Chemistry:Silver hypochlorite
From HandWiki
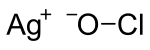
| |
| Names | |
|---|---|
| IUPAC name
Silver(I) hypochlorite
| |
| Other names
Argentous hypochlorite
Silver hypochlorite | |
| Identifiers | |
3D model (JSmol)
|
|
PubChem CID
|
|
| |
| |
| Properties | |
| AgOCl | |
| Molar mass | 159.32 g·mol−1 |
| very soluble | |
| Related compounds | |
Other anions
|
|
Other cations
|
|
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Silver hypochlorite is a chemical compound with the chemical formula AgOCl (also written as AgClO). It is an ionic compound of silver and the polyatomic ion hypochlorite.[1][2] The compound is very unstable and rapidly decomposes.[3] It is the silver(I) salt of hypochlorous acid. The salt consists of silver(I) cations (Ag+
) and hypochlorite anions (−
OCl).
Properties
Silver hypochlorite is very unstable, and its solution will soon disproportionate into silver chlorate and silver chloride:
- 3 AgOCl → AgClO
3 + 2 AgCl
Synthesis
Silver hypochlorite can be prepared by bubbling chlorine through an aqueous suspension of silver oxide:[4]
- 2 Cl
2 + Ag
2O + H
2O → 2 AgCl + 2 HOCl - 2 HOCl + Ag
2O → H
2O + 2 AgOCl
It can also be prepared by the reaction of hypochlorous acid with silver nitrate, producing silver hypochlorite and nitric acid.[5]
- HOCl + AgNO
3 → AgOCl + HNO
3
References
- ↑ Comey, Arthur Messinger (1896) (in en). A Dictionary of Chemical Solubilities; Inorganic. Macmillan and Company. p. 180. https://books.google.com/books?id=S5QMAQAAIAAJ&dq=Silver+hypochlorite&pg=PA180. Retrieved 10 March 2023.
- ↑ "silver hypochlorite" (in en). chemsrc.com. https://www.chemsrc.com/en/cas/475461-52-0_1274065.html.
- ↑ Massey, A. G.; Thompson, N. R.; Johnson, B. F. G. (6 June 2016) (in en). The Chemistry of Copper, Silver and Gold: Pergamon International Library of Science, Technology, Engineering and Social Studies. Elsevier. p. 108. ISBN 978-1-4831-8169-1. https://books.google.com/books?id=GuxPDAAAQBAJ&dq=Silver+hypochlorite&pg=PA108. Retrieved 10 March 2023.
- ↑ Stas, J. A. (1867). "On the Action of Chlorine on Carbonate of Silver" (in en). The Chemical News and Journal of Physical Science: A Journal of Practical Chemistry in All Its Applications to Pharmacy, Arts, and Manufacturers. American Reprint: 173. https://books.google.com/books?id=ePHNAAAAMAAJ&dq=Silver+hypochlorite&pg=PA173. Retrieved 10 March 2023.
- ↑ "Silver Hypochlorite: Formula, Solubility & Molar Mass". study.com. https://study.com/academy/lesson/silver-hypochlorite-formula-solubility-molar-mass.html.
 |
