Chemistry:Cyanogen
Cyanogen is the chemical compound with the formula (CN)
2. Its structure is N≡C–C≡N. The simplest stable carbon nitride, it is a colorless and highly toxic gas with a pungent odor. The molecule is a pseudohalogen. Cyanogen molecules are linear, and consist of two CN groups ‒ analogous to diatomic halogen molecules, such as Cl2, but far less oxidizing. The two cyano groups are bonded together at their carbon atoms, though other isomers have been detected.[1] The name is also used for the CN radical,[2] and hence is used for compounds such as cyanogen bromide (Br–C≡N)[3] (but see also Cyano radical). When burned at increased pressure with oxygen, it is possible to get a blue tinted flame, the temperature of which is about 4,800 °C (8,670 °F) (a higher temperature is possible with ozone). It is as such regarded as the gas with the second highest temperature of burning (after dicyanoacetylene).
Cyanogen is the anhydride of oxamide:
- H
2N–C(=O)–C(=O)–NH
2 → N≡C–C≡N + 2 H
2O
Oxamide is manufactured from cyanogen by hydration:[4]
- N≡C–C≡N + 2 H
2O → H
2N–C(=O)–C(=O)–NH
2
Preparation
Cyanogen is typically generated from cyanide compounds. One laboratory method entails thermal decomposition of mercuric cyanide:
- 2 Hg(CN)
2 → (CN)
2 + Hg
2(CN)
2
Or, one can combine solutions of copper(II) salts (such as copper(II) sulfate) with cyanides; an unstable copper(II) cyanide is formed which rapidly decomposes into copper(I) cyanide and cyanogen.[5]
- 2 CuSO
4 + 4 KCN → (CN)
2 + 2 CuCN + 2 K
2SO
4
Industrially, it is created by the oxidation of hydrogen cyanide, usually using chlorine over an activated silicon dioxide catalyst or nitrogen dioxide over a copper salt. It is also formed when nitrogen and acetylene are reacted by an electrical spark or discharge.[6]
Reactions
For the two less stable isomers of cyanogen, the order of the atoms differs. Isocyanogen (or cyanogen cyanide) is −
C≡N+
–C≡N.[7] It has been detected in the interstellar medium.[8]
Addition of disulfur dichloride to cyanogen gives 3,4-dichloro-1,2,5-thiadiazole.
Paracyanogen
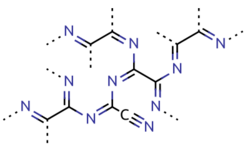
Paracyanogen is a polymer of cyanogen. It can be best prepared by heating mercury(II) cyanide. It can also be prepared by heating silver cyanide, silver cyanate, cyanogen iodide or cyanuric iodide.[9] It can also be prepared by the polymerization of cyanogen at 300 to 500 °C (572 to 932 °F) in the presence of trace impurities. Paracyanogen can also be converted back to cyanogen by heating to 800 °C (1,470 °F).[4] Based on experimental evidence, the structure of this polymeric material is thought to be rather irregular, with most of the carbon atoms being of sp2 type and localized domains of π conjugation.[10]
History
Cyanogen was first synthesized in 1815 by Joseph Louis Gay-Lussac, who determined its empirical formula and named it. Gay-Lussac coined the word "cyanogène" from the Greek words κυανός (kyanos, blue) and γεννάω (gennao, to create), because cyanide was first isolated by Swedish chemist Carl Wilhelm Scheele from the pigment Prussian blue.[11] It attained importance with the growth of the fertilizer industry in the late 19th century and remains an important intermediate in the production of many fertilizers. It is also used as a stabilizer in the production of nitrocellulose.
Cyanogen is commonly found in comets.[12] In 1910 a spectroscopic analysis of Halley's Comet found cyanogen in the comet's tail, which led to public fear that the Earth would be poisoned as it passed through the tail. People in New York wore gas masks, and merchants sold quack "comet pills" claimed to neutralize poisoning.[12] Because of the extremely diffuse nature of the tail, there was no effect when the planet passed through it.[13][14]
Safety
Like other cyanides, cyanogen is very toxic, as it readily undergoes reduction to cyanide, which poisons the cytochrome c oxidase complex, thus interrupting the mitochondrial electron transfer chain. Cyanogen gas is an irritant to the eyes and respiratory system. Inhalation can lead to headache, dizziness, rapid pulse, nausea, vomiting, loss of consciousness, convulsions, and death, depending on exposure.[15] Lethal dose through inhalation typically ranges from 100 to 150 milligrams (1.5 to 2.3 grains).
