Chemistry:Dinitrogen dioxide
From HandWiki
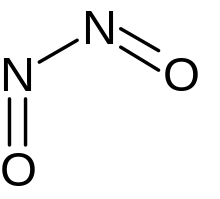
| |
| Identifiers | |
|---|---|
| |
3D model (JSmol)
|
|
| ChEBI |
|
| ChemSpider | |
| 1035 | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| N2O2 | |
| Molar mass | 60.012 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Dinitrogen dioxide is an inorganic compound having molecular formula N2O2. Many structural isomers are possible. The covalent bonding pattern O=N–N=O (a non-cyclic dimer of nitric oxide (NO)) is predicted to be the most stable isomer based on ab initio calculations and is the only one that has been experimentally produced.[1] In the solid form, the molecules have C2v symmetry: the entire structure is planar, with the two oxygen atoms cis across the N–N bond. The O–N distance is 1.15 Å, the N–N distance is 2.33 Å, and the O=N–N angle is 95°.[2]
It is an intermediate in the high-pressure disproportionation of nitric oxide:[3][4]
- 2NO ⇌ N
2O
2 - N
2O
2 + NO → N
2O + NO
2
References
- ↑ Nguyen, Kiet A.; Gordon, Mark S.; Montgomery, John A. Jr.; Michels, H. Harvey (October 1994). "Structures, Bonding, and Energetics of N2O2 Isomers". The Journal of Physical Chemistry 98 (40): 10072–10078. doi:10.1021/j100091a021. https://dr.lib.iastate.edu/bitstreams/be95a54e-5a0e-4578-9fb8-8853ba9bf311/download.
- ↑ Park, Jong Keun; Sun, Hosung (1999). "Theoretical Determination of Geometrical Structures of the Nitric Oxide Dimer, (NO)2" (in ko). Bulletin of the Korean Chemical Society 20 (12): 1399–1408. ISSN 0253-2964. http://www.koreascience.or.kr/article/JAKO199913464470127.page.
- ↑ Agnew, Stephan F.; Swanson, B. I.; Jones, L. H.; Mills, R. L. (April 1985). "Disproportionation of Nitric Oxide at High Pressure". The Journal of Physical Chemistry 89 (9): 1678–1682. doi:10.1021/j100255a026.
- ↑ Tsukahara, Hirokazu; Ishida, Takanobu; Todoroki, Yukiko; Hiraoka, Masahiro; Mayumi, Mitsufumi (January 2003). "Gas-phase Disproportionation of Nitric Oxide at Elevated Pressures". Free Radical Research 37 (2): 171–177. doi:10.1080/1071576021000036489.
- East, Allan L. L. (August 8, 1998). "The 16 valence electronic states of nitric oxide dimer (NO)2". Journal of Chemical Physics 109 (6): 2185–2193. doi:10.1063/1.476786. Bibcode: 1998JChPh.109.2185E.
- Harcourt, Richard D. (April 1990). "The origin of the long N–N bond in N2O2: an ab initio valence bond study". Journal of Molecular Structure: THEOCHEM 206 (3–4): 253–264. doi:10.1016/0166-1280(90)85140-I.[1]
- Dkhissi, Ahmed; Soulard, Pascale; Perrin, Agnès; Lacome, Nelly (May 1997). "The NO Dimer". Journal of Molecular Spectroscopy 183 (1): 12–17. doi:10.1006/jmsp.1996.7249. Bibcode: 1997JMoSp.183...12D.
 |
