Chemistry:Cyclic guanosine monophosphate
| |
| |
| Names | |
|---|---|
| IUPAC name
Guanosine 3′,5′-(hydrogen phosphate)
| |
| Systematic IUPAC name
2-Amino-9-[(4aR,6R,7R,7aS)-2,7-dihydroxy-2-oxotetrahydro-2H,4H-2λ5-furo[3,2-d][1,3,2]dioxaphosphol-6-yl]-3,9-dihydro-6H-purin-6-one | |
| Other names
cGMP; 3′,5′-cyclic GMP; 3′:5′-cyclic GMP; Guanosine cyclic monophosphate; Cyclic 3′,5′-GMP; Guanosine 3′,5′-cyclic phosphate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| MeSH | Cyclic+GMP |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C10H12N5O7P | |
| Molar mass | 345.208 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Cyclic guanosine monophosphate (cGMP) is a cyclic nucleotide derived from guanosine triphosphate (GTP). cGMP acts as a second messenger much like cyclic AMP. Its most likely mechanism of action is activation of intracellular protein kinases in response to the binding of membrane-impermeable peptide hormones to the external cell surface.[1] Through protein kinases activation, cGMP can relax smooth muscle.[2] cGMP concentration in urine can be measured for kidney function and diabetes detection.[3]
History
Cyclic guanosine monophosphate (cGMP) research began after cGMP and cyclic adenosine monophosphate (cAMP) were identified as cellular components and potentially involved with cellular regulation.[4] Upon the synthesis of cGMP in 1960,[4] progress rapidly spread in the understanding of regulation and effects of cGMP. Earl W. Sutherland received the 1971 Nobel Prize in Medicine for his work with cAMP and secondary messengers. This award sparked extensive research into cAMP, while cGMP received less attention, with its biological functions largely unknown until the 1980s.[5] During this period, two pivotal discoveries highlighted cGMP's role in cellular signaling: atrial natriuretic peptide (ANP) was found to stimulate cGMP synthesis through the particulate guanylyl cyclase (pGC) receptor, and nitric oxide (NO), identified as the endothelium-derived relaxing factor, was shown to activate soluble guanylyl cyclase (sGC), producing cGMP to mediate vasodilation in smooth muscle cells.[5] Further components involved with the cGMP were also identified such as cGMP-hydrolyzing phosphodiesterases (PDEs) and cGMP-binding proteins.[5] The awarding of the 1998 Nobel Prize to Robert F. Furchgott, Louis J. Ignarro, and Ferid Murad for their discoveries in the NO-cGMP pathway renewed interest in cGMP research with the 1st International Conference on cGMP being held in 2003.[5]
Synthesis
Guanylate cyclase (GC) catalyzes cGMP synthesis. This enzyme converts GTP to cGMP. Peptide hormones such as the atrial natriuretic factor activate membrane-bound GC, while soluble GC (sGC) is typically activated by nitric oxide to stimulate cGMP synthesis. sGC can be inhibited by ODQ (1H-[1,2,4]oxadiazolo[4,3-a]quinoxalin-1-one).[6]

Functions
cGMP acts as a regulator of ion channel conductance, glycogenolysis, cellular apoptosis, and platelet inhibition. cGMP relaxes smooth muscle tissue leading to vasodilation which increases blood flow. Additionally, cGMP is involved with neurogenesis and neuroplasticity. At presynaptic terminals in the striatum, cGMP controls the efficacy of neurotransmitter release.[7]
cGMP is a secondary messenger in phototransduction in the eye. In the photoreceptors of the mammalian eye, the presence of light activates phosphodiesterase, which degrades cGMP. The sodium ion channels in photoreceptors are cGMP-gated, so degradation of cGMP causes sodium channels to close, which leads to the hyperpolarization of the photoreceptor's plasma membrane and ultimately to visual information being sent to the brain.[8]
cGMP is also seen to mediate the switching on of the attraction of apical dendrites of pyramidal cells in cortical layer V towards semaphorin-3A (Sema3a).[9] Whereas the axons of pyramidal cells are repelled by Sema3a, the apical dendrites are attracted to it. The attraction is mediated by the increased levels of soluble guanylate cyclase (sGC) that are present in the apical dendrites. sGC generates cGMP, leading to a sequence of chemical activations that result in the attraction towards Sema3a. The absence of sGC in the axon causes the repulsion from Sema3a. This strategy ensures the structural polarization of pyramidal neurons and takes place in embryonic development.
