Chemistry:Nitroso
In organic chemistry, nitroso refers to a functional group in which the nitric oxide (–N=O) group is attached to an organic moiety. As such, various nitroso groups can be categorized as C-nitroso compounds (e.g., nitrosoalkanes; R–N=O), S-nitroso compounds (nitrosothiols; RS–N=O), N-nitroso compounds (e.g., nitrosamines, RN(–R’)–N=O), and O-nitroso compounds (alkyl nitrites; RO–N=O).
Synthesis
Nitroso compounds can be prepared by the reduction of nitro compounds[1] or by the oxidation of hydroxylamines.[2] Ortho-nitrosophenols may be produced by the Baudisch reaction. In the Fischer–Hepp rearrangement, aromatic 4-nitrosoanilines are prepared from the corresponding nitrosamines.
Properties
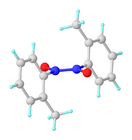
Nitrosoarenes typically participate in a monomer–dimer equilibrium. The azobenzene N,N'-dioxide (Ar(–O)N+=+N(O–)Ar) dimers, which are often pale yellow, are generally favored in the solid state, whereas the deep-green monomers are favored in dilute solution or at higher temperatures. They exist as cis and trans isomers.[4] The central "double bond" in the dimer in fact has a bond order of about 1.5.[5]
When stored in protic media, primary and secondary nitrosoalkanes isomerize to oximes.[6] Some tertiary nitrosoalkanes also isomerize to oximes through C-C bond fission, particularly if the bond is electron-poor.[7] Nitrosophenols and naphthols isomerize to the oxime quinone in solution, but reversibly; nitrosophenol ethers typically dealkylate to facilitate the isomerization. Nitroso tertiary anilines generally do not dealkylate in that way.[8]
Due to the stability of the nitric oxide free radical, nitroso organyls tend to have very low C–N bond dissociation energies: nitrosoalkanes have BDEs on the order of 30–40 kcal/mol (130–170 kJ/mol), while nitrosoarenes have BDEs on the order of 50–60 kcal/mol (210–250 kJ/mol). As a consequence, they are generally heat- and light-sensitive. Compounds containing O–(NO) or N–(NO) bonds generally have even lower bond dissociation energies. For instance, N-nitrosodiphenylamine, Ph2N–N=O, has a N–N bond dissociation energy of only 23 kcal/mol (96 kJ/mol).[9]
Organonitroso compounds serve as a ligands giving transition metal nitroso complexes.[10]
Reactions
Many reactions make use of an intermediate nitroso compound, such as the Barton reaction and Davis–Beirut reaction, as well as the synthesis of indoles, for example: Baeyer–Emmerling indole synthesis, Bartoli indole synthesis. In the Saville reaction, mercury is used to replace a nitrosyl from a thiol group.
C-nitroso compounds are used in organic synthesis as synthons in some well-documented chemical reactions such as hetero Diels-Alder (HDA), nitroso-ene and nitroso-aldol reactions.[11]
Nitrosyl in inorganic chemistry

Nitrosyls are non-organic compounds containing the NO group, for example directly bound to the metal via the N atom, giving a metal–NO moiety. Alternatively, a nonmetal example is the common reagent nitrosyl chloride (Cl–N=O). Nitric oxide is a stable radical, having an unpaired electron. Reduction of nitric oxide gives the nitrosyl anion, NO−
:
- NO + e−
→ NO−
Oxidation of NO yields the nitrosonium cation, NO+
:
- NO → NO+
+ e−
Nitric oxide can serve as a ligand forming metal nitrosyl complexes or just metal nitrosyls. These complexes can be viewed as adducts of NO+
, NO−
, or some intermediate case.
In human health
Nitrosamine formation during digestion
See also
- Nitrosamine, the functional group with the NO attached to an amine, such as R2N–NO
- Nitrosobenzene
- Nitric oxide
- Nitroxyl
References
- ↑ G. H. Coleman; C. M. McCloskey; F. A. Stuart (1945). "Nitrosobenzene". Org. Synth. 25: 80. doi:10.15227/orgsyn.025.0080.
- ↑ Calder, A.; Forrester, A. R.; Hepburn, S. P.. "2-Methyl-2-nitrosopropane and Its Dimer". Organic Syntheses 52: 77. http://www.orgsyn.org/demo.aspx?prep=cv6p0803.; Collective Volume, 6, pp. 803
- ↑ E.Bosch (2014). "Structural Analysis of Methyl-Substituted Nitrosobenzenes and Nitrosoanisoles". J. Chem. Cryst. 98 (2): 44. doi:10.1007/s10870-013-0489-8.
- ↑ Beaudoin, D.; Wuest, J. D. (2016). "Dimerization of Aromatic C-Nitroso Compounds". Chemical Reviews 116 (1): 258–286. doi:10.1021/cr500520s. PMID 26730505.
- ↑ Williams, D. L. H. (1988). Nitrosation. Cambridge, UK: Cambridge University. p. 36. ISBN 0-521-26796-X. https://archive.org/details/nitrosation0000will.
- ↑ Kirby, G. W. (1977). "Electrophilic C-nitroso-compounds". Chemical Society Reviews 6: 2. doi:10.1039/CS9770600001none (Tilden lecture).
- ↑ Williams 1988, p. 36.
- ↑ Williams 1988, pp. 59–61.
- ↑ Luo, Yu-Ran (2007). Comprehensive Handbook of Chemical Bond Energies. Boca Raton, FL: Taylor and Francis. ISBN 9781420007282.
- ↑ Lee, Jonghyuk; Chen, Li; West, Ann H.; Richter-Addo, George B. (2002). "Interactions of Organic Nitroso Compounds with Metals". Chemical Reviews 102 (4): 1019–1066. doi:10.1021/cr0000731. PMID 11942786.
- ↑ Bianchi, P.; Monbaliu, J. C. M. (2022). "Three decades of unveiling the complex chemistry of C-nitroso species with computational chemistry". Organic Chemistry Frontiers 9: 223–264. doi:10.1039/d1qo01415c.
 |
