Physics:Quantum mechanics/Timeline
Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales. It departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter. In most contexts, the term 'Quantum Mechanics' does not refer to theories that incorporate Einstein's theories of Relativity. [1]

What is Quantum Mechanics and what is Old Quantum Mechanics?
Old quantum theory is a collection of results from the years 1900–1925 which predate modern Quantum mechanics. Although the theory was never complete or self-consistent, it yielded enough successes to establish that the classical Newtonian view of particle motion was insufficient. In 1926 Erwin Schrödinger found a completely quantum mechanical wave-equation, which reproduced all the successes of the old quantum theory without ambiguities and inconsistencies. [2] At approximately the same time a matrix version of the theory was developed, and eventually the two versions merged to become equivalent. This essay focuses on the more accessible wave version.[3]
When is a system in the quantum realm?

The Planck constant (denoted ) is a physical constant that appears throughout the theory of quantum mechanics, but that has no basis in the classical physics of Isaac Newton. The units of Plank's constant can be expressed as either, [energy]·[time], or as [mass]·[velocity]·[length]; (both are called action). A particle is likely to be in the quantum realm if either the mass, speed, or length is small:
- Electrons have less mass than protons or neutrons. For this reason, the electrons in an atom must be modeled by a quantum theory, but we have the option of treating the nucleus as a stationary and classical object at the center.[4]
- Electrons confined to a small space, such as the atom (typically 10-10 meters) will exhibit quantum effects.
- Atoms move very slowly when they are very cold. Quantum effects can occur at very low temperatures.[5]
Timeline
- 1900 - Max Planck vaguely [6] and reluctantly [7] introduces what we now call the photon to accurately model the experimentally observed spectrum of Black body radiation.
- 1905 – Albert Einstein uses Max Planck's hypothesis to explain the photoelectric effect. [8]
- 1909 – Geoffrey Ingram Taylor shows that photons interfere with themselves even if only one photon is near the slits at any given time.[9]
- 1913 – Bohr uses the Rydberg formula to propose the Bohr model of the atom.[10]
- 1923 – Arthur Compton demonstrates particle-like behavior of X-rays.[11]
- 1924 – Louis de Broglie proposes matter waves.[12]
- 1926 – Erwin Schrödinger formulates the Schrödinger equation.
- 1926 – Max Born introduces the probability interpretation of the wavefunction.[13]
- 1927 – Davisson–Germer experiment confirms electron diffraction.[14]
- 1927 – Werner Heisenberg formulates the uncertainty principle.[15]
- 1935 – Schrödinger's cat and the EPR paradox highlight foundational issues.[16]
- 1947 – Lamb shift measured, confirming quantum electrodynamics (QED).[17]
- 1948 – QED developed by Feynman, Schwinger, and Tomonaga.[18]
- 1957 – Many-worlds interpretation proposed by Hugh Everett.[19]
- 1964 – Bell's theorem establishes testable limits on local hidden variables.[20]
- 1981 – Richard Feynman proposes quantum simulation.[21]
- 1984 – BB84 protocol introduces quantum cryptography.[22]
- 1994 – Shor's algorithm shows exponential speedup for factoring.[23]
- 1995 – Quantum error correction developed.[24]
- 1998 – First experimental quantum teleportation.[25]
- 2001 – First experimental demonstration of Shor's algorithm.[26]
- 2012 – Higgs boson discovered at CERN.[27]
- 2015 – Loophole-free Bell test experiments performed.[28]
- 2019 – Google claims quantum supremacy.[29]
- 2022 – Nobel Prize awarded for experimental tests of Bell inequalities.[30]
- 2023 – Advances in fault-tolerant quantum computing.[31]
- 2024 – Continued progress in scalable quantum processors and error correction.[32]
- 2025 – Ongoing development of fault-tolerant quantum computers and quantum networks.[33]
Black-body radiation 1900

Old quantum theory came into existence in 1900 with a calculation by Max Planck that precisely matched the observed patterns of black-body radiation. He used the hypothesis that energy is radiated and absorbed in discrete "quanta" (or "energy elements"). In Planck's theory, the constant emerged as a relation between the energy () and the frequency () associated with the interaction of light with the walls of the back body. This relation is called the Planck relation:
-
()
Planck (cautiously) insisted that this was simply an aspect of the processes of absorption and emission of radiation and had nothing to do with the physical reality of the radiation itself.[34] In fact, he considered his quantum hypothesis a mathematical trick to get the right answer rather than a sizeable discovery.
This equation predicts that photon energy is directly proportional to frequency; if you double f then E doubles. Frequency is inversely proportional to wavelength (since the speed of light is
where λ (lambda) is the length of a wave.
How Planck constructed his model and performed his calculation is beyond the scope of this essay.
Photoelectric Effect 1905

In 1905 Albert Einstein published a paper that explained experimental data from the photoelectric effect that occurs when light strikes a metal surface. The photoelectric effect is the emission of an electron from a substance when light is absorbed. electrons are emitted from solids, liquids or gases when they absorb energy from light. Electrons emitted in this manner may be called photoelectrons. His hypothesis was that the photon's energy frequecy obeyed (i.e. Eq. (1) ). Each photon was assumed to give all of its energy to a single electron, and the energy of this electron could be measured by measuring the voltage required to keep the electron from escaping from the metal.
More links on the Photoelectric effect
- http://phet.colorado.edu/en/simulation/photoelectric (Excellent simulation lab by University of Colorado Boulder's PhET effort.)
- https://en.wikiversity.org/wiki/Photoelectric_Effect_for_Beginners (A discussion similar to this one.}
Taylor makes things spooky with very dim light in 1909



