Chemistry:Oteseconazole
 | |
| Clinical data | |
|---|---|
| Trade names | Vivjoa |
| Other names | VT-1161 |
| License data | |
| Routes of administration | By mouth |
| Drug class | Antifungal |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
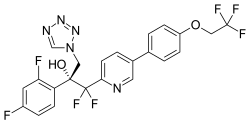
| Formula | C23H16F7N5O2 |
| Molar mass | 527.403 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Oteseconazole, a novel orally bioavailable and selective inhibitor of fungal cytochrome P450 enzyme 51 (CYP51), has shown promising efficacy in the treatment of recurrent vulvovaginal candidiasis (RVVC) in patients.[2][3]
Marketed under the brand name Vivjoa, this medication was developed by Mycovia Pharmaceuticals and received approval for medicinal use from United States Food and Administration (US FDA) in April 2022.[1][4][5]
Society and culture
Names
Oteseconazole is the international nonproprietary name (INN).[6]
Mechanism of action
Oteseconazole targets cytochrome P450 enzymes 51 (CYP51), which a play crucial role in maintaining the integrity and growth of fungal cell membranes. Through inhibition of these enzymes, oteseconazole prevents the synthesis of ergosterol, a key component of fungal cell membranes development. This disruption in ergosterol production leads to fungal membranes permeability, ultimately causing cell death.[2][3][7]
Oteseconazole exhibits selective inhibition of fungal CYP51 and has shown remarkable potency against Candida species during invitro pharmacological studies.[8] This targeted action of oteseconazole makes it a highly effective choice for treating RVVC.[2][9] Additionally, oteseconazole was found to possess superior activity against certain fungi, such as Candida glabrata compared to commonly used antifungals.[10]
Adverse effect and interaction
Oteseconazole has exhibited an outstanding tolerability profile and a low occurrence of adverse effects in clinical trials. In a phase 3 study, the incidence of treatment-emergent adverse events (TEAEs) was comparable between the oteseconazole and fluconazole/placebo groups, with the majority of TEAEs being of mild or moderate severity.[9] No serious adverse events related to the drug, as well as no adverse effects on liver function or QT intervals, were reported.[9] However, it is important to acknowledge that, like any medication, there is a potential risk of adverse effects. Therefore, it is crucial to consult with a healthcare provider prior to initiating oteseconazole or any other medication.
There is currently limited information available on oteseconazole interactions with other medications. The prescribing information for oteseconazole indicates that it is a moderate inhibitor of the CYP3A4 enzyme, suggesting that it may potentially increase the exposure of co-administered medications that are primarily metabolized by CYP3A4.[3] Therefore, patients who are taking medications metabolized by CYP3A4 should be closely monitored for any signs of toxicity or adverse effects when using oteseconazole.[3] It is crucial to have a discussion with a healthcare provider about the use of any medications or supplements to ensure safe and effective usage.
References
- ↑ 1.0 1.1 "Vivjoa- oteseconazole capsule". DailyMed. U.S. National Library of Medicine. 12 July 2022. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=e21d5008-800e-4417-927f-14340341865f.
- ↑ 2.0 2.1 2.2 "Oteseconazole: an advance in treatment of recurrent vulvovaginal candidiasis". Future Microbiology 16 (18): 1453–1461. December 2021. doi:10.2217/fmb-2021-0173. PMID 34783586.
- ↑ 3.0 3.1 3.2 3.3 "Oteseconazole: First Approved Orally Bioavailable and Selective CYP51 Inhibitor for the Treatment of Patients with Recurrent Vulvovaginal Candidiasis". Current Medicinal Chemistry 30 (37): 4170–4175. 2023. doi:10.2174/0929867330666230220130024. PMID 36803759.
- ↑ "Drugs@FDA: FDA-Approved Drugs" (in en). https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=215888.
- ↑ "FDA Approves Mycovia Pharmaceuticals' VIVJOA™ (oteseconazole), the First and Only FDA-Approved Medication for Recurrent Vulvovaginal Candidiasis (Chronic Yeast Infection)" (in en). 2022-04-28. https://www.businesswire.com/news/home/20220428005301/en/FDA-Approves-Mycovia-Pharmaceuticals%E2%80%99-VIVJOA%E2%84%A2-oteseconazole-the-First-and-Only-FDA-Approved-Medication-for-Recurrent-Vulvovaginal-Candidiasis-Chronic-Yeast-Infection.
- ↑ "International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 76". WHO Drug Information 30 (3). 2016.
- ↑ "Treating recurrent vulvovaginal candidiasis" (in en). OBG Manag 34 (11): 22-24, 28-30, 32. November 2022. doi:10.12788/obgm.0238. https://www.mdedge.com/obgyn/article/259225/gynecology/treating-recurrent-vulvovaginal-candidiasis. Retrieved 2023-06-26.
- ↑ "Oteseconazole: A Review" (in en). Journal of Biomedical and Pharmaceutical Research 11 (5). 2022-09-15. doi:10.32553/jbpr.v11i5.918. ISSN 2279-0594. https://jbpr.in/index.php/jbpr/article/view/918.
- ↑ 9.0 9.1 9.2 "107. A phase 3, randomized, double-blind study to evaluate the efficacy and safety of oteseconazole (VT-1161) oral capsules versus fluconazole and placebo in the treatment of acute vulvovaginal candidiasis episodes in subjects with recurrent vulvovaginal candidiasis (ultraViolet).". Open Forum Infectious Diseases (US: Oxford University Press) 8 (Supplement_1): S66–S67. November 2021. doi:10.1093/ofid/ofab466.107.
- ↑ "Review of the alternative therapies for onychomycosis and superficial fungal infections: posaconazole, fosravuconazole, voriconazole, oteseconazole". International Journal of Dermatology 61 (12): 1431–1441. December 2022. doi:10.1111/ijd.15999. PMID 34882787.
External links
- "Oteseconazole". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/oteseconazole.
- Clinical trial number NCT03562156 for "A Study of Oral Oteseconazole for the Treatment of Patients With Recurrent Vaginal Candidiasis (Yeast Infection) (VIOLET)" at ClinicalTrials.gov
- Clinical trial number NCT03561701 for "A Study of Oral Oteseconazole (VT-1161) for the Treatment of Patients With Recurrent Vaginal Candidiasis (Yeast Infection) (VIOLET)" at ClinicalTrials.gov
- Clinical trial number NCT03840616 for "Study of Oral Oteseconazole (VT-1161) for Acute Yeast Infections in Patients With Recurrent Yeast Infections (ultraVIOLET)" at ClinicalTrials.gov
 |
