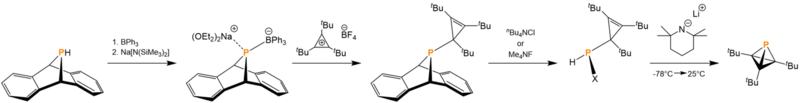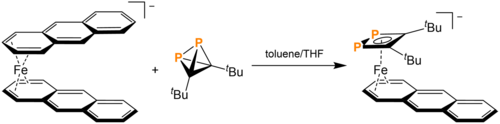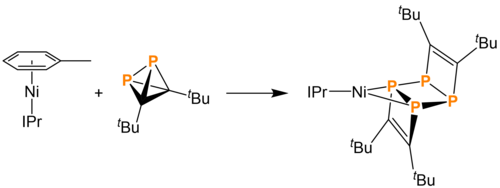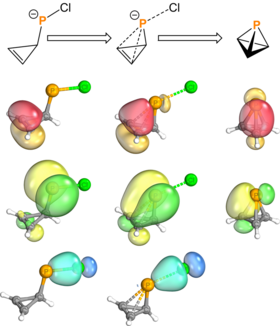Chemistry:Pnictogen-substituted tetrahedranes
Pnictogen-substituted tetrahedranes are pnictogen-containing analogues of tetrahedranes with the formula RxCxPn4-x (Pn = N, P, As, Sb, Bi).[1] Computational work has indicated that the incorporation of pnictogens to the tetrahedral core alleviates the ring strain of tetrahedrane.[2] Although theoretical work on pnictogen-substituted tetrahedranes has existed for decades, only the phosphorus-containing species have been synthesized.[3][4][5] These species exhibit novel reactivities, most often through ring-opening and polymerization pathways. Phosphatetrahedranes are of interest as new retrons for organophosphorus chemistry. Their strain also make them of interest in the development of energy-dense compounds.
History
The first synthetic tetrahedral molecule, tetra-tert-butyltetrahedrane (tBu4C4) was reported in 1978 by Maier and coworkers[6] following the synthesis of other Platonic solid species, like cubane and dodecahedrane. The tert-butyl substituents were used to encumber the tetrahedral core and quell the radical-mediated ring-opening of an otherwise kinetically stable but thermodynamically strained molecule via the corset effect. As of 2023, the unencumbered tetrahedrane (H4C4) has yet to be synthesized.
The substitution of carbons in the tetrahedral core of tetrahedrane with pnictogens to stabilize the highly strained molecule has been suggested due to the known existence of elemental tetrahedral pnictogens. Notably, white phosphorus, the most stable allotrope of phosphorus, is tetrahedral with the molecular formula P4.[7] Arsenic can also exist as a metastable tetrahedral allotrope, As4, known as yellow arsenic. Furthermore, mixed tetrahedral pnictogen molecules have been synthesized, such as AsP3[8] and, more recently, (PbBi3)-.[9] Elements on the extreme ends of the pnictogen family have not yet been observed in a tetrahedral Pn4 configuration, however. Nitrogen's orbitals lack diffusivity, and bismuth’s orbitals undergo minimal hybridization due to relativistic contraction.
Computational studies into mixed pnictogen-tetrel tetrahedranes have suggested that pnictogen-substituted tetrahedranes are more stable than their all tetrel counterparts decades before their first synthesis. In 1990, calculations on azatetrahedranes suggested positive correlation between the number of nitrogens in the tetrahedral core and thermodynamic stability.[2] In the same vein, calculations done in 2010 on pnictacubanes suggested positive correlation between the number of phosphoruses in the cuboidal core and the thermodynamic stability.[10]
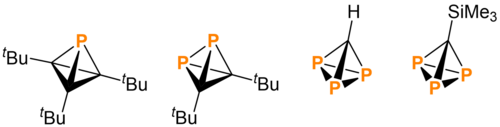
In 2019, Wolf and coworkers synthesized the first pnictogen-substituted tetrahedrane: di-tert-butyldiphosphatetrahedrane (tBu2C2P2), produced from the reaction of nickel catalyst with phosphaalkynes.[4] Shortly thereafter, in 2020, Cummins and coworkers announced that they had synthesized tri-tert-butylmonophosphatetrahedrane (tBu3C3P) from a phosphorus-containing anthracene derivative.[3] In 2021, Cummins and coworkers published the synthesis of triphosphatetrahedrane (HCP3), completing the set of tetrahedral molecules with carbon- and phosphorus-containing cores.[5]
Phosphatetrahedrane Synthesis
Despite the presentation of phosphatetrahedranes as a series of incrementally changing tetrahedrane derivatives, their syntheses are vastly different.[7]
Tri-tert-butylmonophosphatetrahedrane
