Chemistry:Deoxyuridine monophosphate
| |
| |
| Names | |
|---|---|
| IUPAC name
2′-Deoxyuridylic acid
| |
| Systematic IUPAC name
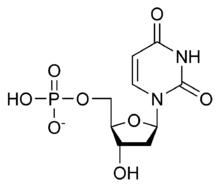
[(2R,3S,5R)-5-(2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-3-hydroxyoxolan-2-yl]methyl dihydrogen phosphate | |
| Other names
dUMP
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| MeSH | uridine-4'-monophosphate |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C9H13N2O8P | |
| Molar mass | 308.182 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Deoxyuridine monophosphate (dUMP), also known as deoxyuridylic acid or deoxyuridylate in its conjugate acid and conjugate base forms, respectively, is a deoxynucleotide.
It is an intermediate in the metabolism of deoxyribonucleotides.
Biosynthesis
Deoxyuridine monophosphate (dUMP) is the deoxygenated form of uridine monophosphate (UMP), and is the precursor to deoxythymidine monophosphate (dTMP), a component of DNA nucleotide biosynthesis.[1] By replacing the hydroxyl group at the 2' carbon of ribose with a hydrogen, UMP becomes deoxygenated to dUMP.
The synthesis of deoxyuridine monophosphate (dUMP) is a multi-step process that begins with uridine monophosphate (UMP), the product of pyrimidine biosynthesis.[2] The enzyme nucleoside monophosphate kinase converts UMP and ATP to uridine diphosphate (UDP) and ADP.
In the presence of excess ATP, the enzyme ribonucleotide reductase initiates a chain reaction with UDP, which catalyzes the formation of deoxyuridine diphosphate (dUDP), which is then converted to deoxyuridine triphosphate (dUTP), then deoxyuridine monophosphate (dUMP) via the addition or removal of phosphate groups.[3]
Interactive pathway map
See also
Notes
- ↑ Berg, J. M.; Tymoczko, J. L.; Stryer, L. (2002). Biochemistry (5th ed.). New York: W H Freeman. ISBN 978-1-4641-2610-9.
- ↑ Shambaugh, G. E. (June 1979). "Pyrimidine biosynthesis". The American Journal of Clinical Nutrition 32 (6): 1290–1297. doi:10.1093/ajcn/32.6.1290. PMID 35970.
- ↑ Garrett, Reginald H.; Grisham, Charles M. (2013). Biochemistry (6th ed.). Belmont, CA: Brooks/Cole, Cengage Learning. pp. 949. ISBN 9781133106296.

