Chemistry:Lithium ruthenate
| |
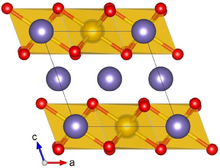
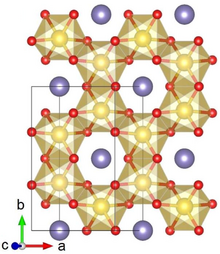 Crystal structure with Ru shown in yellow, Li in purple and O in red
| |
 Scale bar 0.1 mm[1]
| |
| Names | |
|---|---|
| Preferred IUPAC name
Lithium ruthenate | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| Li2RuO3 | |
| Appearance | Dark blue crystals |
| Structure[2] | |
| Monoclinic | |
| P21/m (No. 11) | |
Formula units (Z)
|
4 |
| Related compounds | |
Other anions
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Lithium ruthenate, Li2RuO3, or LRO, is a chemical compound of lithium, ruthenium and oxygen. It has a layered honeycomb crystal structure, and can be prepared by direct calcination of Ru metal or RuO2 and lithium carbonate at ca. 700 °C.[2] The material is a potential lithium-ion battery electrode material[2] and supercapacitor[3] because of its high specific capacity due to lattice oxygen redox.[4] This application is hindered by the high costs of Ru, as compared to the cheaper Li2MnO3 alternative[5]
Despite having a low-spin state of Ru(IV), namely 4d4, above 540 K there have been observed Ru-Ru dimers which decrease the magnetic susceptibility of the material.[6]
The compound is known to react with water in neutral and basic solutions to partially exchange Li2O with H2O in its structure. The new phase, Li2-xHxRuO3, is structurally different from pure Li2RuO3.[7]
References
- ↑ Freund, F.; Williams, S. C.; Johnson, R. D.; Coldea, R.; Gegenwart, P.; Jesche, A. (2016). "Single crystal growth from separated educts and its application to lithium transition-metal oxides". Scientific Reports 6. doi:10.1038/srep35362. PMID 27748402. Bibcode: 2016NatSR...635362F.
- ↑ 2.0 2.1 2.2 O'Malley, Matthew J.; Verweij, Henk; Woodward, Patrick M. (2008). "Structure and properties of ordered Li2IrO3 and Li2PtO3". Journal of Solid State Chemistry 181 (8): 1803. doi:10.1016/j.jssc.2008.04.005. Bibcode: 2008JSSCh.181.1803O.
- ↑ Yong-Qing, Zhao; Guo-Qing, Zhang; Hu-Lin, Li. "Electrochemical characterization on layered lithium ruthenate for electrochemical supercapacitors". Solid State Ionics 177: 1335–1339. doi:10.1016/j.ssi.2006.06.008.
- ↑ Yang, Yu. "Revealing Electronic Signatures of Lattice Oxygen Redox in Lithium Ruthenates and Implications for High-Energy Li-Ion Battery Material Designs". Chemistry of materials. doi:10.1021/acs.chemmater.9b01821.
- ↑ Yoshio, Masaki; Brodd, Ralph J.; Kozawa, Akiya (17 July 2010). Lithium-Ion Batteries: Science and Technologies. Springer Science & Business Media. p. 10. ISBN 978-0-387-34445-4. https://books.google.com/books?id=gkYhDYk6ftQC&pg=PA10.
- ↑ Yoko, Miura. "New-Type Phase Transition of Li2RuO3 with Honeycomb Structure". Journal of the Physical Society of Japan 76 (3). doi:10.1143/JPSJ.76.033705.
- ↑ R., Reshma; Tułodziecki, Michał; Binghong, Han. "Reactivity with Water and Bulk Ruthenium Redox of Lithium Ruthenate in Basic Solutions". Advanced Functional Materials 31 (2). doi:10.1002/adfm.202002249.
 |