While studying medicine at Göttingen in the 1790s, Thomas Young wrote a thesis on the physical and mathematical properties of sound[35] and in 1799, he presented a paper to the Royal Society where he argued that light was also a wave. His idea was furiously opposed because it contradicted Newton, whose views were considered sacred. Nonetheless, he continued to develop his ideas. In 1801, he presented a famous paper to the Royal Society entitled "On the Theory of Light and Colours" [36] which described various interference phenomena, and in 1803 he performed his famous double-slit experiment (strictly speaking, a double hole experiment).
Diffraction occurs also with water and sound. Diffraction allows one to measure wavelength, and the same value of wavelength occurs for diffraction through single slits, double slits, and N-slits. The results are consistent with diffraction through a circular hole.
This experiment was repeated 100 years later by G. I. Taylor in light so dim that only one photon at a time was likely to be involved with the interference. Amazingly, one photon can interfere with itself. Shown in the figure to the right is a simulated animation of what is observed. Since the photons are passing one at a time, a time gap separates when each particle strikes the screen and creates a black dot. [37] The experiment has been repeated in recent years with electrons, atoms, and even molecules containing over 800 atoms.[38]
Bohr-Rutherford Model 1913
The Bohr model, introduced by Niels Bohr in 1913, depicts the atom as a small, positively charged nucleus surrounded by electrons that travel in circular orbelits around the nucleus—similar in structure to the solar system, but with attraction provided by electrostatic forces rather than gravity. The Bohr model is a primitive model of the hydrogen atom that has been surpassed by the more sophisticated Schrodinger's equation. Nevertheless, Bohr's calculation should be viewed as more than a lucky guess. A guess based on dimensional analysis using a new and controversial fundamental constant is also an educated guess. The Bohr model assumes circular orbits of radius, , and follows Newton's laws of physics, but with the following two embellishments to Newton's theory:
-
()
-
()
and is momentum. Lambda, or , is now known as the De Broglie wavelength (although Bohr did not use the De Broglie wavelength to construct this model.) For an electron of mass , and velocity , .[39]

The meaning of allowed orbits can be discerned from the pair of figures situated to the left. The wavelength must be such that the number that fit into a full circumference (2пr) must equal one, or two, or 3, or 4 (and so forth). In one case, the wave fits perfectly in the circles (with exactly four wavelengths). This corresponds to an electron in the third excited state, or n =4. The other wave does not fit into its allowed radius because it doubles over itself. This energy level does not exist for this atom.
Planck's relation (i.e. Eq.(1)) holds for the photons that are emitted from the hydrogen atom, with the photon energy, , being equal to the difference in energy between the two electron orbitals. This can be expressed as an energy conservation law:
- ,
where is the higher energy and is the lower energy of the atom. These energies were calculated using Newton's laws of physics, just as one would calculate the energy of a satellite orbiting the Earth. The frequency of the light, , could be measured by passing light the very hot gas (glowing) through a prism and measuring the wavelength.
While equations(1), (2), and (3) seem to embody all the calculations of Old Quantum Mechanics, they oversimplify the lines of reasoning actually used. Bohr, for example, did not use equation (3). And he wrote (2) not as a statement about "allowed" wavelengths, but about the quantization of angular momentum (L = mvr= nh/(2π)). In fact, the wave behavior of matter particles such as the electron (i.e., matter waves) was not suspected in 1913, and at that time Bohr did not believe in the existence of photons. [40]
Compton Effect 1923


More evidence that Eq.(3) also holds for the momentum of an individual photon was provided by Arthur Holly Compton in 1923. Since the photon is generally understood to be massless, the momentum, , of the photon is not equal to , but equal to , where is the speed of light. The calculation of Compton scattering is a bit more difficult than those collisions usually studied in an undergraduate physics course because it is necessary to perform a relativistic calculation. Nevertheless, the calculation can be performed and predictions were confirmed by experimental observation.
de Broglie Waves 1924
| “ | When I conceived the first basic ideas of wave mechanics in 1923–24, I was guided by the aim to perform a real physical synthesis, valid for all particles, of the coexistence of the wave and of the corpuscular aspects that Einstein had introduced for photons in his theory of light quanta in 1905. | ” |
| — De Broglie[41] | ||

Today, the de Broglie relations[42] are more likely to be expressed using different variables. Instead of , it is more common to use (pronounced "h-bar"). Also, is defined as the wavenumber, is the angular frequency. In these variables, we have , and .
Although we are using the de Broglie relations within the context of the non-relativistic quantum theory, much of de Broglie's argument was based on Einstein's theory of special relativity, which describes how length, time, mass and energy are perceived by observers who are moving relative to one another. The figure on the left shows the crests and troughs of a travelling wave (in blue and red). Einstein's special theory of relativity inertial reference frames describes how different observers (moving at different speeds) will perceive length, time, speed, momentum, and energy. Self-consistency is possible only if frequency-wavenumber is proportional to energy-momentum. Mathematically, De Broglie derived only the need for this proportionality, not its value. But it was clear that ħ is that constant.
Schrödinger Wave Equation 1926


