Biology:Archaellum
This article is missing information about high-resolution structures & tomography, locus regulation, N-glycosylation and metal binding, flexibility. (April 2019) |
The archaellum (pl.: archaella; formerly archaeal flagellum) is a unique structure on the cell surface of many archaea that allows for swimming motility. The archaellum consists of a rigid helical filament that is attached to the cell membrane by a molecular motor. This molecular motor – composed of cytosolic, membrane, and pseudo-periplasmic proteins – is responsible for the assembly of the filament and, once assembled, for its rotation. The rotation of the filament propels archaeal cells in liquid medium, in a manner similar to the propeller of a boat. The bacterial analog of the archaellum is the flagellum, which is also responsible for their swimming motility and can also be compared to a rotating corkscrew. Although the movement of archaella and flagella is sometimes described as "whip-like", this is incorrect, as only cilia from Eukaryotes move in this manner. Indeed, even "flagellum" (word derived from Latin meaning "whip") is a misnomer, as bacterial flagella also work as propeller-like structures.
Early studies on "archaeal flagella" identified several differences between archaella and flagella, although those differences were dismissed as a possible adaptation of archaella to the extreme ecological environments where archaea were at the time known to inhabit. When the first genomes of archaeal organisms were sequenced, it became obvious that archaea do not code for any of the proteins that are part of the flagellum, thus establishing that the motility system of archaea is fundamentally different from that of bacteria. In order to highlight the difference between these two organelles, the name archaellum was proposed in 2012 following studies that showed it to be evolutionarily and structurally different from the bacterial flagella and eukaryotic cilia.[1]
Archaella are evolutionarily and structurally related to type IV filament systems (TFF).[2] The TFF family seems to have originated in the last universal common ancestor, from where it diversified into archaella, Type IV Pili, Type II Secretion Systems, and the Tad pili.[3]
History
The first observations of what is now known to be the archaellum took possibly place more than 100 years ago, even before the identification of the archaea. Archaea were identified initially in 1977 by Carl Woese and George E. Fox,[4] and the three-domain of life (Eucarya, Archaea, and Bacteria) was proposed 10 years later.[5] Also during the 1970s it was suggested for the first time that the proteins that compose the archaellum filament are distinct from those that assemble into the flagella filament, although convincing data was still lacking. In the next decade, it became apparent that all archaella thus far studied possessed some "strange" features, such as the heavy presence of glycosylation in archaellins (that is, the monomers that form the archaella filament), a discovery helped by the cloning of the first archaellins.The "strangeness" of archaella was confirmed in the late 1990s, when the first genome sequences of archaeal species were published, namely those of Methanocaldococcus jannaschii in 1996, Archaeoglobus fulgidus in 1997, and Pyrococcus horikoshii in 1998.[6][7][8] Although genes of archaellins were identified all these three genomes, it was not possible to identify any gene homolog to those involved in flagellation. Besides the evidence that the archaellum is not related to the flagellum, it was also during this time that the similarities between archaella and type iv pili (T4P) became clearer.[9] One of the clearer evidences at the time was the observation that archaellins are synthesised in the cytoplasm as pre-proteins, with a signal peptide that needs to be cleaved prior to their insertion, presumably at the base of the growing archaellar filament. Flagellins, on the other hand, are not synthesised as pre-proteins. Rather, these proteins are synthesised in a mature state, and they travel to the lumen of the flagellar filament (which is therefore hollow) and assemble at its tip.[10] It was based on the similarities between archaella and type IV pili that in 1996 the first proposal for how archaella assemble was published.[11] The next decade saw significant advances in the understanding of archaella. The enzyme responsible for the cleavage of the signal peptide was identified, and so were the other genes thought to be part of the archaella operon (now arl cluster).[9][12] Interestingly, it was also during this period that the archaella from the euryarchaeon Halobacterium salinarum was shown to be powered by ATP hydrolysis.[13] Although the similarities between T4P and archaella suggested that ATP hydrolysis could power this organelle, this discovery identified another major difference between archaella and flagella, as the latter are powered by a cation pump.[14] This time also saw some of the initial research on chemotaxis in archaea, although the initial analyses of archaeal genomes had already suggested that these organisms possess a bacteria-like chemotaxis system. Interestingly, despite having different motors, archaea and bacteria have remarkably similar chemotactic machineries.[15]
During the 2010s, studies on the gene products of the arl operon allowed to establish the function of many of the "accessory" proteins of the archaellum, that is, proteins that compose the motor of this organelle. During this time it was possible to define a minimal set of components necessary for a mature and functional archaellum: the archaellin (either a single type or several), the prepilin peptidase which cleaves the signal peptide off from the pre-archaellin, and the proteins ArlC/D/E/F/G/H/I/J. In the phyla Crenarchaetoa the genes for the proteins ArlC/D/E are not found; instead archaellated members of this phylum code for ArlX, thought to have a similar function to ArlC/D/E.[16] Based on all the evidence that had accumulated on the unique nature of archaella, in 2012 Ken Jarrell and Sonja-Verena Albers proposed that this organelle should not be called "archaeal flagella", but rather "archaella".[1] Despite some initial criticism,[17][18] the name is now widely accepted in the scientific community, and as of 6 June 2021, a PubMed search for the terms "archaella" or "archaellum" retrieves more results in recent years than the terms "archaeal flagella" or "archaeal flagellum".