The original synthesis of tBu3C3P reported by Cummins and coworkers in 2020 begins with the phosphorus derivative of anthracene. The addition of sodium hexamethyldisilazide deprotonates the phosphorus followed by triphenylborane then bonding to the phosphorus. The anionic phosphorus-containing intermediate then forms an ionic interaction with the sodium cation in solution. The addition of tri-tert-butyl cyclopropenium ion produces the thermally stable cyclopropenyl phosphine intermediate. Upon irradiation with 254 nm light in the presence of triflic acid and either tetra-n-butyl ammonium chloride or tetramethylammonium fluoride, the anthracene leaving group is forced out, leaving a halogenated phosphine. Upon addition of lithium tetramethylpiperidide with heating, tBu3C3P and lithium halide salt is generated.[3]
An improved version of tri-tert-butylmonophosphatetrahedrane synthesis, where the anthracene is replaced by two trimethylsilyl groups, was reported by Cummins and coworkers a year later. To replace one of the trimethylsilyl groups with a chloride, hexachloroethane is added, generating trimethylsilyl chloride and tetrachloroethylene as byproducts. Tetramethylammonium fluoride is then added to remove the remaining trimethylsilyl group and the chloride, generating the tBu3C3P. This route has a tBu3C3P yield of 33%[11] as compared to the original route's 19%.[3]
Di-tert-butyldiphosphatetrahedrane
In 2019, Wolf and coworkers reported the synthesis of tBu2C2P2 through the use of a metal catalyst. Ni(IPr)(CO)3, upon addition of 1 equivalent of tert-butylphosphaacetylene (tBuCP), loses two carbon monoxide ligands. The addition of a second equivalent of tBuCP generates the 1,3-diphosphacyclobutadiene ligand, now binding with η4 hapticity. Density functional theory calculations into the catalytic cycle suggest that the 1,3-diphosphacyclobutadiene isomerizes into the desired tetrahedrane. Upon addition of a final tBuCP, (tBu2C2P2) is released and the catalytic cycle can begin again.[4]
Triphosphatetrahedrane
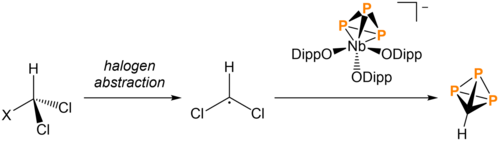
Cummins and coworkers reported the synthesis of HCP3 in 2021. Due to the similarity of HCP3 to AsP3, the [NbII(ODipp)3(P3)]- previously shown to be a retron for AsP3 was used for the synthesis of HCP3. To add a -CH group to [P3]3-, bromodichloromethane undergoes halogen abstraction, leaving a carbon-centered radical. The niobium complex then undergoes P3 transfer to yield HCP3. The use of bromodichloromethyl trimethylsilane instead of bromodichloromethane in this process yields trimethylsilyl triphosphatetrahedrane ((Me3S)CP3).[5][12]
Reactivity
Tri-tert-butylmonophosphatetrahedrane
Lewis Acid-Induced Reactions
Addition of W(CO)5(THF) to tBu3C3P generates a phosphorus-containing housene analogue.[3]
The addition of 0.2 equivalents of triphenylborane in benzene can produce several cycloadducts. In the absence of exogenous reagents, tBu3C3P dimerizes into a ladderane-like compound with a P-P bond. In the presence of excess styrene or an atmosphere of ethylene, [4 + 2] cycloadditions occur to give 1-phosphabicyclo[2.2.0]hexenes.[11]
Silylene Reaction
The cage opening of tBu3C3P can be induced by PhC(NtBu)2SiN(SiMe3)2 over the course of 24 hours to generate the dark red phosphasilene PhC(NtBu)2Si=P(tBu3C3).[13]
Ylide Reaction
Reaction of tBu3C3P with the ylide Ph3P=CH2 over 48 hours and with heat induces cage opening in the same manner as the silylene reaction to generate H2C=P(tBuC)3. Reaction of this product with tBu3C3P generates the symmetric product (tBuC)3P(C)P(tBuC)3.[13]
Formation of Phosphirane
tBu3C3P is a retron for phosphirane synthesis. Upon reaction with Ni(COD)2 (COD = cycloocta-1,5-diene) catalyst in triisopropylphosphine, cage opening occurs. Like the silylene and ylide reactions, the phosphorus bridges the (tBuC)3 and the alkene components. The phosphate undergoes cycloaddition with the double bond to form the phosphirane moiety. This reaction pathway has been demonstrated for styrene, ethylene, and neohexene. Furthermore, this reaction pathway is also capable of synthesizing vinyl-substituted phosphirane as evidenced by tBu3C3P and cyclohexa-1,3-diene.[13]
Ligand Substitution