Schrödinger put forth a wave equation that gave the correct energy levels of the hydrogen atom in 1926. [43] At last we have an equation from which a fundamental wave theory of quantum mechanics might be constructed.
-
()
Although Heisenberg, Born, and Jordan had already developed a parallel effort using matrices that would prove to be a mathematically equivalent theory[44]), we shall take this as the starting point in our quest for a fully developed theory. While this equation looks intimidating to a novice, it is a standard wave equation that is in many respects simpler than Maxwell's Equations. It is completely deterministic, meaning that it allows one to predict how a wave will evolve (given initial conditions). In other words, like Newton's , equation (4) predicts the future of the wave amplitude (We shall soon discover that predicting future values of ψ is not necessarily the same as predicting the future behavior of the particle.)
To the left are some computer generated solutions Schrödinger's equations for a (very tiny) mass attached to a spring. The symbol, ψ, is spelled psi, but often pronounced "sigh", with a hint of "p" at the beginning. It can also be pronounced "psee". It is called the "wavefunction", and is essentially the "amplitude", analogous to the "height" of the wave. The wavefunctions C through F represent states of known energy not unlike the "allowed" orbits of the Bohr atom. Wavefunctions G and H are more complicated and have no counterpart in Bohr's model of "allowed" states. They are known as "mixed energy" states.
What is the nature of the Wavefunction?
The Schrödinger equation details the behavior of ψ but says nothing of its nature. Schrödinger unsuccessfully tried to interpret it as a charge density. [45] More specifically it seemed plausible to interpret the square of the amplitude, |ψ|2, as a charge density, since the total charge can be shown to remain constant if ψ obeys Schrödinger's equation. Schrödinger always opposed a statistical or probabilistic approach, with its associated discontinuities—much like Einstein, who believed that quantum mechanics was a statistical approximation to an underlying deterministic theory— and never reconciled with the Copenhagen interpretation. In 1926, Max Born successfully interpreted ψ as the probability amplitude, whose absolute square,|ψ|2, is equal to probability density. [46] .[47] Like charge, probability also obeys a conservation law, namely that the sum of all probabilities always adds to 1 (i.e., 100%)
Even though Schrödinger is (correctly) credited with inventing this wave equation, a connection between waves and particle motion was understood as early as 1834 when Hamilton wrote equations that describe the path taken by light in the approximation that the wavelength is very small.
Davisson–Germer's accidental diffraction of electrons 1927

While attempting to clean an oxide film off of nickel surface, Davisson and Germer heated the specimen to high temperature, not knowing that this would create large single crystal big enough to caused diffraction of their electron beam.[48][49]
Heisenberg's uncertainty principle 1927
Have you ever noticed that you can't ask a person what they are thinking without changing what they are thinking about? Here is what one of the founders of Quantum Mechanics had to say:
| “ | If a person tries to observe what he is thinking about at the very moment that he is reflecting on a particular subject, it is generally agreed that he introduces unpredictable and uncontrollable changes in the his thoughts proceed thereafter. Why this happens is not definitely known at the present, but some plausible explanations will be suggested later. | ” |
| — David Bohm[50] | ||
Heisenberg came up with more convincing understanding that learning about something changes it in an unpredictable way. His argument imagined a powerful but hypothetical microscope that could see the electron as it orbited around the nucleus. Combining his knowledge of optics with the Compton Effect, Heisenberg concluded that the light required to see the electron would drastically change its orbit. And if the light were gentle, with low-energy photons, the microscope would yield an image so blurred that it would be impossible to track the electron's path.
Measuring a particle at rest in a dark room
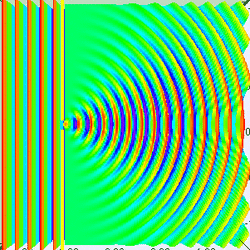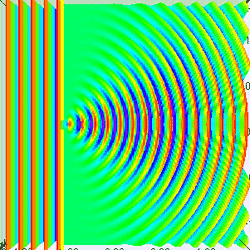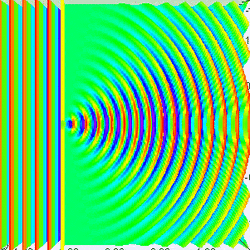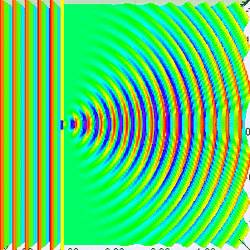
Suppose you are in large dark room with an electron that is known to be at rest. You want to measure its location. The room must be dark because light would interact with and induce motion in the electron. So you put a small hole in a large flat sheet and wave the sheet past where you think the electron is (there is of course no air in the room.) If the electron hits the sheet, your measurement was a failure. But if you repeat the experiment often enough, you will eventually succeed and have knowledge about where the electron is located. Since your sheet never touched the electron, is still at rest, right? Wrong!--No force ever acted on the electron; but nevertheless the electron has now been set in motion! Moreover, you don't know exactly how it is moving.
To understand why, put yourself reference frame of your flat sheet. The sheet is now at rest and an electron is moving towards it. By the de Broglie hypothesis, this electron will have wavelength (given by Eq. (3) to be h/mv). But if the electron acts as a wave, it will undergo single slit diffraction as shown in the figures to the left and right. Notice how decreasing the diameter of the hole increases the motion the spreading of the wave. The more you know about the position, the less you know about the motion, and vice versa. With a bit of basic algebra, the well-known equation for single slit diffraction can be shown to be consistent with the following inequality:
-
()
where , is the uncertainty in position, and is the uncertainty in momentum, . (The symbol is called "sigma"). Equation (5) is known as Heisenberg's uncertainty principle, and renowned theoretical physicists have spent hours attempting to violate it. Under the Copenhagen interpretation of quantum mechanics, it can be proven as a mathematical theorem.
Copenhagen interpretation
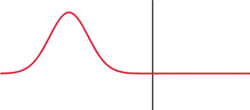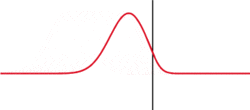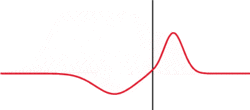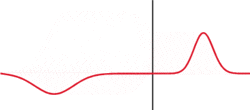
The image to the left illustrates a simple argument for a probabilistic interpretation. Although the wave shown is not a solution to Schrodinger's equation. Wavepackets can become delocalized, hear by impinging on some sort of barrier. What attribute of a particle can be in two places at the same time? Probability! To create this situation in a classroom, tape a wooden block to a stretched snaky spring and strike it.

