Chemistry:1-Octen-3-ol
| |
| Names | |
|---|---|
| Preferred IUPAC name
Oct-1-en-3-ol | |
| Other names
Amyl vinyl carbinol; 1-Vinylhexanol; Matsutake alcohol; Vinyl amyl carbinol; Vinyl hexanol; Matsuica alcohol; Mushroom alcohol; 3-Hydroxy-1-octene
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII |
|
| |
| |
| Properties | |
| C8H16O | |
| Molar mass | 128.215 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
1-Octen-3-ol, octenol for short and also known as mushroom alcohol,[1] is a chemical that attracts biting insects such as mosquitoes. It is contained in human breath and sweat, and it is believed that insect repellent DEET works by blocking the insects' octenol odorant receptors.[2][3][4]
The name “mushroom Alcohol” for 1-octen-3-ol comes from it first isolation by Murahashi in 1936 and 1938 from crushed matsutake mushrooms. [5][6]A recent study on volatiles of this mushroom has shown this compound is only produced upon tissue disruption.[7]
This alcohol is found in many other mushrooms where it may play a role as an antifeedant.[8]
Natural occurrence
Octenol is produced by several plants and fungi, including edible mushrooms and lemon balm. Octenol is formed during oxidative breakdown of linoleic acid.[9]
It is also a wine fault, defined as a cork taint, occurring in wines made with bunch rot contaminated grape.[10]
Properties
1-Octen-3-ol is a secondary alcohol derived from 1-octene. It exists in the form of two enantiomers, (R)-(−)-1-octen-3-ol and (S)-(+)-1-octen-3-ol.
Synthesis
Two possible lab syntheses of 1-octen-3-ol are:[11]
- by the Grignard reaction of acrolein and amyl iodide
- by the selective reduction of 1-octen-3-one
Biochemically, 1-octen-3-ol is generated from the peroxidation of linoleic acid, catalyzed by a lipoxygenase, followed by cleavage of the resulting hydroperoxide with the help of a hydroperoxide lyase. This reaction takes place in cheese and is used in biotechnology to produce the (R)-isomer.[12][13]
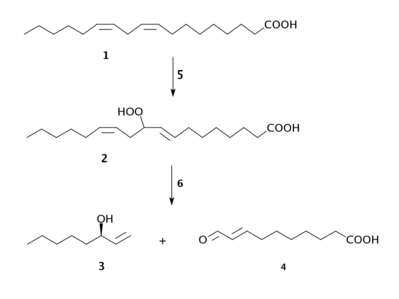
Uses
Octenol is used, sometimes in combination with carbon dioxide, to attract insects in order to kill them with certain electrical devices.[14]
The name 'mushroom alcohol' is used because octenol is the main flavor component of mushrooms.[15]
Health and safety
Octenol is approved by the U.S. Food and Drug Administration as a food additive.[16] It is of moderate toxicity with an LD50 of 340 mg/kg.[14]
In an animal study, octenol has been found to disrupt dopamine homeostasis and may be an environmental agent involved in parkinsonism.[17]
See also
- Olfactory receptor
- Oct-1-en-3-one, the ketone analog that gives blood on skin its typical metallic, mushroom-like smell[18]
- 1-Octen-3-yl acetate, the acetate ester of this compound
References
- ↑ "1-Octen-3-ol, Mushroom alcohol". http://www.hmdb.ca/metabolites/HMDB0031299.
- ↑ Petherick, Anna (2008-03-13). "How DEET jams insects' smell sensors". Nature News. doi:10.1038/news.2008.672. http://www.nature.com/news/2008/080313/full/news.2008.672.html.
- ↑ "Insect odorant receptors are molecular targets of the insect repellent DEET". Science 319 (5871): 1838–42. March 2008. doi:10.1126/science.1153121. PMID 18339904. Bibcode: 2008Sci...319.1838D.
- ↑ "Mosquitoes smell and avoid the insect repellent DEET". Proceedings of the National Academy of Sciences of the United States of America 105 (36): 13598–603. September 2008. doi:10.1073/pnas.0805312105. PMID 18711137.
- ↑ Murahashi, S.. "Sci. Pap. Inst. Phys. Chem. Res. (Jpn.) 34, 155". Chemical Absracts 31: 21617.
- ↑ Murahashi, S.. "Sci. Pap. Inst. Phys. Chem. Res. (Jpn.) 30, 263". Chemical Absracts 32: 27078.
- ↑ Wood W. F.; Lefevre C. K. (2007). "Changing volatile compounds from mycelium and sporocarp of American matsutake mushroom, Tricholoma magnivelare". Biochemical Systematics and Ecology 35 (9): 634–636. doi:10.1016/j.bse.2007.03.001. Bibcode: 2007BioSE..35..634W.
- ↑ Wood, William F.; Archer, Cynthia L.; Largent, David L. (2001). "1-Octen-3-ol, a banana slug antifeedant from mushrooms". Biochemical Systematics and Ecology 29 (5): 531–533. doi:10.1016/s0305-1978(00)00076-4. PMID 11274773. Bibcode: 2001BioSE..29..531W.
- ↑ "Chemical properties of attractants". http://www.nri.org/tsetse/FAQ/chemprop.html.
- ↑ "Grapevine bunch rots: impacts on wine composition, quality, and potential procedures for the removal of wine faults". Journal of Agricultural and Food Chemistry 61 (22): 5189–206. June 2013. doi:10.1021/jf400641r. PMID 23675852.
- ↑ Wnuk, S.; Kinastowski, S.; Kamiński, E. (1983). "Synthesis and analysis of 1-octen-3-ol, the main flavour component of mushrooms". Die Nahrung 27 (5): 479–486. doi:10.1002/food.19830270523. ISSN 0027-769X. PMID 6684212. https://pubmed.ncbi.nlm.nih.gov/6684212/.
- ↑ Matsui, Kenji; Sasahara, Satomi; Akakabe, Yoshihiko; Kajiwara, Tadahiko (2003). "Linoleic acid 10-hydroperoxide as an intermediate during formation of 1-octen-3-ol from linoleic acid in Lentinus decadetes". Bioscience, Biotechnology, and Biochemistry 67 (10): 2280–2282. doi:10.1271/bbb.67.2280. ISSN 0916-8451. PMID 14586122.
- ↑ Min Kuo, Tsung; Gardner, Harold W. (2002). Lipid biotechnology. New York: Marcel Dekker. ISBN 0-585-40371-6. OCLC 48691412. https://www.worldcat.org/oclc/48691412.
- ↑ 14.0 14.1 "Biopesticides Fact Sheet for Octenol". EPA fact sheet. 2007-07-05. https://www3.epa.gov/pesticides/chem_search/reg_actions/registration/fs_G-126_05-Jul-07.pdf.
- ↑ "1-octen-3-ol". http://www.thegoodscentscompany.com/data/rw1024051.html.
- ↑ US FDAs Center for Food Safety and Applied Nutrition. "US FDA/CFSAN – EAFUS List". http://vm.cfsan.fda.gov/~dms/eafus.html.
- ↑ "Fungal-derived semiochemical 1-octen-3-ol disrupts dopamine packaging and causes neurodegeneration". Proceedings of the National Academy of Sciences of the United States of America 110 (48): 19561–6. November 2013. doi:10.1073/pnas.1318830110. PMID 24218591. Bibcode: 2013PNAS..11019561I.
- ↑ "The two odors of iron when touched or pickled: (skin) carbonyl compounds and organophosphines". Angewandte Chemie 45 (42): 7006–9. October 2006. doi:10.1002/anie.200602100. PMID 17009284.
 |