tBu3C3P can be used to replace the ethylene ligand of (Ph3P)Pt(C2H4) in melting THF.[11]
Di-tert-butyldiphosphatetrahedrane
Dimerization Reactions
Above the melting point of tBu2C2P2 (–32 °C), tBu2C2P2 dimerizes into another ladderane-like structure but it is prone to decomposition. This reaction can be hampered by keeping tBu2C2P2 under its melting point and/or by keeping the tBu2C2P2 concentration low.[4]
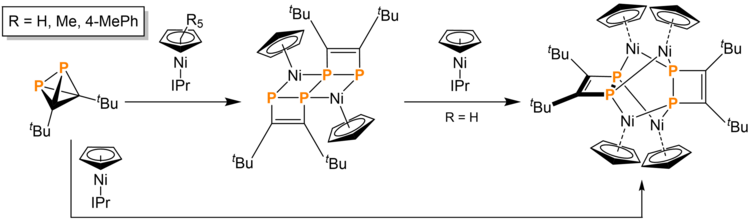
tBu2C2P2 can also be dimerized using nickel complexes to form a variety of exotic structures. tBu2C2P2 reacted with 1 equivalent of Ni(CpR)(IPr) (IPr = 1,3-bis(2,6-diisopropylphenyl)imidazolin-2-ylidene, R = H, CH3, 4-(CH3CH2)-C6H4) generates 0.5 equivalent of a tetracyclo-compound. Upon addition of another equivalent of the same nickel complex, a butterfly-like geometry is adopted, with two nickel atoms coordinated to opposite phosphorus atoms and two coordinated to adjacent phosphorus atoms on different four membered rings. This butterfly-structured compound is a dark red color. The reaction to the butterfly structure is believed to depend on kinetic access to the middle P-P bond. Bulky substituents on CpR kinetically hinder the P-P bond cleavage and transformation into the butterfly-structured product.
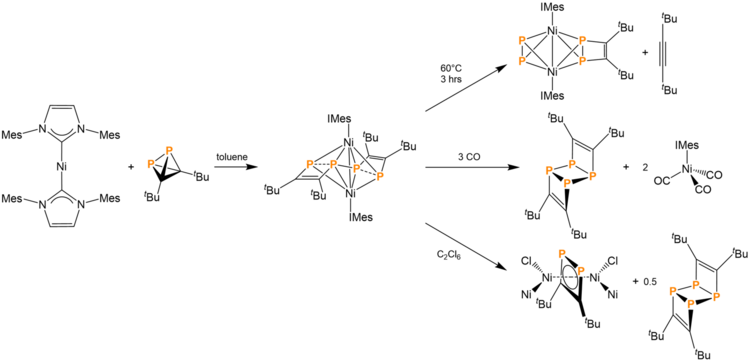
tBu2C2P2 can also be reacted with Ni(IMes)2 (IMes = 1,3-bis(2,4,6-trimethylphenyl)imidazolin-2-ylidene) in toluene to produce 0.5 equivalents of an asymmetric compound with two hexacoordinate Ni atoms, a Ni-Ni bond, and two weak P-P interactions. This product is an intermediate for further chemistry. Heating the product at 60 °C for 3 hours causes the expulsion of a di-tert-butylacetylene and the reformation of P-P bonds. Another reaction pathway involves the addition of 3 equivalents of CO to the product, leading to the production of Ni(IMes)(CO)3 and the ladderane-like compound described at the beginning of this section. A third reaction pathway involves the addition of hexachloroethane. This produces a 1,2-diphosphocyclobutadiene ring (vide supra) that is coordinated to both nickel atoms. This third reaction pathway also produces the ladderane analogue.[14]
Ligand Substitution
In solution with coordination complexes, tBu2C2P2 can cause ligand substitution. Most of these reactions cause cage-opening. The reaction of tBu2C2P2 with [K([18]crown-6)][Fe(anthracene)2] in toluene and THF causes the expulsion of one anthracene and the cage opening of tBu2C2P2 to form the replacement 1,2-diphosphacyclobutadiene ligand. This cage opening is due to P-C bond cleavage. Mono-ligand substitution is also observed in the reaction of tBu2C2P2 with [(DippBIAN)M(COD)] (Dipp = 2,6-diisopropylphenyl, BIAN = bis(arylimino)acenaphthene, M = Fe, Co). The cobalt product, upon reaction with Cy2PCl (Cy = cyclohexyl), forms a 1,2,3-triphospholium ligand. tBu2C2P2 can also displace the toluene in Ni(toluene)(IPr). Toluene is replaced by the aforementioned ladderane analogue in a η2-fashion.[15]
Double-ligand substitution is seen in the reaction of tBu2C2P2 with [Co(COD)2][K(THF)0.2]. The major product has its ligands doubly substituted by 1,2-diphosphacyclobutadiene. A minor product with double substitution by 1,3-diphosphacyclobutadiene ligand is also observed. However, the cobalt complex with one 1,2-diphosphacyclobutadiene ligand and one 1,3-diphosphacyclobutadiene ligand is not observed; this is likely due to steric clash between the tert-butyl substituents. The preference for 1,2-diphosphacyclobutadiene makes tBu2C2P2 a potentially valuable retron as phosphalkynes are known to produce 1,3-diphosphacyclobutadiene.[15]
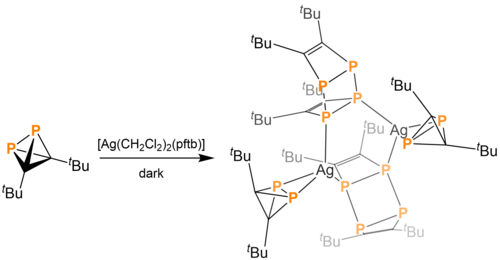