Research of archaella still continues, both related to the basic biology of this organelle, to its ecological roles, and even potential biotechnological applications. Some of the questions that remain open is how is the expression of the arl operon regulated, how does the archaellum motor complex look like, and what is the role of some of the accessory components of the archaellum.[9]
Structure
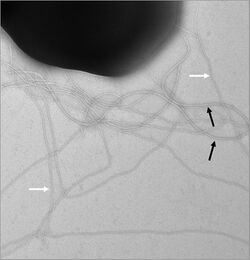
Components

Most proteins that make up the archaellum are encoded within one genetic locus. This genetic locus contains 7-13 genes which encode proteins involved in either assembly or function of the archaellum.[19] The genetic locus contains genes encoding archaellins (arlA and arlB) - the structural components of the filament - and motor components (arlI, arlJ, arlH). The locus furthermore encodes other accessory proteins (arlG, arlF, arlC, arlD, arlE, and arlX). ArlX is only found in Crenarchaeota and ArlCDE (which can exist as individual proteins or as fusion proteins) in Euryarchaeotes. ArlX and ArlCDE are thought to have similar functions, and an unknown protein is also thought to fulfil the same function in Thaumarchaeota.
The archaellum operon used to be historically known as fla (from "flagellum"), but in order to avoid confusion with the bacterial flagellum and to be consistent with the remaining nomenclature (archaellum, archaellins), it has been recently proposed to be renamed to arl (archaellin-related genes).[12] Consequently, the name of the genes is also different (e.g., flaJ is now arlJ). Therefore, in the specialised literature both nomenclatures can be found, with the arl nomenclature being increasingly more used since 2018.
Genetic analysis in different archaea revealed that each of these components is essential for assembly of the archaellum.[20][21][22][23][24] The prepilin peptidase (called PibD in crenarchaeota and ArlK (formerly FlaK) in euryarchaeota) is essential for the maturation of the archaellins and is generally encoded elsewhere on the chromosome.[25]
Functional characterization has been performed for ArlI, a Type II/IV secretion system ATPase super-family member[26] and PibD/ArlK.[25][27][28] ArlI forms a hexamer which hydrolyses ATP and most likely generates energy to assemble the archaellum and to power its rotation.[29][30] PibD cleaves the N-terminus of the archaellins before they can be assembled. ArlH (PDB: 2DR3) has a RecA-like fold and inactive ATPase domains.[31][32] This protein is a homolog of KaiC, a protein central for the regulation of the circadian rhythm in cyanobacteria. However, this function is not thought to be conserved; rather, ArlH also exhibits auto-phosphorylation which seems to modulate its interaction with the ATPase ArlI.[33] Despite arlH deletion resulting in loss of motiliy, rendering this protein essential for archaellation, its role in the archaellum motor remains unknown. ArlI and ArlH interact and, possibly together with the predicted membrane-protein ArlJ, form the central motor complex. In Crenarchaeota, this motor complex might be surrounded by a scaffold formed by a ring composed of ArlX.[34] In Euryarchaeotes, cryo-electron tomograms suggest that ArlCDE form a structure underneath the motor, possibly in the order (from top to bottom) ArlJ-ArlI-ArlH-ArlCDE.[35] ArlF and ArlG possibly form the stator of this complex, providing a static surface against which the rotor can move, and also anchoring the motor to the cell envelope, thus preventing the membrane from rupturing due to archaellar rotation.[36][37] The structure of ArlCDE is unknown, but this complex (or variations thereof) have been shown to link the chemotaxis machinery and the archaellum in Haloferax volcanii.[38]
