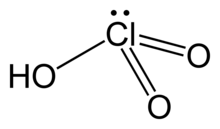
Chemistry:Chloric acid
| |
| |
| Names | |
|---|---|
| Other names
Chloric(V) acid
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| UN number | 2626 |
| |
| |
| Properties | |
| HClO3 | |
| Molar mass | 84.45914 g mol−1 |
| Appearance | colourless solution |
| Density | 1 g/mL, solution (approximate) |
| >40 g/100 ml (20 °C) | |
| Acidity (pKa) | −2.7[1] |
| Conjugate base | Chlorate |
| Structure | |
| pyramidal | |
| Hazards | |
| Main hazards | Oxidant, Corrosive |
| GHS pictograms |  
|
| GHS Signal word | Danger |
| H271, H314 | |
| P210, P220, P221, P260, P264, P280, P283, P301+330+331, P303+361+353, P304+340, P305+351+338, P306+360, P310, P321, P363, P370+378, P371+380+375, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Other anions
|
bromic acid iodic acid |
Other cations
|
ammonium chlorate sodium chlorate potassium chlorate |
Related compounds
|
hydrochloric acid hypochlorous acid chlorous acid perchloric acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Chloric acid, HClO3, is an oxoacid of chlorine, and the formal precursor of chlorate salts. It is a strong acid (pKa ≈ −2.7) and an oxidizing agent.
Properties
Chloric acid is thermodynamically unstable with respect to disproportionation.
Chloric acid is stable in cold aqueous solution up to a concentration of approximately 30%, and solution of up to 40% can be prepared by careful evaporation under reduced pressure.[2] Above these concentrations, chloric acid solutions decompose to give a variety of products, for example:[2]
- 8 HClO
3 → 4 HClO
4 + 2 H
2O + 2 Cl
2 + 3 O
2 - 3 HClO
3 → HClO
4 + H
2O + 2 ClO
2
Hazards
Chloric acid is a powerful oxidizing agent which ignites most organic materials on contact. It is also corrosive.[3]
Production
Chloric acid may be produced from barium chlorate through its reaction with sulfuric acid, resulting in a solution of chloric acid and insoluble barium sulfate precipitate:[4]
- Ba(ClO
3)
2 + H
2SO
4 → 2 HClO
3 + BaSO
4
The chlorate must be dissolved in boiling water and the acid should be somewhat diluted in water and heated before mixing.
Another method which can be used to produce solutions up to 10% concentration is by the use of cation exchange resins and a soluble salt such as NaClO
3, where the Na+ cation will exchange with H+.[4]
Another method is the heating of hypochlorous acid, producing chloric acid and hydrogen chloride:[citation needed]
- 3 HClO → HClO
3 + 2 HCl
Any way it is produced, the acid may be concentrated up to 40% in a vacuum dessicator over H
2SO
4.[4]
See also
References
- ↑ Holleman, Arnold F.; Wiberg, Egon; Wiberg, Nils (2007) (in de). Lehrbuch der anorganischen Chemie. Berlin. ISBN 978-3-11-017770-1. OCLC 180963521.
- ↑ 2.0 2.1 Kirk-Othmer, ed (2001-01-26) (in en). Kirk-Othmer Encyclopedia of Chemical Technology (1 ed.). Wiley. doi:10.1002/0471238961.0308121513051404.a01.pub2. ISBN 978-0-471-48494-3. https://onlinelibrary.wiley.com/doi/book/10.1002/0471238961.
- ↑ "Chloric acid". National Institute of Health. https://pubchem.ncbi.nlm.nih.gov/compound/19654.
- ↑ 4.0 4.1 4.2 Brauer, Georg; Schmeisser, M. (1963). "5. Chlorine, Bromine, Iodine". in Riley, Reed F.. Handbook of Preparative Inorganic Chemistry (2nd ed.). New York, London: Academic Press. pp. 312-313. ISBN 9780121266011. https://archive.org/details/Handbook_of_Preparative_Inorganic_Chemistry_1_2_Brauer/page/n335/mode/2up. Retrieved 6 December 2024.
Additional Information
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- R. Bruce King, ed (1994). "Chloric acid". Encyclopedia of Inorganic Chemistry. 2. Chichester: Wiley. p. 658. ISBN 0-471-93620-0.
 |

