Chemistry:Pseudohalogen
Pseudohalogens are polyatomic analogues of halogens, whose chemistry, resembling that of the true halogens, allows them to substitute for halogens in several classes of chemical compounds.[1] Pseudohalogens occur in pseudohalogen molecules, inorganic molecules of the general forms Ps–Ps or Ps–X (where Ps is a pseudohalogen group), such as cyanogen; pseudohalide anions, such as cyanide ion; inorganic acids, such as hydrogen cyanide; as ligands in coordination complexes, such as ferricyanide; and as functional groups in organic molecules, such as the nitrile group. Well-known pseudohalogen functional groups include cyanide, cyanate, thiocyanate, and azide.
Common pseudohalogens and their nomenclature
Many pseudohalogens are known by specialized common names according to where they occur in a compound. Well-known ones include (the true halogen chlorine is listed for comparison):
| Group | Dimer | Hydrogen compound | Pseudohalide | Ligand name | In organic compounds | Formula | Structural formula |
|---|---|---|---|---|---|---|---|
| True halogens | |||||||
| chloro | chlorine | hydrochloride | chloride | chlorido- chloro- |
-yl chloride | ~ Cl | −Cl |
| Pseudohalogens | |||||||
| cyano | cyanogen | hydrogen cyanide, prussic acid, formonitrile |
cyanide | cyanido- cyano- |
-nitrile -yl cyanide |
~ CN | −C≡N |
| cyapho | cyaphogen | phosphaethyne | cyaphide | cyaphido- cyapho- |
-yl cyaphide | ~ CP | −C≡P |
| isocyano | diisocyanogen | hydrogen isocyanide, isohydrocyanic acid |
isocyanide | isocyanido- isocyano- |
-isonitrile -yl isocyanide |
~ NC | −N+≡C− |
| hydroxyl | hydrogen peroxide | water | hydroxide | hydroxido- hydroxy- |
-ol | ~ OH | −O−H |
| sulfanyl | hydrogen disulfide | hydrogen sulfide | hydrosulfide | sulfanido- thiolato- |
-thiol -yl mercaptane |
~ SH | −S−H |
| cyanate | cyanic acid | cyanate | cyanato- | -yl cyanate | ~ OCN | −O−C≡N | |
| isocyanate | isocyanic acid | isocyanate | isocyanato- | -yl isocyanate | ~ NCO | −N=C=O | |
| fulminate | fulminic acid | fulminate | fulminato- | -nitrile oxide -yl fulminate |
~ CNO | −C≡N+–O− | |
| thiocyanate, rhodanide |
thiocyanogen | thiocyanic acid | thiocyanate | thiocyanato- | -yl thiocyanate | ~ SCN | −S−C≡N |
| isothiocyanate | isothiocyanic acid | isothiocyanate | isothiocyanato- | -yl isothiocyanate | ~ NCS | −N=C=S | |
| selenocyanate, selenorhodanide |
selenocyanogen | selenocyanic acid | selenocyanate | ~ SeCN | −Se−C≡N | ||
| tellurocyanate,[2] tellurorhodanide |
tellurocyanogen | tellurocyanic acid | tellurocyanate | ~ TeCN | −Te−C≡N | ||
| azide | hexazine | hydrazoic acid | azide | azido- | -yl azide | ~ N3 | −N−−N+≡N ↕ −N=N+=N− |
| nitric oxide | dinitrogen dioxide | nitroxyl | nitrosyl | nitroso- | ~ NO | •N=O | |
| nitrogen dioxide | dinitrogen tetroxide | nitryl | nitro- | ~ NO2 | −NO2 | ||
| cobalt carbonyl | dicobalt octacarbonyl | cobalt tetracarbonyl hydride | tetracarbonylcobaltate | ~ Co(CO)4 | −Co(C≡O)4 | ||
| trinitromethanide | hexanitroethane | nitroform, trinitromethane |
trinitromethanide | trinitromethanido- | -yl trinitromethanide | ~ C(NO2)3 | −C(NO2)3 |
| tricyanomethanide | hexacyanoethane | cyanoform, tricyanomethane |
tricyanomethanide | tricyanomethanido- | -yl tricyanomethanide | ~ C(CN)3 | −C(CN)3 |
| ~ CS2N3[3] | 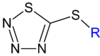
| ||||||
Au− is considered to be a pseudohalogen ion due to its disproportionation reaction with alkali and the ability to form covalent bonds with hydrogen.[4]
Examples of pseudohalogen molecules
Examples of symmetrical pseudohalogens (Ps–Ps) include cyanogen (CN)2, thiocyanogen (SCN)2, hydrogen peroxide H2O2. Another complex symmetrical pseudohalogen is dicobalt octacarbonyl, Co2(CO)8. This substance can be considered as a dimer of the hypothetical cobalt tetracarbonyl, Co(CO)4.
Examples of non-symmetrical pseudohalogens (Ps–X), analogous to the binary interhalogen compounds, are cyanogen halides like ClCN or BrCN; nitryl fluoride, and other compounds. Sometimes nitrosyl chloride NOCl also is considered as pseudohalogen.
Not all combinations are known to be stable.
Pseudohalides
Pseudohalides are the univalent anions (or functional groups) which form hydracids with hydrogen and form insoluble salts with silver such as cyanide, cyanates, fulminate, thiocyanate and azide.
A common complex pseudohalide is a tetracarbonylcobaltate [Co(CO)4]−. The acid HCo(CO)4 is in fact quite a strong acid, though its low solubility renders it not as strong as the true hydrohalic acids.
The behavior and chemical properties of the above pseudohalides are identical to that of the true halide ions. The presence of the internal multiple bonds does not appear to affect their chemical behavior. For example, they can form strong acids of the type HX (compare HCl to HCo(CO)4), and they can react with metals to form compounds like MX (compare NaCl to NaN3).
Nanoclusters of aluminium (often referred to as superatoms) are sometimes considered to be pseudohalides since they, too, behave chemically as halide ions, forming Al13I2− (analogous to I3−) and similar compounds. This is due to the effects of metallic bonding on small scales.
References
- ↑ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "pseudohalogens". doi:10.1351/goldbook.P04930
- ↑ "Tellurocyanate | CNTe | ChemSpider". http://www.chemspider.com/Chemical-Structure.21865273.html.
- ↑ Margaret-Jane Crawford, et al. CS2N3, A Novel Pseudohalogen. J. Am. Chem. Soc. 2000, 122, 9052-9053
- ↑ Mudring, Anjy-Verena; Jansen, Martin (2000-11-28). "ChemInform Abstract: Base-Induced Disproportionation of Elemental Gold.". ChemInform 31 (48): no. doi:10.1002/chin.200048020.