Functional analogs
Despite the limited number of details presently available regarding the structure and assembly of archaellum, it has become increasingly evident from multiple studies that archaella play important roles in a variety of cellular processes in archaea. In spite of the structural dissimilarities with the bacterial flagellum, the main function thus far attributed for archaellum is swimming in liquid[24][39][40] and semi-solid surfaces.[41][42] Increasing biochemical and biophysical information has further consolidated the early observations of archaella mediated swimming in archaea. Like the bacterial flagellum,[43][44] the archaellum also mediates surface attachment and cell-cell communication.[45][46] However, unlike the bacterial flagellum archaellum has not shown to play a role in archaeal biofilm formation.[47] In archaeal biofilms, the only proposed function is thus far during the dispersal phase of biofilm when archaeal cells escape the community using their archaellum to further initiate the next round of biofilm formation. Also, archaellum have been found to be able to have a metal-binding site.[48]
References
- ↑ 1.0 1.1 "The archaellum: an old motility structure with a new name". Trends in Microbiology 20 (7): 307–12. July 2012. doi:10.1016/j.tim.2012.04.007. PMID 22613456.
- ↑ "Exceptionally widespread nanomachines composed of type IV pilins: the prokaryotic Swiss Army knives". FEMS Microbiology Reviews 39 (1): 134–54. January 2015. doi:10.1093/femsre/fuu001. PMID 25793961.
- ↑ "Diversification of the type IV filament superfamily into machines for adhesion, protein secretion, DNA uptake, and motility". PLOS Biology 17 (7): e3000390. July 2019. doi:10.1371/journal.pbio.3000390. PMID 31323028.
- ↑ "Phylogenetic structure of the prokaryotic domain: the primary kingdoms". Proceedings of the National Academy of Sciences of the United States of America 74 (11): 5088–90. November 1977. doi:10.1073/pnas.74.11.5088. PMID 270744. Bibcode: 1977PNAS...74.5088W.
- ↑ "Towards a natural system of organisms: proposal for the domains Archaea, Bacteria, and Eucarya". Proceedings of the National Academy of Sciences of the United States of America 87 (12): 4576–9. June 1990. doi:10.1073/pnas.87.12.4576. PMID 2112744. Bibcode: 1990PNAS...87.4576W.
- ↑ "Complete genome sequence of the methanogenic archaeon, Methanococcus jannaschii". Science 273 (5278): 1058–73. August 1996. doi:10.1126/science.273.5278.1058. PMID 8688087. Bibcode: 1996Sci...273.1058B.
- ↑ "The complete genome sequence of the hyperthermophilic, sulphate-reducing archaeon Archaeoglobus fulgidus". Nature 390 (6658): 364–70. November 1997. doi:10.1038/37052. PMID 9389475. Bibcode: 1997Natur.390..364K.
- ↑ "Complete sequence and gene organization of the genome of a hyper-thermophilic archaebacterium, Pyrococcus horikoshii OT3". DNA Research 5 (2): 55–76. April 1998. doi:10.1093/dnares/5.2.55. PMID 9679194.
- ↑ 9.0 9.1 9.2 "A comprehensive history of motility and Archaellation in Archaea". FEMS Microbes 2: xtab002. 2021-04-16. doi:10.1093/femsmc/xtab002. PMID 37334237.
- ↑ "Type III flagellar protein export and flagellar assembly". Biochimica et Biophysica Acta (BBA) - Molecular Cell Research 1694 (1–3): 207–17. November 2004. doi:10.1016/j.bbamcr.2004.04.005. PMID 15546667.
- ↑ "The archaeal flagellum: a unique motility structure". Journal of Bacteriology 178 (17): 5057–64. September 1996. doi:10.1128/jb.178.17.5057-5064.1996. PMID 8752319.
- ↑ 12.0 12.1 "Archaeal cell surface biogenesis". FEMS Microbiology Reviews 42 (5): 694–717. September 2018. doi:10.1093/femsre/fuy027. PMID 29912330.