A detailed set of rules for interpreting ψ that is beyond our scope. But for reference, three fundamental features of these rules are stated:
- |ψ|2ΔV is the probability of the particle being inside a volume ΔV.
- If a wave function is a superposition of two or more states of known energy, the particle has a probability of being in any of those energy states. In particular, if ψ1 and ψ2 are normalized wavefunctions( ʃ|ψ|2dV=1), and if the (complex) amplitudes a1 and a2 are also normalized (i.e., |a1|2+|a2|2=1), then |a1|2 is the probability of having energy E1 and |a2|2 is the probability of having energy E2.
- If a measurement changes our understanding of the nature of any of the particle's attributes (energy, position, velocity, ...), then the wavefunction immediately rearranges itself in a process called the "collapsing of the wavefunction" .
Example 1: Minimum uncertainty within Copenhagen interpretation from repetitive measurement
We consider the perfect measurement when the dense grid of Maxwell's demons is used to detect the presence of the particle at certain coordinate at certain time who can catch the particle at the coordinates when they see it near. Let us consider the free particle with the mass in one spatial dimension in spreading Gaussian wave packet state:
where the spreading : evolves in time
According to the Copenhagen interpretation the position measurement will collapse the wave function to the eigenstate of the position operator (or back prepare it in this state)
To measure the momentum of the particle two consecutive measurements of the position must me done separated in time by to find the velocity first:
While the delta function may be well approximated by a Gaussian with the infinitesimal spread
- (the exact wave function is proportional to it)
the time evolution of the collapsed state between repetitive measurement which will be therefore
Therefore after each measurement the wave functions reconstructs itself by the free evolution of the spreading near-delta function and is only space shifted so the demolition measurements are nonetheless done effectively on the same quantum state. We can reset the center of the coordinate system to 0 after each measurement so if is the probability density distribution of each square of position measurement the average over many measurements is
The corresponding average of the momentum dispersion is immediately
or
Putting the between-measurement time infinitesimal before we get
or the minimum uncertainty.
Example 2: Minimum uncertainty of position and momentum in 3 dimensions
The uncertainty relation in more dimensions is exotic in the sense that coefficients appear at the front of . We want the uncertainty relation for
We will use the general relation for the uncertainty for the operators
We want the uncertainty relation for i.e. for the operators
The first step is to find the auxiliary operators such that this relation can be used directly. We must make therefore the same trick that Dirac made to calculate the square root of the Klein-Gordon operator to get the Dirac equation:
where are matrices from the Dirac equation:
Therefore we have
We can now readily calculate the commutator while using the anty-comutation properties of matrixes and noticing that the symmetric Gaussian state is annihilating in average the terms containing mixed variable like
Calculating 9 commutators (mixed may be zero by Gaussian example) we obtain
Therefore
which is 3 times (space dimension) more then for the one dimension.
Schrodinger's cat 1935

The idea that an object can be in two places at the same time is unsettling, although there seems to be little evidence that this philosophical mystery has interfered with the pursuit of experimental or even theoretical physics. Three ideas have been put forth that have become cultural icons of Quantum Mechanics. They are the EPR Paradox, Schrodingers Cat, and Bell's Theorem (which has been justifiably called “the most profound discovery of science”.[51]) Of these three cultural icons, Schrodinger's cat is easiest to grasp. Schrödinger wrote:[52]
| “ | One can even set up quite ridiculous cases. A cat is penned up in a steel chamber, along with the following device (which must be secured against direct interference by the cat): in a Geiger counter, there is a tiny bit of radioactive substance, so small, that perhaps in the course of the hour one of the atoms decays, but also, with equal probability, perhaps none; if it happens, the counter tube discharges and through a relay releases a hammer that shatters a small flask of hydrocyanic acid. If one has left this entire system to itself for an hour, one would say that the cat still lives if meanwhile no atom has decayed. The psi-function of the entire system would express this by having in it the living and dead cat (pardon the expression) mixed or smeared out in equal parts.. | ” |
| — Erwin Schrödinger[53] | ||
Developments
- 1964 – John Stewart Bell puts forth Bell's theorem
- 1982 - Bell's theorem was experimentally verified by Alain Aspect[54]
- 2010 - Cleland puts an object visible to the naked eye (but smaller than a cat) into a mixed energy state. [55]
Recent Dvelopments
Lamb shift 1947
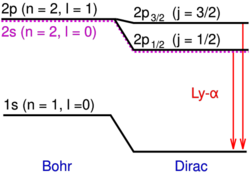
In 1947 Willis Lamb and Robert Retherford measured a tiny shift in the hydrogen spectrum now called the Lamb shift.[56] The result was important because it showed that a simple relativistic wave equation for the electron was not enough. Something else was affecting the atom.
That "something else" was eventually understood to be the interaction of the electron with the quantized electromagnetic field. The Lamb shift therefore became one of the early great successes of modern quantum electrodynamics.[57]
Quantum electrodynamics 1948

By the late 1940s Richard Feynman, Julian Schwinger, and Sin-Itiro Tomonaga had developed the theory now called quantum electrodynamics (QED).[58] QED combines quantum mechanics with the electromagnetic field and describes how light and charged matter interact.
QED was revolutionary because it produced extremely accurate predictions, including corrections associated with the Lamb shift and the magnetic moment of the electron. It also introduced calculational tools that became central to later quantum field theories.
Many-worlds interpretation 1957

In 1957 Hugh Everett III proposed the many-worlds interpretation of quantum mechanics.[59] Rather than assuming that a measurement causes the wavefunction to collapse, Everett suggested that the wavefunction always evolves according to Schrödinger's equation.
In this view, a measurement correlates observer and system, producing effectively separate branches corresponding to different outcomes. Whether this is the best interpretation remains controversial, but Everett's proposal permanently changed the discussion of the foundations of quantum mechanics.
Schrödinger's cat" paradox according to the many-worlds interpretation. In this interpretation, every quantum event is a branch point; the cat is both alive and dead, even after the box is opened, but the "alive" and "dead" cats are in different branches of the multiverse, both of which are equally real, but which do not interact with each other.
Bell's theorem 1964
 In 1964 John Bell derived what is now called Bell's theorem.[60] Bell showed that any theory based on local hidden variables must satisfy certain inequalities. Quantum mechanics predicts situations in which those inequalities are violated.
In 1964 John Bell derived what is now called Bell's theorem.[60] Bell showed that any theory based on local hidden variables must satisfy certain inequalities. Quantum mechanics predicts situations in which those inequalities are violated.
Bell's theorem transformed philosophical debate into experimental science. It showed that the strange correlations predicted by quantum mechanics could be tested in the laboratory, and that "completing" quantum mechanics with local hidden variables would not work.
Quantum simulation 1981

