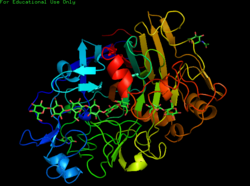
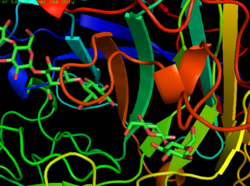
Biology:Cellulose 1,4-β-cellobiosidase (non-reducing end)
| Cellulose 1,4-β-cellobiosidase (non-reducing end) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
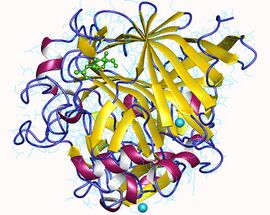 Cellobiohydrolase monomer, Trichoderma reesei | |||||||||
| Identifiers | |||||||||
| EC number | 3.2.1.91 | ||||||||
| CAS number | 37329-65-0 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
Cellulose 1,4-β-cellobiosidase (EC 3.2.1.91, exo-cellobiohydrolase, β-1,4-glucan cellobiohydrolase, β-1,4-glucan cellobiosylhydrolase, 1,4-β-glucan cellobiosidase, exoglucanase, avicelase, CBH 1, C1 cellulase, cellobiohydrolase I, cellobiohydrolase, exo-β-1,4-glucan cellobiohydrolase, 1,4-β-D-glucan cellobiohydrolase, cellobiosidase) is an enzyme of interest for its capability of converting cellulose to useful chemicals, particularly cellulosic ethanol.
The main technological impediment to widespread utilization of cellulose for fuels is still the lack of low-cost technologies to convert cellulose.[1] One solution is the use of organisms that are capable of performing this conversion.[1] Development of such organisms, such as Saccharomyces cerevisiae which is capable of secreting high levels of cellobiohydrolases, is already underway.[1] Cellobiohydrolases are exoglucanases derived from fungi.
The systematic name is 4-β-D-glucan cellobiohydrolase (non-reducing end).[2][3][4][5]
Function
This enzyme catalyses the hydrolysis of (1→4)-β-D-glucosidic linkages in cellulose and cellotetraose, releasing cellobiose from the non-reducing ends of the chains.
CBH1 from yeast, for example, is composed of a carbohydrate binding site, a linker region and a catalytic domain.[6] Once the cellulose chain is bound, it is strung through a tunnel-shaped active site where the cellulose is broken down into two-sugar segments called cellobiose.[6][7] The structure of the enzyme can be seen in the first figure. The second figure shows the activity of the enzyme, and shows both cellulose binding to the enzyme, as well as the product of this step, cellobiose. Research suggests, however, that the activity of CBH1 is very strong inhibited by the product, cellobiose. Determination of an enzyme that is not as strongly inhibited by the product or finding a way to remove cellobiose from the environment of the enzyme are just more examples of the many challenges that face the use of these enzymes for the creation of biofuels.[8]
After above step, the process for creating ethanol is as follows:[9] 3. Separation of sugars from other plant material. 4. Microbial fermentation of the sugar solution to create alcohol. 5. Distillation to purify the products and produce roughly 9% pure alcohol 6. Further purification to bring the ethanol purity to roughly 99.5%
Some notable improvements have been made in this area as well. For example, a strain of yeast capable of producing its own cellulose digesting enzyme has been developed, which would allow the cellulose degradation and the fermentation steps could be at once.[10] This is an important development in the sense that it makes large scale, industrial applications more feasible.
References
- ↑ 1.0 1.1 1.2 "High level secretion of cellobiohydrolases by Saccharomyces cerevisiae". Biotechnology for Biofuels 4: 30. September 2011. doi:10.1186/1754-6834-4-30. PMID 21910902. PMC 3224389. http://www.biotechnologyforbiofuels.com/content/pdf/1754-6834-4-30.pdf.
- ↑ "The mechanism of enzymatic cellulose degradation. Purification of a cellulolytic enzyme from Trichoderma viride' active on highly ordered cellulose". European Journal of Biochemistry 37 (1): 21–30. August 1973. doi:10.1111/j.1432-1033.1973.tb02952.x. PMID 4738092.
- ↑ "Extracellular enzyme system utilized by the fungus Sporotrichum pulverulentum (Chrysosporium lignorum) for the breakdown of cellulose. 3. Purification and physico-chemical characterization of an exo-1,4-β-glucanase". European Journal of Biochemistry 51 (1): 213–8. February 1975. doi:10.1111/j.1432-1033.1975.tb03921.x. PMID 235428.
- ↑ "The role of component C 1 in cellulolytic systems". The Biochemical Journal 127 (2): 43P. April 1972. doi:10.1042/bj1270043Pa. PMID 5076675.
- ↑ "Multidomain structure and cellulosomal localization of the Clostridium thermocellum cellobiohydrolase CbhA". Journal of Bacteriology 180 (12): 3091–9. June 1998. doi:10.1128/JB.180.12.3091-3099.1998. PMID 9620957.
- ↑ 6.0 6.1 "Cellulase Enzymes for the Conversion of Biomass to Biofuels and Chemicals". Energy Innovation Portal. http://techportal.eere.energy.gov/technology.do/techID=171.
- ↑ "Enzymes for Alternative Energy Research". Sigma Aldrich. http://www.sigmaaldrich.com/life-science/metabolomics/enzyme-explorer/analytical-enzymes/enzymes-for-aer.html#cellulase_activities.
- ↑ "Determination of Product Inhibition of CBH1, CBH2, and EG1 Using a Novel Cellulase Activity Assay". Applied Biochemistry and Biotechnology 161 (1–8): 313–7. May 2010. doi:10.1007/s12010-009-8796-4. PMID 19830597.
- ↑ "Sulfite pretreatment (SPORL) for robust enzymatic saccharification of spruce and red pine". Bioresource Technology 100 (8): 2411–8. April 2009. doi:10.1016/j.biortech.2008.10.057. PMID 19119005.
- ↑ Galazka, Jonathan M.; Tian, Chaoguang; Beeson, William T.; Martinez, Bruno; Glass, N. Louise; Cate, Jamie H. D. (2010). "Cellodextrin Transport in Yeast for Improved Biofuel Production". Science 330 (6000): 84–86. doi:10.1126/science.1192838. PMID 20829451. Bibcode: 2010Sci...330...84G. https://www.science.org/doi/abs/10.1126/science.1192838.
External links
- Cellulose+1,4-beta-cellobiosidase+(non-reducing+end) at the US National Library of Medicine Medical Subject Headings (MeSH)
 |