Chemistry:Potassium thiosulfate
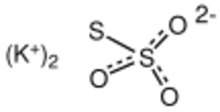
| |
| Names | |
|---|---|
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| K2S2O3 | |
| Molar mass | 190.32 g/mol |
| Appearance | White solid |
| Density | 2.37 g/cm3 |
| 96.1 g/100 ml (0 °C) 155.4 g/100 ml (20 °C) 165 g/100 ml (25 °C) 175.7 g/100 ml (30 °C) 204.7 g/100 ml (40 °C) 215.2 g/100 ml (50 °C) 238.3 g/100 ml (60 °C) 255.2 g/100 ml (70 °C) 293.1 g/100 ml (80 °C) 312 g/100 ml (90 °C)[1] | |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H315, H319 | |
| P264, P280, P302+352, P305+351+338, P321, P332+313, P337+313, P362 | |
| Related compounds | |
Other anions
|
Potassium sulfite; Potassium sulfate |
Other cations
|
Sodium thiosulfate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Potassium thiosulfate, commonly abbreviated KTS, is an inorganic compound with the formula K2S2O3. This salt can form multiple hydrates, such as the monohydrate, dihydrate, and the pentahydrate, all of which are white or colorless solids.[1] It is used as a fertilizer.
Formation and reactions
Thiosulfate salts are produced by the reaction of sulfite ion with elemental sulfur, and by incomplete oxidation of sulfides. For example, this salt is produced by reacting potassium hydroxide with ammonium hydroxide, sulfur dioxide, and elemental sulfur.[2] Thiosulfates are stable in neutral or alkaline solutions, but not in acidic solutions, due to disproportionation to sulfur dioxide and sulfur:[3]
- S2O2−3 + 2 H+ → SO2 + "S" + H2O
Due to this property, it can sequester metals, especially iron.[2]
Thiosulfate reacts with iodine to give tetrathionate, in this case potassium thiosulfate reacts with iodine to produce potassium tetrathionate and potassium iodide:
- 2 K2S2O3 + I2 → K2S4O6 + 2 KI
Thiosulfate extensively forms diverse complexes with transition metals. In the era of silver-based photography, thiosulfate was consumed on a large scale as a "stop" reagent. This application exploits thiosulfate's ability to dissolve silver halides. Thiosulfate is also used to extract or leach gold (sodium thiosulfate) and silver from their ores as a less toxic alternative to cyanide.[3]
Uses
Potassium thiosulfate is commonly used as a fertilizer alone or with urea and/or urea ammonium nitrate[4] due to its ability to delay nitrification.[2] It thus has the ability to reduce the emission of nitrous oxide.[5] It can also reduce the amount of fumigants being released from the soil.[6] If used alone it is used in very dilute solution due to its ability to cause phytotoxicity symptoms. This is caused by the elemental sulfur being oxidized to produce sulfuric acid.[4]
References
- ↑ 1.0 1.1 Atherton Seidell (1919) (in English). Solubilities of inorganic and organic compounds c. 2. D. Van Nostrand Company. p. 568. https://books.google.com/books?id=t4LSvgY7uIEC.
- ↑ 2.0 2.1 2.2 Sulewski, Gavin; Thompson, Michael; Mikkelsen, Robert et al., eds (2020) (in English) (Ebook). Improving Potassium Recommendations for Agricultural Crops. Springer International Publishing. p. 60. ISBN 978-3-030-59197-7. https://books.google.com/books?id=pfsOEAAAQBAJ. Retrieved 6 October 2021.
- ↑ 3.0 3.1 Barbera, J. J.; Metzger, A.; Wolf, M.. "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a25_477.
- ↑ 4.0 4.1 Western Plant Health Association (2018). Barlow, Dave; Pier, Jerome. eds (in English) (Ebook). Western Fertilizer Handbook (Third Horticulture ed.). Waveland Press. p. 186. ISBN 978-1-4786-3884-1. https://books.google.com/books?id=BHeCDwAAQBAJ. Retrieved 6 October 2021.
- ↑ Zejiang Cai; Suduan Gao; Minggang Xu; Bradley D Hanson (2017). "Evaluation of potassium thiosulfate as a nitrification inhibitor to reduce nitrous oxide emissions" (in English). Science of the Total Environment 618: 243–249. doi:10.1016/j.scitotenv.2017.10.274. PMID 29128773.
- ↑ Ruijun Qin; Suduan Gao; Jason A McDonald; Husein Ajwa; Shachar Shem-Tov; David A Sullivan (2008). "Effect of plastic tarps over raised-beds and potassium thiosulfate in furrows on chloropicrin emissions from drip fumigated fields" (in English). Chemosphere 72 (4): 558–563. doi:10.1016/j.chemosphere.2008.03.023. PMID 18440581. Bibcode: 2008Chmsp..72..558Q.
 |