In 1981 Richard Feynman argued that classical computers are poorly suited for simulating quantum systems, and suggested that one should instead build quantum simulators.[61] This idea is one of the conceptual origins of quantum computing.
The basic insight is simple: if nature is quantum mechanical, then a controllable quantum system may efficiently imitate another quantum system. This turned the weirdness of quantum mechanics into a possible computational resource.
Quantum cryptography 1984
In 1984 Charles Bennett and Gilles Brassard proposed the BB84 protocol.[63] It is the first major protocol for quantum cryptography, especially for quantum key distribution.
Its importance lies in the fact that eavesdropping on unknown quantum states generally disturbs them. This makes it possible, in principle, to detect interception of the key. BB84 showed that quantum mechanics is not only a theory of matter and radiation, but also a resource for information security.
Shor's algorithm 1994

In 1994 Peter Shor discovered Shor's algorithm, a quantum algorithm for factoring large integers in polynomial time.[64] This was a turning point in the history of quantum computing.
Its significance is practical as well as theoretical. Many classical cryptographic systems rely on the difficulty of factoring. Shor's work showed that a sufficiently powerful quantum computer would threaten such systems and therefore gave the field a major strategic and scientific push.
Diagram presenting the encryption and the decryption of a document using asymmetric cryptography. Some forms of encryption (including asymmetric cryptography) are at risk of being broken by future quantum computers.
Quantum error correction 1995

One of the apparent obstacles to quantum computing is that quantum states are easily disturbed by noise and decoherence. In 1995, schemes for quantum error correction were developed that showed this obstacle was not fatal.[65]
Quantum error correction works by encoding one logical qubit into a larger entangled state of several physical qubits. Although unknown quantum states cannot be copied, they can still be protected by cleverly distributing information so that certain errors can be detected and corrected.
Quantum teleportation 1998

In 1998 experimental groups demonstrated early forms of quantum teleportation.[66] Quantum teleportation does not move matter itself from place to place. Instead, it transfers the quantum state of one system to another distant system.
Teleportation requires two ingredients: entanglement and an ordinary classical communication channel. It is one of the clearest demonstrations that quantum information behaves differently from classical information.
Comparative prediction of the 13C NMR spectrum of sucrose using various methods. Experimental spectrum is in the middle. Upper spectrum (black) was obtained by empirical routine. Lower spectra (red and green) were obtained by quantum-chemical calculations in PRIRODA and GAUSSIAN respectively. Included information: used theory level/basis set/solvent model, accuracy of prediction (linear correlation factor and root mean square deviation), calculation time on personal computer (blue).
Experimental Shor demonstration 2001
In 2001 a small-scale experimental implementation of Shor's algorithm was reported.[67] The number factored was tiny, but the importance of the experiment was symbolic: it showed that nontrivial quantum algorithms could be realized in the laboratory.
Such demonstrations did not yet threaten real-world cryptography, but they helped establish quantum computing as an experimental discipline rather than merely a theoretical possibility.
Higgs boson 2012

In 2012 experiments at CERN announced the discovery of the Higgs boson.[68] Strictly speaking, this belongs more naturally to high-energy particle physics and quantum field theory than to nonrelativistic quantum mechanics.
Nevertheless, it is historically relevant because it confirmed a central part of the quantum field-theoretic description of fundamental particles. It also illustrates how the quantum viewpoint ultimately extends far beyond atoms and light.
Loophole-free Bell tests 2015
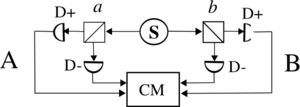
In 2015 several groups reported loophole-free Bell test experiments.[69] Earlier Bell tests strongly supported quantum mechanics, but skeptics could point to experimental "loopholes" such as imperfect detection or possible communication between devices.
The 2015 experiments were designed to close the major loopholes simultaneously. Their results further strengthened the conclusion that nature violates Bell inequalities in the way predicted by quantum mechanics. The source S produces pairs of "photons", sent in opposite directions. Each photon encounters a two-channel polariser whose orientation can be set by the experimenter. Emerging signals from each channel are detected and coincidences counted by the coincidence monitor CM.
Quantum supremacy claim 2019
Demonstrating Quantum Supremacy
In 2019 Google announced that its quantum processor had performed a specific computation faster than a known classical alternative, and described the result as quantum supremacy.[70]
The term itself is controversial, and the practical importance of the particular benchmark was debated. Even so, the announcement marked an important moment in public awareness of quantum computing and intensified work on useful quantum advantage.
Entanglement Nobel Prize 2022

In 2022 the Nobel Prize in Physics was awarded to Alain Aspect, John Clauser, and Anton Zeilinger for experiments involving entangled photons and tests of Bell inequalities.[71]
This award was historically important because it recognized that foundational questions once treated as philosophical had become central experimental physics. Entanglement is now also a practical resource in quantum information science.
Fault-tolerant progress 2023