Only two of tBu2C2P2's ligand substitution reactions are known to preserve the tetrahedral cage. Reacting (pftb)[Ag(CH2Cl2)2] (pftb = Al[PFTB]- = Al[OC(CF3)3]4-) with tBu2C2P2 in lightless conditions leads to the generation of a disilver complex wherein each of the two tBu2C2P2 ligates to one silver atom and each of the two ladderane analogues (vida infra) ligates to both silver atoms.[4] By reacting Ni(CO)4 in THF at –80 °C and in the absence of light, three tBu2C2P2 molecules coordinate to Ni in an η2-fashion. Calculations by Intrinsic Bond Orbital (IBO) theory suggest that the coordination occurs through a 3-center-2-electron bond.[16]
Reactions with N-Heterocyclic Carbenes
tBu2C2P2 can be used a retron to form phosphirenes or phosphaalkenes with the addition of 1 equivalent or 2 equivalents of N-heterocyclic carbenes (NHC), respectively. Upon the addition of 1 equivalent of IPr, IMes, or MesDAC (1,3-bis(2,4,6-trimethylphenyl)diamidocarbene), tBu2C2P2 undergoes ring opening at one phosphorus atom's P-C bonds, creating structures with a bridging P-P bond between the NHC and the phosphirene. IBO calculations and crystallographic evidence support the assignment of double bonding to the P=C bond to resultant molecule. This reaction is very slow, taking several weeks to reach completion.
Upon the addition of 2 equivalents of TMC (2,3,4,5-tetramethylimidazolin-2-ylidene) in benzene, both phosphorus atoms bond to THC. The P-P bond is broken. A double bond also forms between the two carbons of tBu2C2P2, generating a phosphaalkene. This reaction happens significantly faster, with a reported speed of 1 hour.
Selectivity between the two reactions is suggested to be achieved by changing the steric bulk of the NHC used. A bulky NHC should prefer generating a phosphirene, whereas a smaller NHC should prefer generating a phosphaalkene.[17]
Triphosphatetrahedrane
Reaction with (dppe)Fe(Cp*)Cl
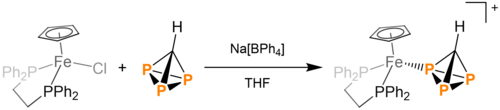
HCP3, upon addition of [(dppe)Fe(Cp*)]Cl (dppe = 1,2-bis-(diphenylphosphino)ethane) in sodium tetraphenylborate and THF, undergoes salt metathesis and produces [(dppe)Fe(Cp*)(HCP3)][BPh4]. This product is crystallizable, producing purple crystals. This product is prone to decomposition back to HCP3.[5]
Theoretical Work
Azatetrahedranes
Bonding Parameters
Ring and cage strain results in poor angular overlap of orbitals, leading to non-linear bonding. Due to the interest in tetrahedrane and their azatetrahedral analogues as highly strained molecules, Politzer and Seminario introduced the "bond deviation index" to determine the deviation of the bond path — defined as the path following maxima between nuclei — and the linear bond between the nuclei.
[math]\displaystyle{ \lambda=\frac{1}{R}\bigg[\frac{1}{N}\sum_i^N{r_i^2}\bigg]^{\frac{1}{2}} }[/math]
The strain of experienced by H4C4 is calculated to be partially alleviated upon the substitution of carbon atoms by nitrogen atoms.[18] The bond deviation index decreases with the number of nitrogens in azatetrahedranes from 0.114 to 0.087 from 0 to 2 nitrogen atoms.[19] The engendered stability is countered by the propensity of the N-N bond in the strained system to escape as dinitrogen. This is evidenced by the calculated bond length: 1.59 Å, which is much higher than that of aromatic N-N bonds: 1.21-1.36 Å.[18][19]
On the basis of the higher electronegativity of nitrogen than carbon, azatetrahedranes have less negative electrostatic potentials at their C-C bonds than H4C4, leading to greater stability against electrophilic attacks.[18] C-C and N-C bonds both contract in azatetrahedranes.[18] This contraction leads to smaller bond deviations and stronger bonding,[20] although the latter is dependent on the distance from the nitrogen atom(s).[19] Azatetrahedranes also seem to distort from an ideal tetrahedral symmetry. These trends directly correlates with the number of nitrogen atoms. Nitration also contracts the tetrahedral core. However, essentially thermodynamically neutral nitro group rotation leads to small amounts of C-N lengthening,[21] weaking the interaction and localizing the electrons.