Cyanogen produces the second-hottest-known natural flame (after dicyanoacetylene aka carbon subnitride) with a temperature of over 4,525 °C (8,177 °F) when it burns in oxygen.[16][17]
See also
References
- ↑ Ringer, A. L.; Sherrill, C. D.; King, R. A.; Crawford, T. D. (2008). "Low-lying singlet excited states of isocyanogen". International Journal of Quantum Chemistry 106 (6): 1137–1140. doi:10.1002/qua.21586. Bibcode: 2008IJQC..108.1137R.
- ↑ Irvine, William M. (2011). "Cyanogen Radical". Encyclopedia of Astrobiology. pp. 402. doi:10.1007/978-3-642-11274-4_1806. ISBN 978-3-642-11271-3.
- ↑ Hartman, W. W.; Dreger, E. E. (1931). "Cyanogen Bromide". Organic Syntheses 11: 30.; Collective Volume, 2, pp. 150
- ↑ 4.0 4.1 Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 320–321. ISBN 978-0-08-037941-8.
- ↑ Brotherton, T. K.; Lynn, J. W. (1959). "The Synthesis And Chemistry Of Cyanogen". Chemical Reviews 59 (5): 841–883. doi:10.1021/cr50029a003.
- ↑ Breneman, A. A. (January 1889). "The Fixation of Atmospheric Nitrogen". Journal of the American Chemical Society 11 (1): 2–27. doi:10.1021/ja02126a001. https://zenodo.org/record/1428969.
- ↑ Bickelhaupt, F. Matthias; Nibbering, Nico M. M.; Van Wezenbeek, Egbert M.; Baerends, Evert Jan (1992). "Central Bond in the Three CN.cntdot.dimers NC-CN, CN-CN and CN-NC: Electron Pair Bonding and Pauli Repulsion Effects". The Journal of Physical Chemistry 96 (12): 4864–4873. doi:10.1021/j100191a027.
- ↑ Agúndez, M.; Marcelino, N.; Cernicharo, J. (2018). "Discovery of Interstellar Isocyanogen (CNCN): Further Evidence that Dicyanopolyynes Are Abundant in Space". The Astrophysical Journal Letters 861 (2): L22. doi:10.3847/2041-8213/aad089. PMID 30186588. Bibcode: 2018ApJ...861L..22A.
- ↑ Bircumshaw, L. L.; F. M. Tayler; D. H. Whiffen (1954). "Paracyanogen: its formation and properties. Part I". J. Chem. Soc.: 931–935. doi:10.1039/JR9540000931.
- ↑ Maya, Leon (1993). "Paracyanogen Reexamined". Journal of Polymer Science Part A 31 (10): 2595–2600. doi:10.1002/pola.1993.080311020. Bibcode: 1993JPoSA..31.2595M. https://zenodo.org/record/1229357.
- ↑ Gay-Lussac, J. L. (1815). "Recherches sur l'acide prussique" (in fr). Annales de Chimie 95: 136–231. https://books.google.com/books?id=m9s3AAAAMAAJ&pg=PA136. Gay-Lussac names cyanogen on p. 163.
- ↑ 12.0 12.1 "Cometary Poison Gas Geyser Heralds Surprises". 2010-11-02. https://science.nasa.gov/science-news/science-at-nasa/2010/02nov_epoxi2/.
- ↑ "Comet's Poisonous Tail". New York Times. 1910-02-08. https://timesmachine.nytimes.com/timesmachine/1910/02/08/104920328.pdf.
- ↑ "Halley's Comet 100 years ago". 2010-05-25. https://www.denverpost.com/2010/05/25/halleys-comet-100-years-ago/.
- ↑ Muir, G. D., ed (1971). Hazards in the Chemical Laboratory. London: The Royal Institute of Chemistry.
- ↑ Thomas, N.; Gaydon, A. G.; Brewer, L. (1952). "Cyanogen Flames and the Dissociation Energy of N2". The Journal of Chemical Physics 20 (3): 369–374. doi:10.1063/1.1700426. Bibcode: 1952JChPh..20..369T.
- ↑ J. B. Conway; R. H. Wilson Jr.; A. V. Grosse (1953). "The Temperature of the Cyanogen-Oxygen Flame". Journal of the American Chemical Society 75 (2): 499. doi:10.1021/ja01098a517.
External links
- National Pollutant Inventory - Cyanide compounds fact sheet
- PhysOrg.com
- CDC - NIOSH Pocket Guide to Chemical Hazards
 |

