Chemistry:Potassium ferrocyanide
| |
| |

| |
| Names | |
|---|---|
| IUPAC name
Potassium hexacyanidoferrate(II)
| |
| Other names | |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII |
|
| |
| |
| Properties | |
| K4[Fe(CN)6] | |
| Molar mass | 368.35 g/mol (anhydrous) 422.388 g/mol (trihydrate) |
| Appearance | Light yellow, crystalline granules |
| Density | 1.85 g/cm3 (trihydrate) |
| Boiling point | (decomposes) |
| trihydrate 28.9 g/100 mL (20 °C) | |
| Solubility | insoluble in ethanol, ether |
| −130.0·10−6 cm3/mol | |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H411 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
1.6—3.2g/kg |
| Related compounds | |
Other anions
|
Potassium ferricyanide |
Other cations
|
Sodium ferrocyanide Prussian blue |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
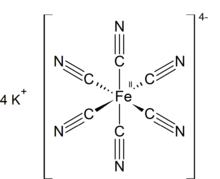
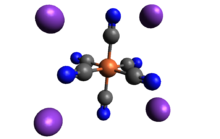
Potassium ferrocyanide is the inorganic compound with formula K4[Fe(CN)6]·3H2O. It is the potassium salt of the coordination complex [Fe(CN)6]4−. This salt forms lemon-yellow monoclinic crystals.
Synthesis
In 1752, the French chemist Pierre Joseph Macquer (1718–1784) first reported the preparation of potassium ferrocyanide, which he achieved by reacting Prussian blue (iron(III) ferrocyanide) with potassium hydroxide.[3][4]
Modern production
Potassium ferrocyanide is produced industrially from hydrogen cyanide, iron(II) chloride, and calcium hydroxide, the combination of which affords Ca2[Fe(CN)6]·11H2O. This solution is then treated with potassium salts to precipitate the mixed calcium-potassium salt CaK2[Fe(CN)6], which in turn is treated with potassium carbonate to give the tetrapotassium salt.[5]
Historical production
Historically, the compound was manufactured from nitrogenous organic material, iron filings, and potassium carbonate.[6] Common nitrogen and carbon sources were torrified horn, leather scrap, offal, or dried blood. It was also obtained commercially from gasworks spent oxide (purification of city gas from hydrogen cyanide).
Chemical reactions
Treatment of potassium ferrocyanide with nitric acid gives H2[Fe(NO)(CN)5]. After neutralization of this intermediate with sodium carbonate, red crystals of sodium nitroprusside can be selectively crystallized.[7]
Upon treatment with chlorine gas, potassium ferrocyanide converts to potassium ferricyanide:
- 2 K4[Fe(CN)6] + Cl2 → 2 K3[Fe(CN)6] + 2 KCl
A famous reaction involves treatment with ferric salts, most commonly iron(III) chloride, to give Prussian blue. In the reaction with iron(III) chloride, producing potassium chloride as a side-product:
3 K4[Fe(CN)6] + 4 FeCl3 → Fe4[Fe(CN)6]3 + 12 KCl
With the composition FeIII4[FeII(CN)6]3, this insoluble but deeply coloured material is the blue of blueprinting, as well as on many famous paintings such as The Great Wave off Kanagawa and The Starry Night.
Applications
Potassium ferrocyanide finds many niche applications in industry. It and the related sodium salt are widely used as anticaking agents for both road salt and table salt. The potassium and sodium ferrocyanides are also used in the purification of tin and the separation of copper from molybdenum ores. Potassium ferrocyanide is used in the production of wine and citric acid.[5]
In the EU, potassium ferrocyanides (E 535–538) were, as of 2017, solely authorised in two food categories as salt additives.
It can also be used in animal feed.[8]
In the laboratory, potassium ferrocyanide is used to determine the concentration of potassium permanganate, a compound often used in titrations based on redox reactions. Potassium ferrocyanide is used in a mixture with potassium ferricyanide and phosphate buffered solution to provide a buffer for beta-galactosidase, which is used to cleave X-Gal, giving a bright blue visualization where an antibody (or other molecule), conjugated to Beta-gal, has bonded to its target. On reacting with Fe(3) it gives a Prussian blue colour. Thus it is used as an identifying reagent for iron in labs.
Potassium ferrocyanide can be used as a fertilizer for plants.[2]
Prior to 1900, before the invention of the Castner process, potassium ferrocyanide was the most important source of alkali metal cyanides.[5] In this historical process, potassium cyanide was produced by decomposing potassium ferrocyanide:[6]
K4[Fe(CN)6] → 4 KCN + FeC2 + N2
Potassium ferrocyanide is a component of white gunpowder or Augendre's powder, a blasting powder of high energy.[9] It comprises 28 parts yellow potassium ferrocyanide, 23 parts cane sugar, and 49 parts of potassium chlorate, mixed under absolute alcohol to prevent premature detonation.[10][11]
Potassium Ferrocyanide is a main component of the traditional formula for the 19th century Cyanotype photographic process.[12]
Structure
Like other metal cyanides, solid potassium ferrocyanide, both as the hydrate and anhydrous salts, has a complicated polymeric structure. The polymer consists of octahedral [Fe(CN)6]4− centers crosslinked with K+ ions that are bound to the CN ligands.[13] The K+---NC linkages break when the solid is dissolved in water.[clarification needed]
Toxicity
The toxicity in rats is low, with lethal dose (LD50) at 1.6—3.2g/kg.[2] The kidneys are the organ for ferrocyanide toxicity.[14]
See also
References
- ↑ Five Hundred Useful and Amusing Experiments in Chemistry, and in the Arts and Manufactures: With Observations on the Properties Employed, and Their Application to Useful Purposes. Thomas Tegg. 1825. https://play.google.com/books/reader?printsec=frontcover&output=reader&id=gXwPAAAAYAAJ&pg=GBS.PA8.
