Chemistry:Danoprevir
 | |
| Clinical data | |
|---|---|
| Other names | ITMN-191, RG-7227 |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| NIAID ChemDB | |
| PDB ligand | |
| Chemical and physical data | |
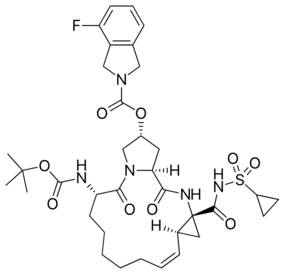
| Formula | C35H46FN5O9S |
| Molar mass | 731.84 g·mol−1 |
| |
| |
Danoprevir (INN)[1] is an orally available[2] 15-membered macrocyclic peptidomimetic inhibitor of NS3/4A HCV protease.[3] It contains acylsulfonamide, fluoroisoindole and tert-butyl carbamate moieties. Danoprevir is a clinical candidate based on its favorable potency profile against multiple HCV genotypes 1–6 and key mutants (GT1b, IC50 = 0.2–0.4 nM; replicon GT1b, EC50 = 1.6 nM).[4] Danoprevir has been evaluated in an open-label, single arm clinical trial in combination with ritonavir for treating COVID-19[5] and favourably compared to lopinavir/ritonavir in a second trial.[6]
History
Danaoprevir was initially developed by Array BioPharma then licensed to Roche for further development and commercialization. In 2013, Danoprevir was licensed to Ascletis by Roche for development and production in China under the tradename Ganovo.[7][8]
References
- ↑ "International Nonproprietary Names for Pharmaceutical Substances (INN). Recommended International Nonproprietary Names: List 64". World Health Organization. p. 265. https://www.who.int/medicines/publications/druginformation/innlists/RL64.pdf.
- ↑ "Danoprevir, a small-molecule NS3/4A protease inhibitor for the potential oral treatment of HCV infection". Current Opinion in Investigational Drugs 11 (8): 951–63. August 2010. PMID 20721837.
- ↑ "Discovery of danoprevir (ITMN-191/R7227), a highly selective and potent inhibitor of hepatitis C virus (HCV) NS3/4A protease". Journal of Medicinal Chemistry 57 (5): 1753–69. March 2014. doi:10.1021/jm400164c. PMID 23672640.
- ↑ Levin, Jeremy, ed (2015). "7.4.1 Danoprevir (ITMN-191)". Macrocycles in Drug Discovery. The Royal Society of Chemistry. doi:10.1039/9781782623113. ISBN 978-1-84973-701-2.
- ↑ Chen H, Zhang Z, Wang L, Huang Z, Gong F, Li X (2020). "First clinical study using HCV protease inhibitor danoprevir to treat COVID-19 patients.". Medicine (Baltimore) 99 (48): e23357. doi:10.1097/MD.0000000000023357. PMID 33235105.
- ↑ Zhang Z, Wang S, Tu X, Peng X, Huang Y, Wang L (2020). "A comparative study on the time to achieve negative nucleic acid testing and hospital stays between danoprevir and lopinavir/ritonavir in the treatment of patients with COVID-19.". J Med Virol 92 (11): 2631–2636. doi:10.1002/jmv.26141. PMID 32501538.
- ↑ Ng, Eric (17 July 2018). "Singapore sovereign wealth fund GIC to invest US$75 million in Chinese drug maker Ascletis' Hong Kong IPO". South China Morning Post. https://www.scmp.com/business/investor-relations/ipo-quote-profile/article/2155708/singapore-sovereign-wealth-fund-invest.
- ↑ Speights, Keith (21 February 2017). "How High Can Array BioPharma Inc. Stock Go?". The Motley Fool. https://www.fool.com/investing/2017/02/21/how-high-can-array-biopharma-stock-go.aspx.
Further reading
"Preclinical characteristics of the hepatitis C virus NS3/4A protease inhibitor ITMN-191 (R7227)". Antimicrobial Agents and Chemotherapy 52 (12): 4432–41. December 2008. doi:10.1128/AAC.00699-08. PMID 18824605.
 |
