Chemistry:Potassium chromate

| |
| |
| Names | |
|---|---|
| IUPAC name
Potassium chromate
| |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 3077 |
| |
| |
| Properties[1] | |
| K 2CrO 4 | |
| Molar mass | 194.189 g·mol−1 |
| Appearance | Yellow orthorhombic crystals |
| Odor | odorless |
| Density | 2.73 g/cm3 |
| Melting point | 975 °C (1,787 °F; 1,248 K) |
| 65.0 g/100 mL | |
| −3.9×10−6 cm3/mol | |
Refractive index (nD)
|
1.74[citation needed] |
| Structure | |
| |
| Hazards | |
| Safety data sheet | Fisher Scientific[2] |
| GHS pictograms |   
|
| GHS Signal word | Danger |
| H315, H317, H319, H335, H340, H350, H410 | |
| P201, P202, P261, P264, P271, P272, P273, P280, P302+352, P304+340+312Script error: No such module "Preview warning".Category:GHS errors, P305+351+338, P308+313, P333+313, P337+313, P362, P391, P403+233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Other anions
|
Potassium dichromate |
Other cations
|
|
Related chromates
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
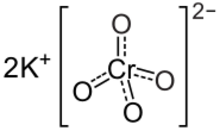
Potassium chromate is the inorganic compound with the formula K
2CrO
4. This yellow solid is the potassium salt of the chromate anion. It is a common laboratory chemical, whereas sodium chromate is important industrially.
Production and reactions
It is prepared by treating potassium dichromate with potassium hydroxide:[citation needed]
- K
2Cr
2O
7(aq) + 2 KOH → 2 K
2CrO
4 + H
2O
Or with potassium carbonate:[4]
- K
2Cr
2O
7 + K
2CO
3 → 2 K
2CrO
4 + CO
2
Or, the fusion of potassium hydroxide and chromium trioxide:[citation needed]
- 2 KOH + CrO
3 → K
2CrO
4 + H
2O
When treated with lead(II) nitrate, it gives an orange-yellow precipitate, lead(II) chromate.[citation needed]
Applications
Unlike the less expensive sodium salt, the potassium salt is mainly used for laboratory work in situations where an anhydrous salt is required, or as an oxidizing agent in organic synthesis.[5]
It is used in qualitative inorganic analysis, e.g. as a colorimetric test for silver ion. It is also used as an indicator in precipitation titrations with silver nitrate to measure levels of chloride ion (the Mohr method of determining chloride) since red silver chromate is precipitated in the presence of any excess of silver ions when potassium chromate is present. This titration proceeds by the following reactions, where silver nitrate is used as the titrant:[6]
- Ag+
+ Cl−
→ AgCl - 2 Ag+
+ CrO−
4 → Ag
2CrO
4
Structure
Two crystalline forms are known, both being very similar to the corresponding potassium sulfate. Orthorhombic β-K
2CrO
4 is the common form, but it converts to an α-form above 666 °C (1,231 °F).[5]
-
Structure of β-K
2CrO
4 -
Coordination sphere of one of two types of K+
site -
The environment about the tetrahedral CrO2−
4 center in β-K
2CrO
4
Safety
As with other Cr(VI) compounds, potassium chromate is carcinogenic. Positive associations with lung cancer at a very high rate, and nasal / sinus cancer at a 100x lower rate than lung cancer have been found using worker exposure data. In general, less soluble chromates are a larger chronic hazard as they can be encapsulated in the lung without being absorbed and excreted, giving more time for reactive intermediates to be produced. Animal data indicates a potential for impaired fertility, heritable genetic damage and harm to unborn children, along with other types of cancer via less common exposure routes.[7]
As a highly soluble hexavalent chromium compound, potassium chromate is also acutely toxic, though it is poorly absorbed from the intestinal tract.[7] The compound is also corrosive and exposure may produce severe eye damage or blindness.[2]
References
- ↑ Lide, David R., ed (2004). CRC Handbook of Chemistry and Physics (85th ed.). Boca Raton, Florida: CRC Press. pp. 4-76,4-147. ISBN 978-0-8493-0485-9.
- ↑ 2.0 2.1 2.2 "SDS - Potassium Chromate". Thermo Fisher Scientific. 29 March 2024. https://documents.thermofisher.com/DirectWebViewer/private/document.aspx?prd=ALFAA12610~~PDF~~MTR~~AGHS~~EN~~2024-03-30%2000:10:50~~Potassium%20chromate~~.
- ↑ Sigma-Aldrich Co., Potassium chromate.
- ↑ Fedoroff, Basil T.; Sheffield, Oliver E. (1 January 1966). "C - Chromates - Potassium Chromate". Encyclopedia of Explosives and Related Items (PDF) (Technical report). Vol. 3, Chlorides through Detonating Relays. Picatinny Arsenal, NJ: U.S. Army Armament Research Development And Engineering Center - TACOM, ARDEC - Warheads, Energetics And Combat Support Armaments Center. pp. C281-2. AD0653029, PATR 2700.
- ↑ 5.0 5.1 Anger, Gerd; Halstenberg, Jost; Hochgeschwender, Klaus; Ulrich Korallus, Christoph Scherhag; Knopf, Herbert; Schmidt, Peter; Ohlinger, Manfred. "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a07_067.
- ↑ Chloride, titrimetric, Mohr (Report). National Environment Methods Index (NEMI). I-1183-85. https://www.nemi.gov/methods/method_pdf/9597/.
- ↑ 7.0 7.1 Volume 100C: Arsenic, Metals, Fibres, and Dusts. Lyon, France: World Health Organization - International Agency for Research on Cancer. 2012. pp. 153–64. ISBN 978-92-832-0135-9. https://publications.iarc.who.int/_publications/media/download/6143/ef2dcba35d394362f6f5346d042bd48e5792ded3.pdf. Retrieved 2020-01-05.
Template:Chromates and dichromates
 |