In 2023 researchers reported further progress in fault-tolerant quantum computing and in experiments showing that logical qubits can be protected better than the underlying physical qubits.[72]
This matters because large-scale quantum computing requires not merely a few good qubits, but a scalable architecture in which errors can be suppressed faster than they accumulate. Quantum error correction therefore remains central to the field.
Scalable processors and error correction 2024
In 2024 work continued on larger and more reliable quantum processors, improved control systems, and better implementations of error-correcting codes.[73] These advances did not yet amount to fully general fault-tolerant quantum computing, but they represented steady progress toward that goal.
By this stage, the field had clearly moved beyond proof-of-principle demonstrations and into engineering questions involving architecture, fabrication, calibration, and error management.
Quantum computing and networking 2025
In 2025 research continued on fault-tolerant quantum computers and on the development of quantum networks, including protocols for distributed entanglement and secure communication.[74]
Whether these efforts soon produce broadly useful machines remains uncertain, but they show that quantum mechanics has evolved from a theory explaining atomic spectra into a framework supporting new technologies for computation, communication, and sensing.
See also
Index
Core theory Foundations Conceptual and interpretations Mathematical structure and systems Atomic and spectroscopy Wavefunctions and modes Quantum dynamics and evolution Measurement and information Quantum information and computing
Applications and extensions Quantum optics and experiments Open quantum systems Quantum field theory Statistical mechanics and kinetic theory Condensed matter and solid-state physics Plasma and fusion physics Timeline Advanced and frontier topics
Quantum Book II
Quantum Book III
Quantum Book IV
Full contents
- Physics:Quantum basics
- Physics:Quantum Postulates
- Physics:Quantum Hilbert space
- Physics:Quantum Observables and operators
- Physics:Quantum mechanics
- Physics:Quantum mechanics measurements
- Physics:Quantum state
- Physics:Quantum system
- Physics:Quantum superposition
- Physics:Quantum probability
- Physics:Quantum Mathematical Foundations of Quantum Theory
- Physics:Quantum Interpretations of quantum mechanics
- Physics:Quantum Wave–particle duality
- Physics:Quantum Complementarity principle
- Physics:Quantum Uncertainty principle
- Physics:Quantum Measurement problem
- Physics:Quantum Bell's theorem
- Physics:Quantum Hidden variable theory
- Physics:Quantum nonlocality
- Physics:Quantum contextuality
- Physics:Quantum Darwinism
- Physics:Quantum A Spooky Action at a Distance
- Physics:Quantum A Walk Through the Universe
- Physics:Quantum The Secret of Cohesion and How Waves Hold Matter Together
- Physics:Quantum measurement problem
- Physics:Quantum Density matrix
- Physics:Quantum Exactly solvable quantum systems
- Physics:Quantum Formulas Collection
- Physics:Quantum A Matter Of Size
- Physics:Quantum Symmetry in quantum mechanics
- Physics:Quantum Angular momentum operator
- Physics:Quantum Runge–Lenz vector
- Physics:Quantum Approximation Methods
- Physics:Quantum Matter Elements and Particles
- Physics:Quantum Dirac equation
- Physics:Quantum Klein–Gordon equation
- Physics:Quantum pendulum
- Physics:Quantum configuration space
- Physics:Quantum Atomic structure and spectroscopy
- Physics:Quantum Hydrogen atom
- Physics:Quantum number
- Physics:Quantum Multi-electron atoms
- Physics:Quantum Fine structure
- Physics:Quantum Hyperfine structure
- Physics:Quantum Isotopic shift
- Physics:Quantum defect
- Physics:Quantum Zeeman effect
- Physics:Quantum Stark effect
- Physics:Quantum Spectral lines and series
- Physics:Quantum Selection rules
- Physics:Quantum Fermi's golden rule
- Physics:Quantum beats
- Physics:Quantum Wavefunction
- Physics:Quantum Superposition principle
- Physics:Quantum Eigenstates and eigenvalues
- Physics:Quantum Boundary conditions and quantization
- Physics:Quantum Standing waves and modes
- Physics:Quantum Normal modes and field quantization
- Physics:Number of independent spatial modes in a spherical volume
- Physics:Quantum Density of states
- Physics:Quantum carpet
- Physics:Quantum Time evolution
- Physics:Quantum Schrödinger equation
- Physics:Quantum Time-dependent Schrödinger equation
- Physics:Quantum Stationary states
- Physics:Quantum Perturbation theory
- Physics:Quantum Time-dependent perturbation theory
