Biology:Lipocalin
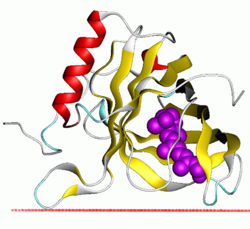 | |||||||||
| Identifiers | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Symbol | Lipocalin | ||||||||
| Pfam | PF00061 | ||||||||
| Pfam clan | CL0116 | ||||||||
| InterPro | IPR000566 | ||||||||
| PROSITE | PDOC00187 | ||||||||
| SCOP2 | 1hms / SCOPe / SUPFAM | ||||||||
| OPM superfamily | 50 | ||||||||
| OPM protein | 1kt6 | ||||||||
| |||||||||
| Lipocalin-like domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
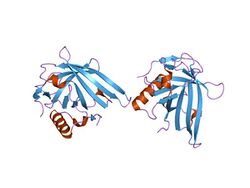 Structure of the Escherichia coli lipocalin.[1] | |||||||||
| Identifiers | |||||||||
| Symbol | Lipocalin_2 | ||||||||
| Pfam | PF08212 | ||||||||
| Pfam clan | CL0116 | ||||||||
| InterPro | IPR013208 | ||||||||
| |||||||||
The lipocalins are a family of proteins which transport small hydrophobic molecules such as steroids, bilins, retinoids, and lipids, and most lipocalins are also able to bind to complexed iron (via siderophores[2] or flavonoids[3]) as well as heme.[4] They share limited regions of sequence homology and a common tertiary structure architecture.[5][6][7][8][9] This is an eight stranded antiparallel beta barrel with a repeated + 1 topology enclosing an internal ligand binding site.[7][8]
These proteins are found in gram negative bacteria, vertebrate cells, and invertebrate cells, and in plants. Lipocalins have been associated with many biological processes, among them immune response, pheromone transport, biological prostaglandin synthesis, retinoid binding, and cancer cell interactions.[10]
Family members
The name "lipocalin" has been proposed[5] for this protein family, but cytosolic fatty acid binding proteins are also included. The sequences of most members of the family, the core or kernel lipocalins, are characterised by three short conserved stretches of residues, while others, the outlier lipocalin group, share only one or two of these.[8][11] Proteins known to belong to this family include alpha-1-microglobulin (protein HC); major urinary proteins; alpha-1-acid glycoprotein (orosomucoid);[12] aphrodisin; apolipoprotein D; beta-lactoglobulin; complement component C8 gamma chain;[13] crustacyanin;[14] epididymal-retinoic acid binding protein (E-RABP);[15] insectacyanin; odorant binding protein (OBP); human pregnancy-associated endometrial alpha-2 globulin (PAEP); probasin (PB), a prostatic protein; prostaglandin D synthase;[16] purpurin; Von Ebner's gland protein (VEGP);[17] and lizard epididymal secretory protein IV (LESP IV).[18]
Human proteins that contain lipocalin domain include:
- AMBP, APOD
- C8G, CRABP1, CRABP2
- FABP1, FABP2, FABP3, FABP4, FABP5, FABP6, FABP7
- LCN1, LCN2, LCN8, LCN9, LCN10, LCN12
- OBP2A, OBP2B
- ORM1, ORM2
- PAEP, PERF15, PMP2, PTGDS
- RBP1, RBP2, RBP4, RBP5, RBP7
- UNQ2541
Structure
Although lipocalins are a broad family of greatly varied proteins, their three-dimensional structure is a unifying characteristic. Lipocalins have an eight-stranded, antiparallel, symmetrical β-barrel fold, which is, in essence, a beta sheet which has been rolled into a cylindrical shape. Inside this barrel is located a ligand binding site, which plays an important role in the lipocalin classification as a transport protein.[19] If lipocalins are genetically engineered in the attempt to modify their binding properties, they are called anticalins.[20]
Function
Immune response
Lipocalin proteins are important key players of nutritional immunity by withholding and sequestering micronutrients.[21] They are thereby able to regulate inflammatory and detoxification processes caused by immune system activation in mammals. They are known respiratory allergens of mice, cats, dogs, horses, and other animals. Examples of lipocalin proteins involved in immune system responses include alpha-1-microglobulin, alpha-1-acid glycoprotein, and C8gamma. Structural information for many immune system influencing lipocalin proteins is available, while their exact role in biological systems is still somewhat unclear. Lipocalin allergens have been shown to evoke a Th2-deviated immune response, important for allergic sensitization, when applied in their apo-form (with an empty calyx devoid of ligands), whereas the holo-form seemed to exert immune-suppressive properties in vitro.[22]
Pheromone transport
The lipocalin family has been connected with the transport of mammalian pheromones due to easily observable protein-pheromone interactions. Lipocalins are comparatively small in size, and are thus less complicated to study as opposed to large, bulky proteins. They can also bind to various ligands for different biological purposes. Lipocalins have been detected as carrier proteins of important pheromones in the nasal mucus of rodents. Major urinary proteins, a lipocalin subfamily, are found in mouse and rat urine and may act as protein pheromones themselves.[23]
Prostaglandin synthesis
This family of proteins plays a part in the biological system of terminal prostaglandin synthesis.
