Chemistry:Sulfonyl
From HandWiki
This article does not cite any external source. HandWiki requires at least one external source. See citing external sources. (June 2018) (Learn how and when to remove this template message) |
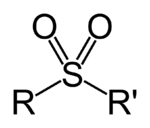
A sulfonyl group can refer either to a functional group found primarily in sulfones or to a substituent obtained from a sulfonic acid by the removal of the hydroxyl group similarly to acyl groups. Sulfonyl groups can be written as having the general formula R−S(=O)2−R′, where there are two double bonds between the sulfur and oxygen.
Sulfonyl groups can be reduced to the hydrocarbon with lithium aluminium hydride (LiAlH4).
In inorganic chemistry, when the group −S(=O)2− is not connected to any carbon atoms, it is referred to as sulfuryl.
Examples of sulfonyl group substituents
The names of sulfonyl groups typically end in -syl, such as:
Group name Full name Pseudoelement symbol Example Tosyl p-toluenesulfonyl Ts Tosyl chloride (p-toluenesulfonyl chloride)
CH3C6H4SO2ClBrosyl p-bromobenzenesulfonyl Bs Nosyl o- or p-nitrobenzenesulfonyl Ns Mesyl methanesulfonyl Ms Mesyl chloride (methanesulfonyl chloride)
CH3SO2ClTriflyl trifluoromethanesulfonyl Tf Tresyl 2,2,2-trifluoroethyl-1-sulfonyl Dansyl 5-(dimethylamino)naphthalene-1-sulfonyl Ds Dansyl chloride
See also
References
