Chemistry:Telcagepant
 | |
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | 5–8 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
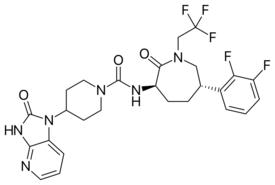
| Formula | C26H27F5N6O3 |
| Molar mass | 566.533 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Telcagepant (INN) (code name MK-0974) is a calcitonin gene-related peptide receptor antagonist which was an investigational drug for the acute treatment and prevention of migraine, developed by Merck & Co.[1]
In the acute treatment of migraine, it was found to have equal efficacy to rizatriptan[2] and zolmitriptan.[3]
A Phase IIa clinical trial studying telcagepant for the prophylaxis of episodic migraine was stopped on March 26, 2009, after the "identification of two patients with significant elevations in serum transaminases".[4] A memo to study locations stated that telcagepant had preliminarily been reported to increase the hepatic liver enzyme alanine transaminase (ALT) levels in "11 out of 660 randomized (double-blinded) study participants." All study participants were told to stop taking the medication.[5]
In July 2011, Merck announced that it had discontinued development of telcagepant.[6]
See also
References
- ↑ Molecule of the Month February 2009
- ↑ "Randomized controlled trial of an oral CGRP receptor antagonist, MK-0974, in acute treatment of migraine". Neurology 70 (16): 1304–12. April 2008. doi:10.1212/01.WNL.0000286940.29755.61. PMID 17914062.
- ↑ "Efficacy and tolerability of MK-0974 (telcagepant), a new oral antagonist of calcitonin gene-related peptide receptor, compared with zolmitriptan for acute migraine: a randomised, placebo-controlled, parallel-treatment trial". Lancet 372 (9656): 2115–23. December 2008. doi:10.1016/S0140-6736(08)61626-8. PMID 19036425.
- ↑ Clinical trial number NCT00797667 for "MK0974 for Migraine Prophylaxis in Patients With Episodic Migraine" at ClinicalTrials.gov
- ↑ Merck & Co.: Memo to all US study locations involved in protocol MK0974-049
- ↑ "Press release: Merck Announces Second Quarter 2011 Financial Results". Merck. July 29, 2011. http://www.mercknewsroom.com/press-release/financial-news/merck-announces-second-quarter-2011-financial-results.
 |