- ↑ "Flagellar rotation in the archaeon Halobacterium salinarum depends on ATP". Journal of Molecular Biology 384 (1): 1–8. December 2008. doi:10.1016/j.jmb.2008.08.057. PMID 18786541.
- ↑ "Evolution of the Stator Elements of Rotary Prokaryote Motors". Journal of Bacteriology 202 (3). January 2020. doi:10.1128/JB.00557-19. PMID 31591272.
- ↑ "Taxis in archaea". Emerging Topics in Life Sciences 2 (4): 535–546. December 2018. doi:10.1042/ETLS20180089. PMID 33525831.
- ↑ 16.0 16.1 "The archaellum: how Archaea swim". Frontiers in Microbiology 6: 23. 27 January 2015. doi:10.3389/fmicb.2015.00023. PMID 25699024.
- ↑ "Response to Jarrell and Albers: the name says it all". Trends in Microbiology 20 (11): 512–3. November 2012. doi:10.1016/j.tim.2012.08.007. PMID 22944242.
- ↑ "Response to Jarrell and Albers: seven letters less does not say more". Trends in Microbiology 20 (11): 511–2. November 2012. doi:10.1016/j.tim.2012.07.007. PMID 22889944.
- ↑ "Assembly and function of the archaeal flagellum". Biochemical Society Transactions 39 (1): 64–9. January 2011. doi:10.1042/BST0390064. PMID 21265748.
- ↑ "The fla gene cluster is involved in the biogenesis of flagella in Halobacterium salinarum". Molecular Microbiology 41 (3): 653–63. August 2001. doi:10.1046/j.1365-2958.2001.02542.x. PMID 11532133.
- ↑ "The archaeal flagellum: a different kind of prokaryotic motility structure". FEMS Microbiology Reviews 25 (2): 147–74. April 2001. doi:10.1111/j.1574-6976.2001.tb00575.x. PMID 11250034.
- ↑ "Mutants in flaI and flaJ of the archaeon Methanococcus voltae are deficient in flagellum assembly". Molecular Microbiology 46 (3): 879–87. November 2002. doi:10.1046/j.1365-2958.2002.03220.x. PMID 12410843.
- ↑ "Systematic deletion analyses of the fla genes in the flagella operon identify several genes essential for proper assembly and function of flagella in the archaeon, Methanococcus maripaludis". Molecular Microbiology 66 (3): 596–609. November 2007. doi:10.1111/j.1365-2958.2007.05913.x. PMID 17887963.
- ↑ 24.0 24.1 "Molecular analysis of the crenarchaeal flagellum". Molecular Microbiology 83 (1): 110–24. January 2012. doi:10.1111/j.1365-2958.2011.07916.x. PMID 22081969.
- ↑ 25.0 25.1 "Cleavage of preflagellins by an aspartic acid signal peptidase is essential for flagellation in the archaeon Methanococcus voltae". Molecular Microbiology 50 (4): 1339–47. November 2003. doi:10.1046/j.1365-2958.2003.03758.x. PMID 14622420.
- ↑ "Archaeal flagellar ATPase motor shows ATP-dependent hexameric assembly and activity stimulation by specific lipid binding". The Biochemical Journal 437 (1): 43–52. July 2011. doi:10.1042/BJ20110410. PMID 21506936.
- ↑ "FlaK of the archaeon Methanococcus maripaludis possesses preflagellin peptidase activity". FEMS Microbiology Letters 208 (1): 53–9. February 2002. doi:10.1111/j.1574-6968.2002.tb11060.x. PMID 11934494.
- ↑ "Identification of diverse archaeal proteins with class III signal peptides cleaved by distinct archaeal prepilin peptidases". Journal of Bacteriology 189 (3): 772–8. February 2007. doi:10.1128/JB.01547-06. PMID 17114255.
- ↑ "Characterization of the ATPase FlaI of the motor complex of the Pyrococcus furiosus archaellum and its interactions between the ATP-binding protein FlaH". PeerJ 6: e4984. 2018-06-18. doi:10.7717/peerj.4984. PMID 29938130.
- ↑ "Insights into FlaI functions in archaeal motor assembly and motility from structures, conformations, and genetics". Molecular Cell 49 (6): 1069–82. March 2013. doi:10.1016/j.molcel.2013.01.014. PMID 23416110.