- Physics:Quantum Adiabatic theorem
- Physics:Quantum Scattering theory
- Physics:Quantum S-matrix
- Physics:Quantum tunnelling
- Physics:Quantum speed limit
- Physics:Quantum revival
- Physics:Quantum reflection
- Physics:Quantum oscillations
- Physics:Quantum jump
- Physics:Quantum boomerang effect
- Physics:Quantum chaos
- Physics:Quantum Measurement theory
- Physics:Quantum Measurement operators
- Physics:Quantum Projective measurement
- Physics:Quantum POVM
- Physics:Quantum Weak measurement
- Physics:Quantum Measurement collapse
- Physics:Quantum entanglement
- Physics:Quantum Zeno effect
- Physics:Quantum limit
- Physics:Quantum information theory
- Physics:Quantum Qubit
- Physics:Quantum Entanglement
- Physics:Quantum Gates and circuits
- Physics:Quantum Computing Algorithms in the NISQ Era
- Physics:Quantum Noisy Qubits
- Physics:Quantum random access code
- Physics:Quantum pseudo-telepathy
- Physics:Quantum network
- Physics:Quantum money
- Physics:Quantum Nonlinear King plot anomaly in calcium isotope spectroscopy
- Physics:Quantum optics beam splitter experiments
- Physics:Quantum Ultra fast lasers
- Physics:Quantum Experimental quantum physics
- Physics:Quantum optics
- Template:Quantum optics operators
- Physics:Quantum Open systems
- Physics:Quantum Master equation
- Physics:Quantum Lindblad equation
- Physics:Quantum Decoherence
- Physics:Quantum dissipation
- Physics:Quantum Markov semigroup
- Physics:Quantum Markovian dynamics
- Physics:Quantum Non-Markovian dynamics
- Physics:Quantum Trajectories
- Physics:Quantum field theory (QFT) basics
- Physics:Quantum field theory (QFT) core
- Physics:Quantum Fields and Particles
- Physics:Quantum Second quantization
- Physics:Quantum Harmonic Oscillator field modes
- Physics:Quantum Creation and annihilation operators
- Physics:Quantum vacuum fluctuations
- Physics:Quantum Propagators in quantum field theory
- Physics:Quantum Feynman diagrams
- Physics:Quantum Path integral formulation
- Physics:Quantum Renormalization in field theory
- Physics:Quantum Renormalization group
- Physics:Quantum Field Theory Gauge symmetry
- Physics:Quantum Non-Abelian gauge theory
- Physics:Quantum Electrodynamics (QED)
- Physics:Quantum chromodynamics (QCD)
- Physics:Quantum Electroweak theory
- Physics:Quantum Standard Model
- Physics:Quantum triviality
- Physics:Quantum confinement problem
- Physics:Quantum Statistical mechanics
- Physics:Quantum Partition function
- Physics:Quantum Distribution functions
- Physics:Quantum Liouville equation
- Physics:Quantum Kinetic theory
- Physics:Quantum Boltzmann equation
- Physics:Quantum BBGKY hierarchy
- Physics:Quantum Relaxation and thermalization
- Physics:Quantum Thermodynamics
- Physics:Quantum Band structure
- Physics:Quantum Fermi surfaces
- Physics:Quantum Semiconductor physics
- Physics:Quantum Phonons
- Physics:Quantum Electron-phonon interaction
- Physics:Quantum Superconductivity
- Physics:Quantum Topological phases of matter
- Physics:Quantum well
- Physics:Quantum spin liquid
- Physics:Quantum spin Hall effect
- Physics:Quantum phase transition
- Physics:Quantum critical point
- Physics:Quantum dot
- Physics:Quantum Fusion reactions and Lawson criterion
- Physics:Quantum Plasma (fusion context)
- Physics:Quantum Magnetic confinement fusion
- Physics:Quantum Inertial confinement fusion
- Physics:Quantum Plasma instabilities and turbulence
- Physics:Quantum Tokamak core plasma
- Physics:Quantum Tokamak edge physics and recycling asymmetries
- Physics:Quantum Stellarator
- Physics:Quantum mechanics/Timeline
- Physics:Quantum mechanics/Timeline/Pre-quantum era
- Physics:Quantum mechanics/Timeline/Old quantum theory
- Physics:Quantum mechanics/Timeline/Modern quantum mechanics
- Physics:Quantum mechanics/Timeline/Quantum field theory era
- Physics:Quantum mechanics/Timeline/Quantum information era
- Physics:Quantum mechanics/Timeline/Quantum technology era
- Physics:Quantum mechanics/Timeline/Quiz
- Physics:Quantum topology
- Physics:Quantum battery
- Physics:Quantum Supersymmetry
- Physics:Quantum Black hole thermodynamics
- Physics:Quantum Holographic principle
- Physics:Quantum gravity
- Physics:Quantum De Sitter invariant special relativity
- Physics:Quantum Doubly special relativity
- Physics:Quantum arithmetic geometry
- Physics:Quantum unsolved problems
- Physics:Quantum Yang-Mills mass gap
- Physics:Quantum gravity problem
- Physics:Quantum black hole information paradox
- Physics:Quantum dark matter problem
- Physics:Quantum neutrino mass problem
- Physics:Quantum matter-antimatter asymmetry problem
Foundations