Ionization of all azatetrahedranes cores in the series led to cage-opening at the G4MP2 and G4 levels of theory.[22]
Thermodynamics
Early work on azatetrahedranes has also utilized isodesmic reactions — aphysical reactions where compounds are changing but bond types are not — to understand molecular stability.[2][23] The isodesmic reaction energy is thus a metric of the stabilization/destabilization relative to the starting reagents. Overall, as the number of nitrogens increase, the stability of the system increases. The isodesmic reaction energy goes from 151.8 kcal/mol to 81.8 kcal/mol for 0 to 4 nitrogen atoms. Nitration destabilizes the energy dramatically, as exemplified by 2,4-dinitro-1,3-diazatetrahedrane ((O2NC)2N2) having an isodesmic reaction energy of 152.9 kcal/mol.[23]
Due to the instability of many azatetrahedranes, isodesmic comparisons to azacyclobutadiene analogues have been used to determine which core structures are the most synthetically feasible. Alkorta, Elguero, and Rozas reported that every member of the azatetrahedrane core series is always slightly more unstable than their azacyclobutadiene analogue(s).[2] Jursic's calculations suggest that the energetic differential between the azatetrahedrane and the azacyclobutadiene starts off large and decreases as the number of nitrogen atoms increase in the cage. Furthermore, Jursic's calculations suggest that tetraazatetrahedrane may be slightly more stable (difference of 4.4 kcal/mol at 0 K and the CBSQ level of theory) than its azacyclobutadiene analogue. Despite the instability, non-sterically hindered azatetrahedranes may still be detectable in gaseous matrices.[24]
Phosphatetrahedranes
Bonding Parameters
Much like azatetrahedranes, phosphatetrahedranes show bond deviation. The molecular graph of tBu3C3P shows significantly warped bond paths; the bond critical points lie far from the linear representation.[3]
Investigations into the bonds of phosphatetrahedranes used quasi-atomic orbital analysis with isodesmic reactions. In contrast to azatetrahedranes, the lower electronegativity of phosphorus relative to carbon leads to a localization of negative charge in the carbons and the localization of positive charge in the phosphorus(es). This leads to greater occupancy in the C-C-C ring from 4.17 to 4.28 electrons with the addition of one phosphorus atom. Overall, the extent of charge transfer increases with the number of phosphorus atoms in the tetrahedral core.[25]
Of the phosphatetrahedranes, only the triphosphatetrahedrane core did not show evidence of cage-opening upon ionization.[22]
Thermodynamics
Ivanov, Bozhenko, and Boldyrev studied the energetic landscape of the phosphatetrahedrane series ((HC)xP4-x). Their calculations suggest that substitution of phosphorus for carbon increasingly favors the tetrahedral structure. The tetrahedral structure is the absolute minima starting for triphosphatetrahedrane, but diphosphatetrahedrane is only 2.3 kcal/mol higher in energy than the absolute minima. They attribute the stabilization of the tetrahedral structure to phosphorus' amiability towards more acute bond angles. The more diffuse orbitals of phosphorus versus carbon also favor the tetrahedral structure's σ-interactions over the planar phosphacyclobutadiene's π-interactions.[1]
Riu, Ye, and Cummins report similar computational findings. Their calculations show decreasing strain energy with the number of phosphorus atoms in the tetrahedral cage. They also attribute the stabilization to the diffusitivity of phosphorus orbitals. They also note that the accumulation of p-character on the bond orbitals leads to greater s-character on the lone pairs.[5]
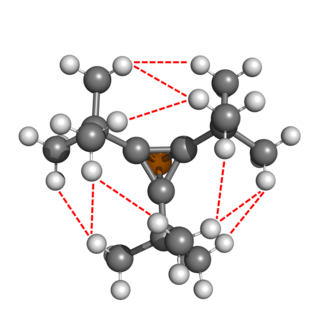
The isolability of tBu3C3P was attributed to the controversial hydrogen-hydrogen bonds (HHB), which some chemists have argued may not exist. Each HHB of the tert-butyl network were calculated (in absence of steric repulsion) to contribute 0.7 kcal/mol of stabilization. Calculations with one of the tert-butyl substituents with a methyl, ethyl, or isopropyl group result in net repulsion due to the loss of HHBs.[26] In total, this forms the basis of the corset effect.[5] Non-Lewis donation of electron density from the tetrahedral core to the tert-butyl substituents also stabilizes tBu3C3P according to natural bond orbital theory.[5] This effect was also demonstrated in silico for unsubstituted monophosphatetrahedrane.[27]
Substitution by Heavier Congeners
Schaefer and coworkers, in light of the synthesis of tBu3C3P, ran calculations on the mono-pnictogen-substituted tetrahedrane series, represented by R3C3Pn (R = H, tBu, Pn = N, P, As, Sb, Bi). These calculations yielded a series of well correlated trends.
