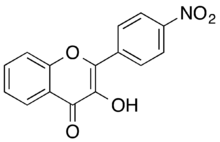
Chemistry:4'-Nitroflavonol
From HandWiki
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
PubChem CID
|
|
| |
| |
| Properties | |
| C15H9NO5 | |
| Molar mass | 283.239 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
4'-Nitroflavonol is a pale yellow solid. This substance belongs to the subclass of flavonols of the class of flavonoids.
Synthesis
4'-Nitroflavonol can be synthesized by the Algar–Flynn–Oyamada reaction of the chalcone formed from 4-nitrobenzaldehyde and 2-hydroxyacetophenone.[1]
Properties
Fluorescence is reported[1] despite the presence of a nitro group at the 4' position of the molecule, which[clarification needed] is known as a fluorescence quencher.[2] An excited state intramolecular proton transfer reaction for 4'-nitroflavonol has also been observed.[1]
References
- ↑ 1.0 1.1 1.2 Chumak, Andrii Yu.; Mudrak, Vladyslav O.; Kotlyar, Volodymyr M.; Doroshenko, Andrey O. (2021-02-01). "4'-Nitroflavonol fluorescence: Excited state intramolecular proton transfer reaction from the non-emissive excited state" (in en). Journal of Photochemistry and Photobiology A: Chemistry 406: 112978. doi:10.1016/j.jphotochem.2020.112978. ISSN 1010-6030.
- ↑ Lippert, Ernst; Kelm, Jürgen (1978-01-25). "Spektroskopische Untersuchungen über die Rolle des Käfig-Effektes bei der Prädissoziation aromatischer Nitroverbindungen" (in en). Helvetica Chimica Acta 61 (1): 279–285. doi:10.1002/hlca.19780610125.
 |