Conceptual and interpretations

Mathematical structure and systems

Atomic and spectroscopy

Wavefunctions and modes

Quantum dynamics and evolution

Measurement and information

Quantum information and computing

Quantum optics and experiments

Open quantum systems

Quantum field theory

Statistical mechanics and kinetic theory

Condensed matter and solid-state physics

Plasma and fusion physics

Timeline

Advanced and frontier topics

See the subpage for a quiz on this topic.
References and Endnotes
- Most of material was lifted out of Wikipedia
- ↑ https://en.wikipedia.org/wiki/Quantum_mechanics
- ↑ https://en.wikipedia.org/wiki/Old_quantum_theory
- ↑ https://en.wikipedia.org/w/index.php?title=Matrix_mechanics&oldid=577613976
- ↑ This analysis assumes that the speed is much less than that of light (i.e. Wikipedia:nonrelativistic).
- ↑ There is a fourth condition for quantum mechanics to occur: when elementary particles collide at very high energy, they can interact with each in such a way as to create new particles. This is the field of High Energy Physics (or particle physics), and will not be part of our discussion, except in one minor way. In the Compton effect, the x-ray was "high energy" (in those days) and acts as a classical particle (like a billiard ball) that rips apart the atom.
- ↑ Planck thought (perhaps correctly?) that it is not light but the way atoms behaved that was quantized.
- ↑ https://en.wikipedia.org/wiki/History_of_quantum_mechanics
- ↑ This and Planck's work begin to establish wave-particle duality for electromagnetic radiation.
- ↑ Taylor, G. I. (1909).
- ↑ https://en.wikipedia.org/wiki/Bohr_model
- ↑ https://en.wikipedia.org/wiki/Compton_scattering
- ↑ https://en.wikipedia.org/wiki/Matter_wave
- ↑ https://en.wikipedia.org/wiki/Born_rule
- ↑ https://en.wikipedia.org/wiki/Davisson%E2%80%93Germer_experiment
- ↑ https://en.wikipedia.org/wiki/Uncertainty_principle
- ↑ https://en.wikipedia.org/wiki/EPR_paradox
- ↑ https://en.wikipedia.org/wiki/Lamb_shift
- ↑ https://en.wikipedia.org/wiki/Quantum_electrodynamics
- ↑ https://en.wikipedia.org/wiki/Many-worlds_interpretation
- ↑ https://en.wikipedia.org/wiki/Bell%27s_theorem
- ↑ https://en.wikipedia.org/wiki/Quantum_computing
- ↑ https://en.wikipedia.org/wiki/BB84
- ↑ https://en.wikipedia.org/wiki/Shor%27s_algorithm
- ↑ https://en.wikipedia.org/wiki/Quantum_error_correction
- ↑ https://en.wikipedia.org/wiki/Quantum_teleportation
- ↑ https://en.wikipedia.org/wiki/Shor%27s_algorithm
- ↑ https://en.wikipedia.org/wiki/Higgs_boson
- ↑ https://en.wikipedia.org/wiki/Bell_test_experiments
- ↑ https://en.wikipedia.org/wiki/Quantum_supremacy
- ↑ https://www.nobelprize.org/prizes/physics/2022/summary/
- ↑ https://en.wikipedia.org/wiki/Quantum_error_correction
- ↑ https://en.wikipedia.org/wiki/Quantum_computing
- ↑ https://en.wikipedia.org/wiki/Quantum_technology
- ↑ Kuhn, T. S. (1978). Black-body theory and the quantum discontinuity 1894-1912. Oxford: Clarendon Press. ISBN 0195023838.
- ↑ Mason, P. (1981). The Light Fantastic. Penguin Books. ISBN 978-0-14-006129-1.
- ↑ Young, T. (1802). "The Bakerian Lecture: On the Theory of Light and Colours". Philosophical Transactions of the Royal Society of London 92: 12–48.
- ↑ Taylor, Geoffrey Ingram. "Interference fringes with feeble light." Proceedings of the Cambridge Philosophical Society. Vol. 15. No. 1. 1909.
- ↑ https://en.wikipedia.org/w/index.php?title=Double-slit_experiment&oldid=618587827
- ↑ Incidentally, this equation also holds for photons, where is the photon's momentum.
- ↑ https://en.wikipedia.org/wiki/Bohr_model
- ↑ Louis de Broglie "The Reinterpretation of Wave Mechanics" Foundations of Physics, Vol. 1 No. 1 (1970)
- ↑ L. de Broglie, Recherches sur la théorie des quanta (Researches on the quantum theory), Thesis (Paris), 1924; L. de Broglie, Ann. Phys. (Paris) 3, 22 (1925).
- ↑ https://en.wikipedia.org/wiki/Schrodinger
- ↑ https://en.wikipedia.org/wiki/Quantum_mechanics
- ↑ Moore, W.J. (1992). Schrödinger: Life and Thought. Cambridge University Press. p. 219. ISBN 0-521-43767-9.
- ↑ Moore, W.J. (1992). Schrödinger: Life and Thought. Cambridge University Press. p. 220. ISBN 0-521-43767-9.
- ↑ It is clear that even in his last year of life, as shown in a letter to Max Born, that Schrödinger never accepted the Copenhagen interpretation (cf. p. 220). Moore, W.J. (1992). Schrödinger: Life and Thought. Cambridge University Press. p. 479. ISBN 0-521-43767-9.
- ↑ Hugh D. Young, Roger A. Freedman: University Physics, Ed. 11. Pearson Education, Addison Wesley, San Francisco 2004, 0-321-20469-7, S. 1493-1494.
- ↑ https://en.wikipedia.org/w/index.php?title=Davisson%E2%80%93Germer_experiment&oldid=580087519
- ↑ Bohm, David. Quantum Theory, Dourier Dover Publications, 1951]
- ↑ Henry P. Stapp, "Bell's Theorem and World Process", Nuovo Cimento, Vol. 29B, No. 2, p. 270 (1975). (Quote on p. 271)
- ↑ Schroedinger: "The Present Situation in Quantum Mechanics." 5. Are the Variables Really Blurred?
- ↑ Die gegenwärtige Situation in der Quantenmechanik (The present situation in quantum mechanics), Naturwissenschaften
(translated by John D. Trimmer in Proceedings of the American Philosophical Society)] - ↑ in what might be the most philosophically profound experiment ever performed
- ↑ http://www.nature.com/news/2010/100317/full/news.2010.130.html Andrew Cleland at the University of California, Santa Barbara, and his team cooled a tiny metal paddle until it reached its quantum mechanical 'ground state' — the lowest-energy state permitted by quantum mechanics. They then used the weird rules of quantum mechanics to simultaneously set the paddle moving while leaving it standing still.
- ↑ https://en.wikipedia.org/wiki/Lamb_shift
- ↑ https://en.wikipedia.org/wiki/Quantum_electrodynamics
- ↑ https://en.wikipedia.org/wiki/Quantum_electrodynamics
- ↑ https://en.wikipedia.org/wiki/Many-worlds_interpretation
- ↑ https://en.wikipedia.org/wiki/Bell%27s_theorem
- ↑ https://en.wikipedia.org/wiki/Quantum_computing
- ↑ Migdał, Piotr; Jankiewicz, Klementyna; Grabarz, Paweł; Decaroli, Chiara; Cochin, Philippe (2022). "Visualizing quantum mechanics in an interactive simulation - Virtual Lab by Quantum Flytrap". Optical Engineering 61 (8). doi:10.1117/1.OE.61.8.081808.
- ↑ https://en.wikipedia.org/wiki/BB84
- ↑ https://en.wikipedia.org/wiki/Shor%27s_algorithm
- ↑ https://en.wikipedia.org/wiki/Quantum_error_correction
- ↑ https://en.wikipedia.org/wiki/Quantum_teleportation
- ↑ https://en.wikipedia.org/wiki/Shor%27s_algorithm
- ↑ https://en.wikipedia.org/wiki/Higgs_boson
- ↑ https://en.wikipedia.org/wiki/Bell_test_experiments
- ↑ https://en.wikipedia.org/wiki/Quantum_supremacy
- ↑ https://www.nobelprize.org/prizes/physics/2022/summary/
- ↑ https://en.wikipedia.org/wiki/Quantum_error_correction
- ↑ https://en.wikipedia.org/wiki/Quantum_computing
- ↑ https://en.wikipedia.org/wiki/Quantum_technology

![An interactive simulation of an optical implementation of the BB84 quantum key distribution protocol in the Virtual Lab by Quantum Flytrap,[62] available online. In this optical setup, bits are encoded using orthogonal polarization states of photons. Alice and Bob select their measurement bases by rotating the polarization by 0 or 45 degrees using Faraday rotators. Single-photon detectors measure the output after the photons pass through a polarizing beam splitter, which separates the polarizations.](/wiki/images/e/e6/BB84_QKD_protocol_-_Virtual_Lab_by_Quantum_Flytrap.png)