Consistent with a perturbation of the pnictogen residing above a C-C-C ring, the C-Pn bonds elongate from 1.493 Å to 2.289 Å, and C-Pn-C angle decreases from 58.0° to 37.1° as heavier congeners are used. This is due to the larger atomic radius of the heavier pnictogens. The H-[C-C-C plane] angle increases from 9.1° to 31.1°, which is also attributed to the diffusivity of the heavier congener's orbitals.
As noted above with the aza- and phosphatetrahedranes, the change in pnictogen electronegativity changes the interaction between the Pn atom and the C-C-C ring. The C-C-C ring becomes increasingly more negatively charged with the heavier pnictogens.
Isodesmic reactions show greater stabilization of the cage structure due to the diffusivity of the pnictogen's orbitals, although even with bismuth, the mono-pnictogen-substituted tetrahedrane is unstable. Delocalization plays a large part in the stabilization of the heavier analogues. For example, electron density is increasingly transferred from the Pn-C bonds into the Pn lone pair in the heavier congeners. These lone pairs are also noted to follow Bent's rule.
As noted above with tBu3C3P, non-Lewis interactions stabilize the tetrahedral core. These effects also become more pronounced with the heavier pnictogens. Second order perturbations suggest that the key non-Lewis interactions are C-C to C-R* and C-Pn to C-H* (i.e., cage-opening), as well as interactions to Pn-C*. The former set of interactions stabilize the tetrahedral core most when the substituent is an electron-withdrawing group (e.g., fluoride), although decreased electron density in C-C and C-Pn can facilitate cage-opening as well.[27]
See also
- Tetrahedrane
- White Phosphorus
- Yellow Arsenic
- Prismane
- Cubane
- Cyclobutadiene
References
- ↑ Jump up to: 1.0 1.1 Ivanov, Alexander S.; Bozhenko, Konstantin V.; Boldyrev, Alexander I. (2012-01-10). "Peculiar Transformations in the C x H x P 4– x ( x = 0–4) Series" (in en). Journal of Chemical Theory and Computation 8 (1): 135–140. doi:10.1021/ct200727z. ISSN 1549-9618. https://pubs.acs.org/doi/10.1021/ct200727z.
- ↑ Jump up to: 2.0 2.1 2.2 2.3 Alkorta, Ibon; Elguero, Jose; Rozas, Isabel; Balaban, Alexandru T. (August 1990). "Theoretical studies on aza-analogs of platonic hydrocarbons" (in en). Journal of Molecular Structure 208 (1-2): 63–77. doi:10.1016/0166-1280(92)80008-A. https://linkinghub.elsevier.com/retrieve/pii/016612809280008A.
- ↑ Jump up to: 3.0 3.1 3.2 3.3 3.4 3.5 Riu, Martin-Louis Y.; Jones, Rebecca L.; Transue, Wesley J.; Müller, Peter; Cummins, Christopher C. (2020-03-27). "Isolation of an elusive phosphatetrahedrane" (in en). Science Advances 6 (13). doi:10.1126/sciadv.aaz3168. ISSN 2375-2548. PMID 32232162. PMC 7096166. https://www.science.org/doi/10.1126/sciadv.aaz3168.
- ↑ Jump up to: 4.0 4.1 4.2 4.3 4.4 Hierlmeier, Gabriele; Coburger, Peter; Bodensteiner, Michael; Wolf, Robert (2019-11-18). "Di‐ tert ‐butyldiphosphatetrahedrane: Catalytic Synthesis of the Elusive Phosphaalkyne Dimer" (in en). Angewandte Chemie International Edition 58 (47): 16918–16922. doi:10.1002/anie.201910505. ISSN 1433-7851. PMID 31591760. PMC 6899750. https://onlinelibrary.wiley.com/doi/10.1002/anie.201910505.
- ↑ Jump up to: 5.0 5.1 5.2 5.3 5.4 5.5 5.6 Riu, Martin-Louis Y.; Ye, Mengshan; Cummins, Christopher C. (2021-10-13). "Alleviating Strain in Organic Molecules by Incorporation of Phosphorus: Synthesis of Triphosphatetrahedrane" (in en). Journal of the American Chemical Society 143 (40): 16354–16357. doi:10.1021/jacs.1c07959. ISSN 0002-7863. https://pubs.acs.org/doi/10.1021/jacs.1c07959.
