Chemistry:Centaureidin
From HandWiki
| |
| Names | |
|---|---|
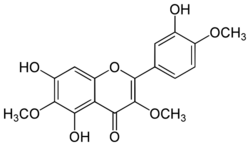
| IUPAC name
3′,5,7-Trihydroxy-3,4′,6-trimethoxyflavone
| |
| Systematic IUPAC name
5,7-Dihydroxy-2-(3-hydroxy-4-methoxyphenyl)-3,6-dimethoxy-4H-1-benzopyran-4-one | |
| Other names
Desmethoxycentaureidine
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C18H16O8 | |
| Molar mass | 360.31 g/mol |
| Density | 1.542 g/mL |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Centaureidin is an O-methylated flavonol. It can be isolated from Tanacetum microphyllum,[1] Achillea millefolium,[2] Brickellia veronicaefolia, Bidens pilosa[3] and Polymnia fruticosa.[4]
References
- ↑ Abad, Maria Jose; Bermejo, Paulina; Villar, Angel (1995). "The activity of flavonoids extracted from Tanacetum microphyllum DC. (Compositae) on soybean lipoxygenase and prostaglandin synthetase". General Pharmacology: The Vascular System 26 (4): 815–9. doi:10.1016/0306-3623(94)00242-F. PMID 7635257.
- ↑ "Assessment Report on Achillea Millefolium L., Herba". 15 September 2010. https://www.ema.europa.eu/documents/herbal-report/draft-assessment-report-achillea-millefolium-l-herba_en.pdf.
- ↑ Chang, Shu-Lin; Chiang, Yi-Ming; Chang, Cicero Lee-Tian; Yeh, Hsu-Hua; Shyur, Lie-Fen; Kuo, Yueh-Hsiung; Wu, Tung-Kung; Yang, Wen-Chin (2007). "Flavonoids, centaurein and centaureidin, from Bidens pilosa, stimulate IFN-γ expression". Journal of Ethnopharmacology 112 (2): 232–6. doi:10.1016/j.jep.2007.03.001. PMID 17408892.
- ↑ Beutler, John A.; Cardellina, John H.; Lin, Chii M.; Hamel, Ernest; Cragg, Gordon M.; Boyd, Michael R. (1993). "Centaureidin, a cytotoxic flavone from Polymnia fruticosa, inhibits tubulin polymerization". Bioorganic & Medicinal Chemistry Letters 3 (4): 581–4. doi:10.1016/S0960-894X(01)81233-6.
 |
