Chemistry:Silver fulminate
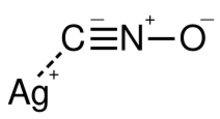
| |
| Names | |
|---|---|
| IUPAC names | |
| Other names
Silver fulminate
Silver(I) fulminate Argentous fulminate | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| AgCNO | |
| Molar mass | 149.885 g/mol |
| Density | 3.938 g/cm3 |
| Explosive data | |
| Shock sensitivity | Extremely high |
| Friction sensitivity | Extremely high |
| Hazards | |
| Main hazards | Sensitive high explosive |
| NFPA 704 (fire diamond) | |
| 170 °C (338 °F; 443 K) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Silver fulminate (AgCNO) is the highly explosive silver salt of fulminic acid.
Silver fulminate is a primary explosive, but has limited use as such due to its extreme sensitivity to impact, heat, pressure, and electricity. The compound becomes progressively sensitive as it is aggregated, even in small amounts; the touch of a falling feather, the impact of a single water droplet, or a small static discharge are all capable of explosively detonating an unconfined pile of silver fulminate no larger than a dime and no heavier than a few milligrams. Aggregating larger quantities is impossible, due to the compound's tendency to self-detonate under its own weight.
Silver fulminate was first prepared in 1800 by Edward Charles Howard in his research project to prepare a large variety of fulminates. Along with mercury fulminate, it is the only fulminate stable enough for commercial use. Detonators using silver fulminate were used to initiate picric acid in 1885, but since have been used only by the Italian Navy.[3] The current commercial use has been in producing non-damaging novelty noisemakers as children's toys.
Structure
Silver fulminate occurs in two polymorphic forms, an orthorhombic one and a trigonal one with a rhombohedral lattice.[4] The trigonal polymorph consists of cyclic hexamers, (AgCNO)6.[5]
Properties
Fulminates are toxic, about the same as cyanides.[3] When pure, silver fulminate is chemically stable, not decomposing after years of storage. Like many silver salts, it darkens with light exposure. It is slightly soluble in cold water and can be recrystallized using hot water.[3][6] It can also be recrystallized from a 20% solution of ammonium acetate.[3] It is not hygroscopic and can explode when moist or under water; it was reported to remain explosive after 37 years under water.[3] It explodes upon contact with concentrated sulfuric acid or chlorine or bromine, but not when contacting iodine.[3] It is insoluble in nitric acid, but dissolves in ammonia, alkali chlorides, alkali cyanides, aniline, pyridine, and potassium iodide by forming complexes.[3] Concentrated hydrochloric acid decomposes it non-explosively with a hissing noise; thiosulfate also decomposes it non-explosively, and can be used for disposal.[3]
Preparation
This compound can be prepared by pouring a solution of silver nitrate in nitric acid into ethanol, under careful control of the reaction conditions, to avoid an explosion.[7] The reaction is usually done at 80–90 °C; at 30 °C, the precipitate may not form.[3] Only tiny amounts of silver fulminate should be prepared at once, as even the weight of the crystals can cause them to self-detonate.
Another way to make silver fulminate is to react silver carbonate with ammonia in solution.[citation needed]
Silver fulminate also forms when nitrogen oxide gas is passed through a solution of silver nitrate in ethanol.[3]
Silver fulminate can be prepared unintentionally, when an acidic solution of silver nitrate comes in contact with alcohol.[7] This is a hazard in some formulations of chemically silvering mirrors.
Novelty explosive
Silver fulminate, often in combination with potassium chlorate, is used in trick noise-makers known as "throw-downs", "crackers", "snappers", "whippersnappers", "pop-its", or "bang snaps", a popular type of novelty firework. They contain approximately 200 milligrams of fine gravel coated with a minute quantity (approximately 80 micrograms)[8] of silver fulminate. When thrown against a hard surface, the impact is sufficient to detonate the tiny quantity of explosive, creating a small salute from the supersonic detonation. Snaps are designed to be incapable of producing damage (even when detonated against skin) due to the buffering effect provided by the much greater mass of the gravel medium. It is also the chemical found in Christmas crackers[9] having first been used for that purpose by Tom Smith in 1860. The chemical is painted on one of two narrow strips of card, with abrasive on the second. When the cracker is pulled, the abrasive detonates the silver fulminate.
A fulminate mixture with 10–20% potassium chlorate is cheaper and more brisant than the fulminate alone.[3]
Silver fulminate and "fulminating silver"
Silver fulminate is often confused with silver nitride, silver azide, or fulminating silver. "Fulminating silver", though always referring to an explosive silver-containing substance, is an ambiguous term. While it may be a synonym of silver fulminate, it may also refer to the nitride or azide, the decomposition product of Tollen's reagent, or an alchemical mixture, which does not contain the fulminate anion.
See also
- Primary explosive
- Justus von Liebig
- Friedrich Woehler
- Silver cyanate
- Isomerism
- Fulminate
- Fulminic acid
- Potassium fulminate
- Mercury(II) fulminate
References
- ↑ International Union of Pure and Applied Chemistry (2005). Nomenclature of Inorganic Chemistry (IUPAC Recommendations 2005). Cambridge (UK): RSC–IUPAC. ISBN:0-85404-438-8. p. 291. Electronic version.
- ↑ "Silver fulminate". ChemBase. http://www.chembase.com/cbid_62585.htm. Retrieved 8 June 2012.[yes|permanent dead link|dead link}}]
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 Matyas, Robert; Pachman, Jiri (Mar 12, 2013). Primary Explosives. Springer Science & Business Media, 338 pages.
- ↑ Britton, D.; Dunitz, J. D. (1965). "The Crystal Structure of Silver Fulminate". Acta Crystallographica 19 (4): 662–668. doi:10.1107/S0365110X6500405X. Bibcode: 1965AcCry..19..662B.
- ↑ Britton, D. (1991). "A Redetermination of the Trigonal Silver Fulminate Structure". Acta Crystallographica C 47 (12): 2646–2647. doi:10.1107/S0108270191008855. Bibcode: 1991AcCrC..47.2646B.
- ↑ 0.0075 gm at 13 °C, 0.018 gm at 30 °C, and 0.25 gm per 100 gm H2O at 100 °C
- ↑ 7.0 7.1 Collins, P. H.; Holloway, K. J. (1978). "A Reappraisal of silver fulminate as a detonant". Propellants, Explosives, Pyrotechnics 3 (6): 159–162. doi:10.1002/prep.19780030603.
- ↑ package disclosure of Alexron Co. Ltd, Hong Kong, Phantom Brand Bang Snaps, (c)2013
- ↑ Spectrum. "Comment #70". Old Firework Factory Locations. UK Pyrotechnics Society. http://www.pyrosociety.org.uk/forum/topic/4101-old-firework-factory-locations/page__st__60. Retrieved 20 December 2011.
Further reading
- Singh, K. (1959). "Crystal structure of silver fulminate". Acta Crystallographica 12 (12): 1053. doi:10.1107/S0365110X5900295X. Bibcode: 1959AcCry..12.1053S.
 |