- ↑ Maier, Günther; Pfriem, Stephan; Schäfer, Ulrich; Matusch, Rudolf (July 1978). "Tetra‐ tert ‐butyltetrahedrane" (in en). Angewandte Chemie International Edition in English 17 (7): 520–521. doi:10.1002/anie.197805201. ISSN 0570-0833. https://onlinelibrary.wiley.com/doi/10.1002/anie.197805201.
- ↑ Jump up to: 7.0 7.1 Jupp, Andrew R.; Slootweg, J. Chris (2020-06-26). "Mixed Phosphatetrahedranes" (in en). Angewandte Chemie International Edition 59 (27): 10698–10700. doi:10.1002/anie.202004514. ISSN 1433-7851. https://onlinelibrary.wiley.com/doi/10.1002/anie.202004514.
- ↑ Cossairt, Brandi M.; Diawara, Mariam-Céline; Cummins, Christopher C. (2009-01-30). "Facile Synthesis of AsP 3" (in en). Science 323 (5914): 602–602. doi:10.1126/science.1168260. ISSN 0036-8075. https://www.science.org/doi/10.1126/science.1168260.
- ↑ Pan, Fuxing; Guggolz, Lukas; Dehnen, Stefanie (July 2022). "Capture the missing: formation of (PbBi 3 ) − and {[AuPb 5 Bi 3 2 } 4− via atom exchange or reorganization of the pseudo‐ tetrahedral Zintl anion (Pb 2 Bi 2 ) 2−"] (in en). Natural Sciences 2 (3). doi:10.1002/ntls.202103302. ISSN 2698-6248. https://onlinelibrary.wiley.com/doi/10.1002/ntls.202103302.
- ↑ Rayne, Sierra; Forest, Kaya (2010-11-01). "Structures, enthalpies of formation, and ionization energies for the parent and binary mixed carbon, silicon, nitrogen, and phosphorus cubane derivatives: A G4MP2 theoretical study" (in en). Nature Precedings. doi:10.1038/npre.2010.5154.1. ISSN 1756-0357. https://www.nature.com/articles/npre.2010.5154.1.
- ↑ Jump up to: 11.0 11.1 11.2 Riu, Martin-Louis Y.; Eckhardt, André K.; Cummins, Christopher C. (2021-08-25). "Dimerization and Cycloaddition Reactions of Transient Tri- tert -butylphosphacyclobutadiene Generated by Lewis Acid Induced Isomerization of Tri- tert -butylphosphatetrahedrane" (in en). Journal of the American Chemical Society 143 (33): 13005–13009. doi:10.1021/jacs.1c06840. ISSN 0002-7863. https://pubs.acs.org/doi/10.1021/jacs.1c06840.
- ↑ Riu, Martin-Louis Y. (September 2022). Synthesis and Reactivity of Phosphorus-Containing Heterocycles and Tetrahedranes (Thesis thesis). Massachusetts Institute of Technology.
- ↑ Jump up to: 13.0 13.1 13.2 Riu, Martin-Louis Y.; Eckhardt, André K.; Cummins, Christopher C. (2022-05-04). "Reactions of Tri- tert -Butylphosphatetrahedrane as a Spring-Loaded Phosphinidene Synthon Featuring Nickel-Catalyzed Transfer to Unactivated Alkenes" (in en). Journal of the American Chemical Society 144 (17): 7578–7582. doi:10.1021/jacs.2c02236. ISSN 0002-7863. https://pubs.acs.org/doi/10.1021/jacs.2c02236.
- ↑ Hierlmeier, Gabriele; Wolf, Robert (2021-03-15). "Activation of Di‐ tert ‐butyldiphosphatetrahedrane: Access to ( t BuCP) n ( n= 2, 4) Ligand Frameworks by P−C Bond Cleavage" (in en). Angewandte Chemie International Edition 60 (12): 6435–6440. doi:10.1002/anie.202015680. ISSN 1433-7851. PMID 33403771. PMC 7986217. https://onlinelibrary.wiley.com/doi/10.1002/anie.202015680.
- ↑ Jump up to: 15.0 15.1 Hierlmeier, Gabriele; Coburger, Peter; Scott, Daniel J.; Maier, Thomas M.; Pelties, Stefan; Wolf, Robert; Pividori, Daniel M.; Meyer, Karsten et al. (2021-10-25). "Di ‐tert ‐butyldiphosphatetrahedrane as a Source of 1,2‐Diphosphacyclobutadiene Ligands" (in en). Chemistry – A European Journal 27 (60): 14936–14946. doi:10.1002/chem.202102335. ISSN 0947-6539. PMID 34424579. PMC 8596834. https://chemistry-europe.onlinelibrary.wiley.com/doi/10.1002/chem.202102335.
- ↑ Uttendorfer, Maria K.; Hierlmeier, Gabriele; Wolf, Robert (2022-10-14). "A Homoleptic Diphosphatetrahedrane Nickel(0) Complex" (in en). Zeitschrift für anorganische und allgemeine Chemie 648 (19). doi:10.1002/zaac.202200124. ISSN 0044-2313. https://onlinelibrary.wiley.com/doi/10.1002/zaac.202200124.