cGMP, like cAMP, gets synthesized when olfactory receptors receive odorous input. cGMP is produced slowly and has a more sustained life than cAMP, which has implicated it in long-term cellular responses to odor stimulation, such as long-term potentiation. cGMP in the olfactory is synthesized by both membrane guanylyl cyclase (mGC) as well as soluble guanylyl cyclase (sGC). Studies have found that cGMP synthesis in the olfactory is due to sGC activation by nitric oxide, a neurotransmitter. cGMP also requires increased intracellular levels of cAMP and the link between the two second messengers appears to be due to rising intracellular calcium levels.[10]

Pathology
Role in Cardiovascular Events
The nitric oxide (NO)-cyclic guanosine monophosphate (cGMP)-phosphodiesterase (PDE) pathway has become a target in developing treatments for heart failure. A deficit in cGMP levels has been associated with adverse cardiovascular outcomes, promoting factors like myocardial fibrosis, vasoconstriction, and inflammation, all of which accelerate heart failure progression.[11] Some soluble guanylate cyclase (sGC) stimulators, have yielded promising outcomes in reducing cardiovascular events.[11] Their effectiveness is thought to result from increased sensitivity of sGC to endogenous NO.
Elevated plasma cGMP levels, regulated predominantly by natriuretic peptides (NP) rather than nitric oxide (NO), were found to correlate with a higher risk of heart failure, atherosclerotic cardiovascular disease, and coronary heart disease.[12]
Role in Major Depression Disorder
The cGMP signaling pathway plays a role in the regulation of neuroplasticity, an area of interest in understanding the pathophysiology of major depressive disorder (MDD).[13] The cGMP signaling pathway in the brain operates as a second messenger system, amplifying neurotransmitter signals, influencing gene expression and neuronal function. Within neurons, cGMP levels are modulated by guanylate cyclase enzymes, which synthesize cGMP, and by PDEs, which degrade cGMP.[13]
Enhancing cGMP levels, either by stimulating guanylate cyclase or inhibiting PDEs, promotes neurogenesis and synaptic plasticity, particularly in brain regions implicated in MDD, such as the hippocampus and prefrontal cortex.[13] Animal studies also demonstrate that chronic antidepressant treatment can elevate cGMP levels in these areas.[13] Genetic research has further highlighted specific polymorphisms in PDE genes associated with MDD susceptibility and treatment response.[13]
Role in Infectious Disease Pathogenesis
Certain pathogens, such as Enterotoxigenic Escherichia coli (ETEC), elevate cGMP to evade host immune defenses and establish infection. ETEC's heat-stable toxin induces significant cGMP production within intestinal epithelial cells, and this cGMP is often secreted into the extracellular space, where it serves as a signaling molecule.[14] Extracellular cGMP, in turn, triggers the release of IL-33 release which modulate inflammation and impact the immune system's ability to mount effective responses, dampening both innate and adaptive immunity.[14][15]
Degradation
Numerous cyclic nucleotide phosphodiesterases (PDE) can degrade cGMP by hydrolyzing cGMP into 5'-GMP. PDE 5, -6 and -9 are cGMP-specific while PDE1, -2, -3, -10 and -11 can hydrolyse both cAMP and cGMP.
