Chemistry:Potassium bisulfite
| |
| |
| Names | |
|---|---|
| IUPAC name
Potassium hydrogen sulfite
| |
| Other names
Potassium bisulfite, potassium bisulphite, monopotassium salt, monopotassium sulfite, potassium hydrosulfite
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
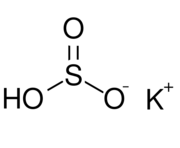
| KHSO3 | |
| Molar mass | 120.1561 g/mol |
| Appearance | White crystalline powder |
| Odor | SO2-like |
| Melting point | 190 °C (374 °F; 463 K) (decomposes) |
| 49 g/100 mL (20 °C) 115 g/100 mL (100 °C) | |
| Solubility | Insoluble in alcohol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Potassium bisulfite (or potassium hydrogen sulfite) is a chemical mixture with the approximate chemical formula KHSO3. Potassium bisulfite in fact is not a real compound,[1] but a mixture of salts that dissolve in water to give solutions composed of potassium ions and bisulfite ions. It is a white solid with an odor of sulfur dioxide. Attempts to crystallize potassium bisulfite yield potassium metabisulfite, K2S2O5.[2]
Potassium bisulfite is used as a sterilising agent in the production of alcoholic beverages.[3] This additive is classified as E number E228 under the current EU-approved food additive legislation.[4]
Production
It is made by the reaction of sulfur dioxide and potassium carbonate. The sulfur dioxide is passed through a solution of the potassium carbonate until no more carbon dioxide is evolved. The solution is concentrated.
See also
References
- ↑ Tudela, David; Jenkins, H. Donald B. (2003). "New Methods to Estimate Lattice Energies: Application to the Relative Stabilities of Bisulfite (HSO3−) and Metabisulfite (S2O52-) Salts". Journal of Chemical Education 80 (12): 1482. doi:10.1021/ed080p1482. Bibcode: 2003JChEd..80.1482T.
- ↑ Johnstone, H. F. (1946). "Sulfites and Pyrosulfites of the Alkali Metals". Inorganic Syntheses. Inorganic Syntheses. 2. pp. 162–167. doi:10.1002/9780470132333.ch49. ISBN 9780470132333.
- ↑ Barberá, José Jiménez; Metzger, Adolf; Wolf, Manfred (2000). "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a25_477.
- ↑ "Approved additives and E numbers" (in en). http://www.food.gov.uk/business-guidance/approved-additives-and-e-numbers.
 |
