Chemistry:Lithium methoxide
From HandWiki
 | |
| Names | |
|---|---|
| IUPAC name
Lithium methoxide
| |
| Other names
Lithium methanolate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| CH3LiO | |
| Molar mass | 37.975 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
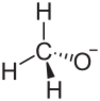
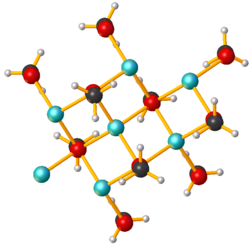
Lithium methoxide is a compound with formula LiCH3O. It is the lithium salt of methanol. Like other alkali metal alkoxides, lithium methoxide adopts a polymeric structure[1] Its solubility in common polar aprotic solvents like THF is low; however, it is soluble in methanol and is available commercially as a 10% solution.[2]
See also
References
- ↑ WHEATLEY, P. J. (1960-03-05). "Structure of Lithium Methoxide" (in en). Nature 185 (4714): 681–682. doi:10.1038/185681b0. Bibcode: 1960Natur.185..681W.
- ↑ "Lithium Methoxide (LiOMe) in 10% Methanol". 2016. http://www.fmclithium.com/Portals/FMCLithium/content/docs/DataSheet/QS-PDS-014%20r2.pdf.
 |
