Chemistry:Prehnitene
From HandWiki
Short description: Organic compound
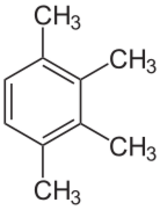
| |
| Names | |
|---|---|
| Preferred IUPAC name
1,2,3,4-Tetramethylbenzene | |
| Other names
Prehnitene
| |
| Identifiers | |
3D model (JSmol)
|
|
| 1904390 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
| 101866 | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C10H14 | |
| Molar mass | 134.22 |
| Appearance | colorless liquid |
| Density | 0.90 g/cm3 |
| Melting point | −6.2 °C (20.8 °F; 266.9 K) |
| Boiling point | 205 °C (401 °F; 478 K) |
| 33.9 mg/l | |
| Hazards | |
| Main hazards | Flammable |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+352, P304+340, P305+351+338, P312, P321, P332+313, P337+313, P362, P403+233, P405, P501 | |
| Flash point | 68.3 °C (154.9 °F; 341.4 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Prehnitene or 1,2,3,4-tetramethylbenzene is an organic compound with the formula C6H2(CH3)4, classified as an aromatic hydrocarbon. It is a flammable colorless liquid which is nearly insoluble in water but soluble in organic solvents. It occurs naturally in coal tar. Prehnitene is one of three isomers of tetramethylbenzene, the other two being isodurene (1,2,3,5-tetramethylbenzene) and durene (1,2,4,5-tetramethylbenzene).[1] It is a relatively easily oxidized benzene derivative, with E1/2 of 2.0 V vs NHE.[2]
Production
Industrially, prehnitene can be isolated from the reformed fraction of oil refineries. It may also be produced by methylation of toluene, xylenes and the trimethylbenzenes hemimellitene and pseudocumene.[1]
References
- ↑ 1.0 1.1 Griesbaum, Karl; Behr, Arno; Biedenkapp, Dieter; Voges, Heinz-Werner; Garbe, Dorothea; Paetz, Christian; Collin, Gerd; Mayer, Dieter et al. (2002). "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a13_227.
- ↑ Howell, J. O.; Goncalves, J. M.; Amatore, C.; Klasinc, L.; Wightman, R. M.; Kochi, J. K. (1984). "Electron transfer from aromatic hydrocarbons and their pi-complexes with metals. Comparison of the standard oxidation potentials and vertical ionization potentials". Journal of the American Chemical Society 106 (14): 3968–3976. doi:10.1021/ja00326a014.
 |