Phosphodiesterase inhibitors prevent the degradation of cGMP, thereby enhancing and/or prolonging its effects. For example, Sildenafil (Viagra) and similar drugs enhance the vasodilatory effects of cGMP within the corpus cavernosum by inhibiting PDE 5 (or PDE V). This is used as a treatment for erectile dysfunction. However, the drug can inhibit PDE6 in retina (albeit with less affinity than PDE5). This has been shown to result in loss of visual sensitivity but is unlikely to impair common visual tasks, except under conditions of reduced visibility when objects are already near visual threshold.[16] This effect is largely avoided by other PDE5 inhibitors, such as tadalafil.[17]
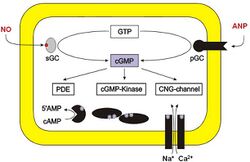
Protein kinase activation
The cGMP-dependent protein kinase (PKG) activation pathway begins with the production of cGMP by guanylyl cyclase enzymes, which can be activated by signaling molecules such as nitric oxide (NO) or natriuretic peptides. Elevated cGMP levels then lead to the activation of some protein-dependent kinases like PKG.[5] For example, PKG (protein kinase G) is a dimer consisting of one catalytic and one regulatory unit, with the regulatory units blocking the active sites of the catalytic units.
cGMP binds to sites on the regulatory units of PKG and activates the catalytic units, enabling them to phosphorylate their substrates. Unlike with the activation of some other protein kinases, notably PKA, the PKG is activated but the catalytic and regulatory units do not disassociate.
Once activated, PKG phosphorylates various target proteins, altering their function and contributing to cellular processes such as smooth muscle relaxation, ion channel regulation, and inhibition of platelet aggregation. This pathway is also significant in cardiovascular physiology, where it helps maintain vascular tone and blood pressure.[11]
See also
- Cyclic adenosine monophosphate (cAMP)
- 8-Bromoguanosine 3',5'-cyclic monophosphate (8-Br-cGMP)
- Guanosine triphosphate (GTP)
- Guanylate cyclase
- Protein Kinase G
References
- ↑ "Cyclic nucleotide-dependent protein kinases: intracellular receptors for cAMP and cGMP action". Critical Reviews in Clinical Laboratory Sciences 36 (4): 275–328. August 1999. doi:10.1080/10408369991239213. PMID 10486703.
- ↑ "Molecular mechanism of cGMP-mediated smooth muscle relaxation". Journal of Cellular Physiology 184 (3): 409–420. September 2000. doi:10.1002/1097-4652(200009)184:3<409::aid-jcp16>3.0.co;2-k. PMID 10911373.
- ↑ Shimosawa, Tatsuo, ed (2018-04-12). "Urinary cGMP predicts major adverse renal events in patients with mild renal impairment and/or diabetes mellitus before exposure to contrast medium". PLOS ONE 13 (4). doi:10.1371/journal.pone.0195828. PMID 29649334. Bibcode: 2018PLoSO..1395828C.
- ↑ 4.0 4.1 Kots, Alexander Y.; Martin, Emil; Sharina, Iraida G.; Murad, Ferid (2009), Schmidt, Harald H. H. W.; Hofmann, Franz; Stasch, Johannes-Peter, eds., "A Short History of cGMP, Guanylyl Cyclases, and cGMP-Dependent Protein Kinases" (in en), cGMP: Generators, Effectors and Therapeutic Implications (Berlin, Heidelberg: Springer Berlin Heidelberg) 191: pp. 1–14, doi:10.1007/978-3-540-68964-5_1, ISBN 978-3-540-68960-7, PMID 19089322, PMC 3932363, http://link.springer.com/10.1007/978-3-540-68964-5_1, retrieved 2024-11-27
- ↑ 5.0 5.1 5.2 5.3 5.4 Feil, Robert; Kemp-Harper, Barbara (Feb 2006). "cGMP signalling: from bench to bedside: Conference on cGMP Generators, Effectors and Therapeutic Implications" (in en). EMBO Reports 7 (2): 149–153. doi:10.1038/sj.embor.7400627. ISSN 1469-221X. PMID 16439998.
- ↑ "Potent and selective inhibition of nitric oxide-sensitive guanylyl cyclase by 1H-[1,2,4oxadiazolo[4,3-a]quinoxalin-1-one"]. Molecular Pharmacology 48 (2): 184–188. August 1995. PMID 7544433. https://molpharm.aspetjournals.org/content/48/2/184.
- ↑ "Presynaptic cGMP sets synaptic strength in the striatum and is important for motor learning". EMBO Reports 23 (8). August 2022. doi:10.15252/embr.202154361. PMID 35735260.
