Chemistry:Silver iodate
From HandWiki
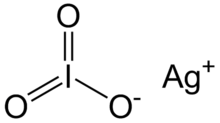
| |
| Names | |
|---|---|
| IUPAC name
Silver(I) iodate
| |
| Systematic IUPAC name
Silver(I) iodate(V) | |
| Other names
Argentous iodate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| AgIO3 | |
| Molar mass | 282.77 g/mol |
| Appearance | white crystals |
| Odor | odorless |
| Density | 5.525 g/cm3 |
| Melting point | ~200 °C |
| Boiling point | ~1150 °C |
| 0.003 g/100 mL (10 °C) 0.019 g/100 mL (50 °C) | |
Solubility product (Ksp)
|
3.17×10−8[1] |
| Solubility | soluble in ammonia |
| Structure | |
| orthorhombic | |
| Hazards | |
| GHS pictograms |  
|
| GHS Signal word | Danger |
| HH272Script error: No such module "Preview warning".Category:GHS errors, HH315Script error: No such module "Preview warning".Category:GHS errors, HH319Script error: No such module "Preview warning".Category:GHS errors, HH335Script error: No such module "Preview warning".Category:GHS errors | |
| PP210Script error: No such module "Preview warning".Category:GHS errors, PP220Script error: No such module "Preview warning".Category:GHS errors, PP261Script error: No such module "Preview warning".Category:GHS errors, PP264Script error: No such module "Preview warning".Category:GHS errors, PP264+P265Script error: No such module "Preview warning".Category:GHS errors, PP271Script error: No such module "Preview warning".Category:GHS errors, PP280Script error: No such module "Preview warning".Category:GHS errors, PP302+P352Script error: No such module "Preview warning".Category:GHS errors, PP304+P340Script error: No such module "Preview warning".Category:GHS errors, PP305+P351+P338Script error: No such module "Preview warning".Category:GHS errors, PP319Script error: No such module "Preview warning".Category:GHS errors, PP321Script error: No such module "Preview warning".Category:GHS errors, PP332+P317Script error: No such module "Preview warning".Category:GHS errors, PP337+P317Script error: No such module "Preview warning".Category:GHS errors, PP362+P364Script error: No such module "Preview warning".Category:GHS errors, PP370+P378Script error: No such module "Preview warning".Category:GHS errors, PP403+P233Script error: No such module "Preview warning".Category:GHS errors, PP405Script error: No such module "Preview warning".Category:GHS errors, PP501Script error: No such module "Preview warning".Category:GHS errors | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-combustable |
| Related compounds | |
Other anions
|
silver iodide silver chlorate |
Other cations
|
sodium iodate potassium iodate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Silver iodate (AgIO3) is a light-sensitive, white crystal composed of silver, iodine and oxygen. Unlike most metal iodates, it is practically insoluble in water.
Production
Silver iodate can be obtained by reacting silver nitrate (AgNO3) with sodium iodate or potassium iodate. The by-product of the reaction is sodium nitrate.[2]
Alternatively, it can be created by the action of iodine in a solution of silver oxide.
Uses
Silver iodate is used to detect traces of chlorides in blood.[3]
References
- ↑ John Rumble (June 18, 2018) (in English). CRC Handbook of Chemistry and Physics (99 ed.). CRC Press. pp. 5–189. ISBN 978-1138561632.
- ↑ Qiu, Chao; Sheng Han; Xingguo Cheng; Tianhui Ren (2005). "Distribution of Thioethers in Hydrotreated Transformer Base Oil by Oxidation and ICP-AES Analysis". Industrial & Engineering Chemistry Research 44 (11): 4151–4155. doi:10.1021/ie048833b. "Silver nitrate reacts with iodate to form the precipitate of silver iodate, and the precipitate is transferred to silver nitrate.".
- ↑ Rodkey, F Lee; Sendroy, Julius (1963-12-01). "Microdetermination of Chloride in Blood Plasma and Cells, by Spectrophotometric Analysis Using Solid Silver Iodate" (in en). Clinical Chemistry 9 (6): 668–681. doi:10.1093/clinchem/9.6.668. ISSN 0009-9147. https://academic.oup.com/clinchem/article/9/6/668/5672555.
 |

