Chemistry:Syringetin
From HandWiki
| |
| Names | |
|---|---|
| IUPAC name
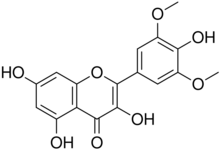
3,4′,5,7-Tetrahydroxy-3′,5′-dimethoxyflavone
| |
| Systematic IUPAC name
3,5,7-Trihydroxy-2-(4-hydroxy-3,5-dimethoxyphenyl)-4H-1-benzopyran-4-one | |
| Other names
3′,5′-O-Dimethylmyricetin
3′,5′-Dimethoxy-3,5,7,4′-tetrahydroxyflavone 3,5,7,4′-Tetrahydroxy-3′,5′-dimethoxyflavone | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C17H14O8 | |
| Molar mass | 346.291 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Syringetin is an O-methylated flavonol, a type of flavonoid. It is found in red grape (absent in white grape),[1] in Lysimachia congestiflora[2] and in Vaccinium uliginosum (bog billberries).[3] It is one of the phenolic compounds present in wine.[4]
It induces human osteoblast differentiation through bone morphogenetic protein-2/extracellular signal-regulated kinase 1/2 pathway.[4]
Metabolism
Syringetin is formed from laricitrin by the action of the enzyme laricitrin 5′-O-methyltransferase[1][5] (myricetin O-methyltransferase).[6]
Glycosides
- Syringetin-3-O-galactoside[1][7]
- Syringetin-3-O-glucoside[8][9]
- Syringetin 3-rhamnoside (CAS number 93126-00-2)
- Syringetin-3-O-rutinoside[9] found in Larix sibirica[10]
- Syringetin 3-O-(6′′-acetyl)-β-glucopyranoside found in Picea abies (Norway spruce)[11]
References
- ↑ 1.0 1.1 1.2 Mattivi, Fulvio; Guzzon, Raffaele; Vrhovsek, Urska; Stefanini, Marco; Velasco, Riccardo (2006). "Metabolite profiling of grape: Flavonols and anthocyanins". Journal of Agricultural and Food Chemistry 54 (20): 7692–7702. doi:10.1021/jf061538c. PMID 17002441.
- ↑ Guo, Jian; Yu, Dong-Lei; Xu, Lizhen; Zhu, Min; Yang, Shi-Lin (1998). "Flavonol glycosides from Lysimachia congestiflora". Phytochemistry 48 (8): 1445–1447. doi:10.1016/s0031-9422(97)01025-x.
- ↑ Lätti, Anja K.; Jaakola, Laura; Riihinen, Kaisu R.; Kainulainen, Pirjo S. (2010). "Anthocyanin and flavonol variation in bog bilberries (Vaccinium uliginosum L.) in Finland". Journal of Agricultural and Food Chemistry 58 (1): 427–433. doi:10.1021/jf903033m. PMID 20000402.
- ↑ 4.0 4.1 Hsu, Ya-Ling; Liang, Hsin-Lin; Hung, Chih-Hsing; Kuo, Po-Lin (2009). "Syringetin, a flavonoid derivative in grape and wine, induces human osteoblast differentiation through bone morphogenetic protein-2/extracellular signal-regulated kinase 1/2 pathway". Molecular Nutrition & Food Research 53 (11): 1452–1461. doi:10.1002/mnfr.200800483. PMID 19784998.
- ↑ "Laricitrin 5′-O-methyltransferase activity". Gene Ontology Consortium. 2009-02-28. http://amigo.geneontology.org/amigo/term/GO:0070448.
- ↑ Foerster, Hartmut (2006-11-03). "MetaCyc pathway: Syringetin biosynthesis". SRI International. https://metacyc.org/META/NEW-IMAGE?object=PWY-5391.
- ↑ Matsuda, F.; Suzuki, M.; Sawada, Y. (2016-01-19). "Syringetin-3-O-galactoside; LC-ESI-QTOF; MS2; CE:Ramp 5-60 V; [M+H+"]. http://www.massbank.jp/RecordDisplay?id=PR100366.
- ↑ Tohge, T. (2016-01-19). "Syringetin-3-O-glucoside; LC-ESI-QTOF; MS". http://www.massbank.jp/RecordDisplay?id=PR020084.
- ↑ 9.0 9.1 Slimestad, Rune; Hostettmann, Kurt (1996). "Characterisation of phenolic constituents from juvenile and mature needles of Norway spruce by means of high performance liquid chromatography–mass spectrometry". Phytochemical Analysis 7 (1): 42–48. doi:10.1002/(SICI)1099-1565(199601)7:1<42::AID-PCA282>3.0.CO;2-K.
- ↑ Tyukavkina, N. A.; Medvedeva, S. A.; Ivanova, S. Z. (1974). "New flavonol glycosides from the needles of Larix sibirica". Chemistry of Natural Compounds 10 (2): 170–172. doi:10.1007/BF00563605.
- ↑ Slimestad, Rune; Andersen, Øyvind M.; Francis, George W.; Marston, Andrew; Hostettmann, Kurt (1995). "Syringetin 3-O-(6′′-acetyl)-β-glucopyranoside and other flavonols from needles of Norway spruce, Picea abies". Phytochemistry 40 (5): 1537–1542. doi:10.1016/0031-9422(95)00383-I.
 |