- ↑ Hierlmeier, Gabriele; Uttendorfer, Maria K.; Wolf, Robert (2021). "Di- tert -butyldiphosphatetrahedrane as a building block for phosphaalkenes and phosphirenes" (in en). Chemical Communications 57 (19): 2356–2359. doi:10.1039/D0CC07103J. ISSN 1359-7345. http://xlink.rsc.org/?DOI=D0CC07103J.
- ↑ Jump up to: 18.0 18.1 18.2 18.3 Politzer, Peter; Seminario, Jorge M. (January 1989). "Computational determination of the structures and some properties of tetrahedrane, prismane, and some of their aza analogs" (in en). The Journal of Physical Chemistry 93 (2): 588–592. doi:10.1021/j100339a019. ISSN 0022-3654. https://pubs.acs.org/doi/abs/10.1021/j100339a019.
- ↑ Jump up to: 19.0 19.1 19.2 Politzer, Peter; Seminario, Jorge M. (January 1990). "Relative bond strengths in tetrahedrane, prismane, and some of their aza analogs" (in en). Structural Chemistry 1 (1): 29–32. doi:10.1007/BF00675781. ISSN 1040-0400. http://link.springer.com/10.1007/BF00675781.
- ↑ Seminario, Jorge M.; Politzer, P. (June 1989). "Analysis of different computational treatments of highly strained molecules" (in en). Chemical Physics Letters 159 (1): 27–31. doi:10.1016/S0009-2614(89)87447-0. https://linkinghub.elsevier.com/retrieve/pii/S0009261489874470.
- ↑ Politzer, Peter; Seminario, Jorge M. (June 1989). "Computational analysis of the structures, bond properties, and electrostatic potentials of some nitrotetrahedranes and nitroazatetrahedranes" (in en). The Journal of Physical Chemistry 93 (12): 4742–4745. doi:10.1021/j100349a013. ISSN 0022-3654. https://pubs.acs.org/doi/abs/10.1021/j100349a013.
- ↑ Jump up to: 22.0 22.1 Rayne, Sierra; Forest, Kaya (2011-08-29). "Theoretical studies in the molecular Platonic solids: Pure and mixed carbon, nitrogen, phosphorus, and silicon tetrahedranes" (in en). Nature Precedings. doi:10.1038/npre.2011.6299.1. ISSN 1756-0357. https://www.nature.com/articles/npre.2011.6299.1.
- ↑ Jump up to: 23.0 23.1 Murray, Jane S.; Seminario, Jorge M.; Lane, Pat; Politzer, Peter (June 1990). "Anomalous energy effects associated with the presence of aza nitrogens and nitro substituents in some strained systems" (in en). Journal of Molecular Structure 207 (3-4): 193–200. doi:10.1016/0166-1280(90)85023-G. https://linkinghub.elsevier.com/retrieve/pii/016612809085023G.
- ↑ Jursic, B.S. (February 2001). "Structures and properties of nitrogen derivatives of tetrahedrane" (in en). Journal of Molecular Structure 536 (2-3): 143–154. doi:10.1016/S0166-1280(00)00619-9. https://linkinghub.elsevier.com/retrieve/pii/S0166128000006199.
- ↑ Del Angel Cruz, Daniel; Galvez Vallejo, Jorge L.; Gordon, Mark S. (2023). "Analysis of the bonding in tetrahedrane and phosphorus-substituted tetrahedranes" (in en). Physical Chemistry Chemical Physics 25 (40): 27276–27292. doi:10.1039/D3CP03619G. ISSN 1463-9076. http://xlink.rsc.org/?DOI=D3CP03619G.
- ↑ Riu, Martin-Louis Y.; Bistoni, Giovanni; Cummins, Christopher C. (2021-07-22). "Understanding the Nature and Properties of Hydrogen–Hydrogen Bonds: The Stability of a Bulky Phosphatetrahedrane as a Case Study" (in en). The Journal of Physical Chemistry A 125 (28): 6151–6157. doi:10.1021/acs.jpca.1c04046. ISSN 1089-5639. https://pubs.acs.org/doi/10.1021/acs.jpca.1c04046.
- ↑ Jump up to: 27.0 27.1 Wolf, Mark E.; Doty, Elizabeth A.; Turney, Justin M.; Schaefer III, Henry F. (2021-04-01). "Highly Strained Pn(CH) 3 (Pn = N, P, As, Sb, Bi) Tetrahedranes: Theoretical Characterization" (in en). The Journal of Physical Chemistry A 125 (12): 2612–2621. doi:10.1021/acs.jpca.1c01022. ISSN 1089-5639. https://pubs.acs.org/doi/10.1021/acs.jpca.1c01022.
 |