BCS-AA10717-A new herbicide for pre-emergent control of grasses and broadleaf weeds for turf and ornamentals". WSSA Meeting Abstracts. http://wssa.net/meeting/meetingabstracts.
- ↑ Mark D. Parrish; R. Darren Unland; William J. Bertges (7-10 December 2009). "Introduction of Indaziflam for Weed Control in Fruit, Nut, and Grape Crops". North Central Weed Science Society Proceedings (Kansas City, Mo: North Central Weed Science Society) 64: 164.
- ↑ "Bayer CropSciences new herbicide indaziflam received first registration in U.S." (in en). 6 September 2010. http://news.agropages.com/News/Detail-2830.htm.
- ↑ Brabham, C.; Lei, L.; Gu, Y.; Stork, J.; Barrett, M.; DeBolt, S. (30 July 2014). "Indaziflam Herbicidal Action: A Potent Cellulose Biosynthesis Inhibitor". Plant Physiology (American Society of Plant Biologists (OUP)) 166 (3): 1177–1185. doi:10.1104/pp.114.241950. ISSN 0032-0889. PMID 25077797.
- ↑ 11.0 11.1 Weed Science Society of America (3 December 2020). "WSSA-Herbicide Site of Action (SOA) Classification List". http://wssa.net/wp-content/uploads/WSSA-Herbicide-SOA-20201203.xlsx.
- ↑ Weed Science Society of America. "Summary of Herbicide Mechanism of Action According to the Weed Science Society of America (WSSA)". http://wssa.net/wp-content/uploads/WSSA-Mechanism-of-Action.pdf.
- ↑ "Herbicide Resistant Weeds by Individual Herbicide" (in en). Herbicide Resistance Action Committee. http://www.weedscience.org/summary/ResistByActive.aspx.
- ↑ "List of Herbicide Resistant Weeds by Herbicide Mode of Action (L/26)" (in en). Herbicide Resistance Action Committee. http://www.weedscience.org/summary/MOA.aspx?MOAID=19.
- ↑ Grossmann, Klaus; Kwiatkowski, Jacek (2000). "The Mechanism of Quinclorac Selectivity in Grasses". Pesticide Biochemistry and Physiology (Elsevier) 66 (2): 83–91. doi:10.1006/pest.1999.2461. ISSN 0048-3575.
- ↑ Tresch, Stefan; Grossmann, Klaus (2003). "Quinclorac does not inhibit cellulose (cell wall) biosynthesis in sensitive barnyard grass and maize roots". Pesticide Biochemistry and Physiology (Elsevier) 75 (3): 73–78. doi:10.1016/s0048-3575(03)00013-0. ISSN 0048-3575.
- ↑ Tresch, Stefan; Grossmann, Klaus (2003). "Erratum to "Quinclorac does not inhibit cellulose (cell wall) biosynthesis in sensitive barnyard grass and maize roots"". Pesticide Biochemistry and Physiology (Elsevier) 76 (2): 70–71. doi:10.1016/s0048-3575(03)00064-6. ISSN 0048-3575.
- ↑ "Bayer completes sale of its Environmental Science Professional business to Cinven". https://www.us.envu.com/pressrelease/bayer-completes-sale-of-environmental-science.
- ↑ "Bayer completes sale of its Environmental Science Professional business to Cinven". https://www.bayer.com/en/agriculture/environmental-science.
- ↑ "Envu's Rejuvra (indaziflam) Herbicide". https://www.us.envu.com/vegetation-management/range-and-pasture/products/rejuvra.
- ↑ "Esplanade 200 SC IVM Product". http://www.environmentalscience.bayer.us/vegetation-management/industrial-vegetation-management/products/esplanade-200-sc.
- ↑ "Esplanade EZ IVM Product". http://www.environmentalscience.bayer.us/vegetation-management/industrial-vegetation-management/products/esplanade-ez.
- ↑ Spesard, Bruce (8 June 2018). "Broadleaf and Grassy Weed Control". http://www.environmentalscience.bayer.us/turf-and-ornamentals-management/production-ornamentals/ornamentals-insights/broadleaf-and-grassy-weed-control.
- ↑ 24.0 24.1 "Marengo (indaziflam) or Specticle". NC State Ag Extension. 30 May 2014. http://content.ces.ncsu.edu/marengo-indaziflam.
- ↑ "Specticle Flo". http://www.environmentalscience.bayer.us/-/media/prfunitedstates/documents/resource-library/product-labels/specticle-flo.ashx.
- ↑ "US84467332F (170705Fv2) ALION SC 32 FOZ ETL 0119.indd". Bayer. 1 November 2019. http://www.cropscience.bayer.us/products/herbicides/alion/label-msds.
- ↑ 27.0 27.1 "PRIA Label Amendment – IR-4 tolerance petition and related amendments: (R190) to establish new uses on Hops, Caneberry subgroup 13-07A, Coffee, Bushberry subgroup 13-07B, Tropicals 23A and another amendment, and (R175) for crop group conversions in Stone Fruits 12-12 and Tree Nuts 14-12 Product Name: Indaziflam 200 SC Herbicide EPA Registration Number: 264-1106 Petition Number: 6E8452 Application Date: February 18, 2016 Decision Number(s): 514431, 514432, 514435, 514436". US EPA. 5 July 2017. http://www3.epa.gov/pesticides/chem_search/ppls/000264-01106-20170705.pdf.
- ↑ "Indaziflam". NCBI, NLM, US NIH. http://pubchem.ncbi.nlm.nih.gov/compound/44146693. CID=CID 44146693 from PubChem
 |