- ↑ "The nucleotide-dependent interaction of FlaH and FlaI is essential for assembly and function of the archaellum motor". Molecular Microbiology 99 (4): 674–85. February 2016. doi:10.1111/mmi.13260. PMID 26508112.
- ↑ "Crystal structure of the flagellar accessory protein FlaH of Methanocaldococcus jannaschii suggests a regulatory role in archaeal flagellum assembly". Protein Science 25 (6): 1147–55. June 2016. doi:10.1002/pro.2932. PMID 27060465.
- ↑ "Autophosphorylation of the KaiC-like protein ArlH inhibits oligomerisation and interaction with ArlI, the motor ATPase of the archaellum". Molecular Microbiology 116 (3): 943–956. July 2021. doi:10.1111/mmi.14781. PMID 34219289.
- ↑ "FlaX, a unique component of the crenarchaeal archaellum, forms oligomeric ring-shaped structures and interacts with the motor ATPase FlaI". The Journal of Biological Chemistry 287 (52): 43322–30. December 2012. doi:10.1074/jbc.M112.414383. PMID 23129770.
- ↑ "Structure and in situ organisation of the Pyrococcus furiosus archaellum machinery". eLife 6: e27470. June 2017. doi:10.7554/eLife.27470. PMID 28653905.
- ↑ "The structure of the periplasmic FlaG-FlaF complex and its essential role for archaellar swimming motility". Nature Microbiology 5 (1): 216–225. January 2020. doi:10.1038/s41564-019-0622-3. PMID 31844299.
- ↑ "FlaF Is a β-Sandwich Protein that Anchors the Archaellum in the Archaeal Cell Envelope by Binding the S-Layer Protein". Structure 23 (5): 863–872. May 2015. doi:10.1016/j.str.2015.03.001. PMID 25865246.
- ↑ "The switch complex ArlCDE connects the chemotaxis system and the archaellum". Molecular Microbiology 114 (3): 468–479. September 2020. doi:10.1111/mmi.14527. PMID 32416640.
- ↑ "Flagella and motility behaviour of square bacteria". The EMBO Journal 3 (12): 2899–903. December 1984. doi:10.1002/j.1460-2075.1984.tb02229.x. PMID 6526006.
- ↑ "Swimming behavior of selected species of Archaea". Applied and Environmental Microbiology 78 (6): 1670–4. March 2012. doi:10.1128/AEM.06723-11. PMID 22247169. Bibcode: 2012ApEnM..78.1670H.
- ↑ "Flagellar motility and structure in the hyperthermoacidophilic archaeon Sulfolobus solfataricus". Journal of Bacteriology 189 (11): 4305–9. June 2007. doi:10.1128/JB.00042-07. PMID 17416662.
- ↑ "Isolation and characterization of insertional mutations in flagellin genes in the archaeon Methanococcus voltae". Molecular Microbiology 20 (3): 657–66. May 1996. doi:10.1046/j.1365-2958.1996.5371058.x. PMID 8736544.
- ↑ "Bacterial surface translocation: a survey and a classification". Bacteriological Reviews 36 (4): 478–503. December 1972. doi:10.1128/MMBR.36.4.478-503.1972. PMID 4631369.
- ↑ "The surprisingly diverse ways that prokaryotes move". Nature Reviews. Microbiology 6 (6): 466–76. June 2008. doi:10.1038/nrmicro1900. PMID 18461074.
- ↑ "Flagella of Pyrococcus furiosus: multifunctional organelles, made for swimming, adhesion to various surfaces, and cell-cell contacts". Journal of Bacteriology 188 (19): 6915–23. October 2006. doi:10.1128/JB.00527-06. PMID 16980494.
- ↑ "Appendage-mediated surface adherence of Sulfolobus solfataricus". Journal of Bacteriology 192 (1): 104–10. January 2010. doi:10.1128/JB.01061-09. PMID 19854908.
- ↑ "Crenarchaeal biofilm formation under extreme conditions". PLOS ONE 5 (11): e14104. November 2010. doi:10.1371/journal.pone.0014104. PMID 21124788. Bibcode: 2010PLoSO...514104K.
- ↑ "High-resolution archaellum structure reveals a conserved metal-binding site". EMBO Reports 20 (5). May 2019. doi:10.15252/embr.201846340. PMID 30898768.
 |