- ↑ "The pharmacology of cyclic nucleotide-gated channels: emerging from the darkness". Current Pharmaceutical Design 12 (28): 3597–3613. 2006. doi:10.2174/138161206778522100. NIHMSID: NIHMS47625. PMID 17073662.
- ↑ "Semaphorin 3A is a chemoattractant for cortical apical dendrites". Nature 404 (6778): 567–573. April 2000. doi:10.1038/35007001. PMID 10766232. Bibcode: 2000Natur.404..567P.
- ↑ "Interplay among cGMP, cAMP, and Ca2+ in living olfactory sensory neurons in vitro and in vivo". The Journal of Neuroscience 31 (23): 8395–8405. June 2011. doi:10.1523/JNEUROSCI.6722-10.2011. PMID 21653844.
- ↑ 11.0 11.1 11.2 Emdin, Michele; Aimo, Alberto; Castiglione, Vincenzo; Vergaro, Giuseppe; Georgiopoulos, Georgios; Saccaro, Luigi Francesco; Lombardi, Carlo Mario; Passino, Claudio et al. (2020-10-13). "Targeting Cyclic Guanosine Monophosphate to Treat Heart Failure: JACC Review Topic of the Week". Journal of the American College of Cardiology 76 (15): 1795–1807. doi:10.1016/j.jacc.2020.08.031. ISSN 0735-1097. https://linkinghub.elsevier.com/retrieve/pii/S0735109720363336.
- ↑ Zhao, Di; Guallar, Eliseo; Vaidya, Dhananjay; Ndumele, Chiadi E.; Ouyang, Pamela; Post, Wendy S.; Lima, Joao A.; Ying, Wendy et al. (2020-01-21). "Cyclic Guanosine Monophosphate and Risk of Incident Heart Failure and Other Cardiovascular Events: the ARIC Study" (in en). Journal of the American Heart Association 9 (2). doi:10.1161/JAHA.119.013966. ISSN 2047-9980. PMID 31928156.
- ↑ 13.0 13.1 13.2 13.3 13.4 W. Reierson, Gillian; Guo, Shuyu; Mastronardi, Claudio; Licinio, Julio; Wong, Ma-Li (2011-12-01). "cGMP Signaling, Phosphodiesterases and Major Depressive Disorder" (in en). Current Neuropharmacology 9 (4): 715–727. doi:10.2174/157015911798376271. PMID 22654729. PMC 3263465. http://www.eurekaselect.com/openurl/content.php?genre=article&issn=1570-159X&volume=9&issue=4&spage=715.
- ↑ 14.0 14.1 Motyka, Natalya I.; Stewart, Sydney R.; Hollifield, Ian E.; Kyllo, Thomas R.; Mansfield, Joshua A.; Norton, Elizabeth B.; Clements, John D.; Bitoun, Jacob P. (2021-03-17). Torres, Victor J.. ed. "Elevated Extracellular cGMP Produced after Exposure to Enterotoxigenic Escherichia coli Heat-Stable Toxin Induces Epithelial IL-33 Release and Alters Intestinal Immunity" (in en). Infection and Immunity 89 (4). doi:10.1128/IAI.00707-20. ISSN 0019-9567. PMID 33431701. PMC 8090939. https://journals.asm.org/doi/10.1128/IAI.00707-20.
- ↑ Wang, Haixiu; Zhong, Zifu; Luo, Yu; Cox, Eric; Devriendt, Bert (2019-01-08). "Heat-Stable Enterotoxins of Enterotoxigenic Escherichia coli and Their Impact on Host Immunity" (in en). Toxins 11 (1): 24. doi:10.3390/toxins11010024. ISSN 2072-6651. PMID 30626031.
- ↑ "The effect of sildenafil citrate (Viagra) on visual sensitivity". Journal of Vision 7 (8): 4. June 2007. doi:10.1167/7.8.4. PMID 17685811.
- ↑ "The discovery of tadalafil: a novel and highly selective PDE5 inhibitor. 2: 2,3,6,7,12,12a-hexahydropyrazino[1',2':1,6]pyrido[3,4-b]indole-1,4-dione analogues". Journal of Medicinal Chemistry 46 (21): 4533–4542. October 2003. doi:10.1021/jm0300577. PMID 14521415.
 |