- ↑ 2.0 2.1 2.2 Kopras, Elizabeth J. (2012). Bingham, Eula; Cohrssen, Barbara. eds. Patty's Toxicology. 1 (6 ed.). Hoboken, NJ: Wiley. p. 990. ISBN 978-0-470-41081-3.
- ↑ Macquer (1752). "Éxamen chymique de bleu de Prusse" (in fr). Histoire de l'Académie Royale des Sciences …, § Mémoires de l'Académie royale des Sciences: 60–77. https://www.biodiversitylibrary.org/item/88112#page/250/mode/1up. From pp. 63-64: "Après avoir essayé ainsi inutilement de décomposer le bleu de Prusse par les acides, … n'avoit plus qu'une couleur jaune un peu rousse." (After having tried so vainly to decompose Prussian blue by acids, I made recourse to alkalies. I put a half ounce of this [Prussian] blue in a flask, and I poured on it ten ounces of a solution of nitre fixed by tartar [i.e., potassium nitrate (nitre) which is mixed with crude cream of tartar and then ignited, producing potassium carbonate]. As soon as these two substances had been mixed together, I saw with astonishment that, without the aid of heat, the blue color had entirely disappeared; the powder [i.e., precipitate] at the bottom of the flask had only a rather gray color: having put this vessel on a sand bath in order to heat the solution until it simmered, this gray color also disappeared entirely, and all that was contained in the flask, both the powder [i.e., precipitate] and the solution, had only a yellow color [that was] a little red.)
- ↑ Munroe, Charles E.; Chatard, Thomas M. (1902). "Manufactures: Chemicals and Allied Products". Twelfth Census of the United States: Bulletins (210): 1–306. https://books.google.com/books?id=8UIUAQAAMAAJ&pg=RA1-PA31.; see p. 31.
- ↑ 5.0 5.1 5.2 Gail, E.; Gos, S.; Kulzer, R.; Lorösch, J.; Rubo, A.; Sauer, M.; Kellens, R.; Reddy, J. et al. (October 2011). "Cyano Compounds, Inorganic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a08_159.pub3. ISBN 978-3527306732.
- ↑ 6.0 6.1 Von Wagner, Rudolf (1897). Manual of chemical technology. New York: D. Appleton & Co.. p. 474 & 477. https://archive.org/details/manualofchemical00wagnuoft.
- ↑ Seel, F. (1965). "Sodium nitrosyl cyanoferrate". in Brauer, G.. Handbook of Preparative Inorganic Chemistry. 2 (2nd ed.). New York: Academic Press. p. 1768. https://www.scribd.com/doc/27443280/Handbook-of-Preparative-Inorganic-Chemistry-Vol-2-2d-Ed-George-Brauer. Retrieved 2017-09-10.
- ↑ "EuSalt Expert Meeting on E 535 and E 536 as Feed Additives". EUSalt. https://eusalt.com/events/eusalt-expert-meeting-e-535-and-e-536-feed-additives.
- ↑ Fay, Albert (1918). A Glossary of the Mining and Mineral Industry (1949 ed.). Washington, DC: United States Bureau of Mines. p. 741.
- ↑ (in German) Chemischen Technologie (1872 ed.). New York: D. Appleton & Company. p. 155.
- ↑ Brannt, William T. (William Theodore); Wahl, William H. (William Henry) (1919). The Techno-chemical receipt book. New York: Henry Carey Baird & Co.. p. 30. https://archive.org/details/technochemicalre00bran/page/30/mode/1up.
- ↑ Farber, Richard (1998). Historic Photographic Processes. New York: Allworth Press. p. 59. ISBN 1-880559-93-5. https://archive.org/details/aa100-HistoricPhotographicProcesses/page/n73/mode/2up?ref=ol&q=ferrocyanide.
- ↑ Willans, Mathew J.; Wasylishen, Roderick E.; McDonald, Robert (2009-05-18). "Polymorphism of Potassium Ferrocyanide Trihydrate as Studied by Solid-State Multinuclear NMR Spectroscopy and X-ray Diffraction" (in en). Inorganic Chemistry 48 (10): 4342–4353. doi:10.1021/ic802134j. ISSN 0020-1669. PMID 19425611. https://pubs.acs.org/doi/10.1021/ic802134j.
- ↑ Peter Aggett, Fernando Aguilar, Riccardo Crebelli, Birgit Dusemund, Metka Filipič, Maria Jose Frutos, Pierre Galtier, David Gott, Ursula Gundert-Remy, Gunter Georg Kuhnle, Claude Lambré, Jean-Charles Leblanc, Inger Therese Lillegaard, Peter Moldeus, Alicja Mortensen, Agneta Oskarsson, Ivan Stankovic, Ine Waalkens-Berendsen, Rudolf Antonius Woutersen, Matthew Wright and Maged Younes. (2018). "Re-evaluation of sodium ferrocyanide (E 535), potassium ferrocyanide (E 536) and calcium ferrocyanide (E 538) as food additives". EFSA Journal 16 (7): 5374. doi:10.2903/j.efsa.2018.5374. PMID 32626000. PMC 7009536. https://www.efsa.europa.eu/en/efsajournal/pub/5374.
External links
- "Cyanide (inorganic) compounds fact sheet". National Pollutant Inventory Australia. http://www.npi.gov.au/substances/cyanide/index.html.
- "Potassium Ferrocyanide in Salt Is Entirely Safe To Consume". rediff.com. http://blogs.rediff.com/thinko/2019/07/31/customers-can-rely-on-tata-salt-as-report-says-it-is-safe-to-consume/. * Photo of large potassium ferrocyanide crystals
 |