Retinoid binding
Retinol, (vitamin A), is an important micronutrient that affects eyesight, cell differentiation, immune system function, bone growth, and tumor suppression. Retinol absorption and metabolism depends on lipocalins that act as binding proteins. Retinyl esters (present in meats) and beta-carotene (present in plants) are the two main sources of retinoids in the diet. After intake, they are converted to retinol, successively metabolized, and finally bound to retinol binding proteins (lipocalins) in the blood plasma.
Cancer cell interactions
Because lipocalins are extracellular proteins, their intracellular effects are not obvious, and demand further study. However, lipophilic ligands, present as substituents to the lipocalins, have the ability to enter the cell, where they can act as tumor protease inhibitors. This research suggests another possible route of protein-tumor investigations.
Hormone
Allergens
Some of the proteins in this family are allergens. Allergies are hypersensitivity reactions of the immune system to specific substances called allergens (such as pollen, stings, drugs, or food) that, in most people, result in no symptoms. A nomenclature system has been established for antigens (allergens) that cause IgE-mediated atopic allergies in humans.[24] This nomenclature system is defined by a designation that is composed of the first three letters of the genus; a space; the first letter of the species name; a space and an Arabic number. In the event that two species names have identical designations, they are discriminated from one another by adding one or more letters (as necessary) to each species designation.
The allergens in this family include allergens with the following designations:[25]
- Insects
- German cockroach: Bla g 4
- American cockroach: Per a 4
- Carnivorans
- Domestic dog: Can f 1, Can f 2, Can f 4, Can f 6
- Domestic cat: Fel d 4, Fel d 7
- Rodents
- Guinea pig: Cav p 1, Cav p 2, Cav p 3, Cav p 6
- Golden hamster: Mes a 1
- House mouse: Mus m 1
- Brown rat: Rat n 1
- Siberian hamster: Phod s 1
- Other mammals
- Domestic cattle: Bos d 2, Bos d 5
- Domestic horse: Equ c 1, Equ c 2
- Domestic rabbit: Ory c 1, Ory c 2, Ory c 4
See also
- Retinol binding protein
References
- ↑ "The crystal structure of the Escherichia coli lipocalin Blc suggests a possible role in phospholipid binding". FEBS Letters 562 (1–3): 183–188. March 2004. doi:10.1016/S0014-5793(04)00199-1. PMID 15044022. Bibcode: 2004FEBSL.562..183C.
- ↑ "The neutrophil lipocalin NGAL is a bacteriostatic agent that interferes with siderophore-mediated iron acquisition". Molecular Cell 10 (5): 1033–1043. November 2002. doi:10.1016/s1097-2765(02)00708-6. PMID 12453412.
- ↑ "Linking iron-deficiency with allergy: role of molecular allergens and the microbiome". Metallomics 9 (12): 1676–1692. December 2017. doi:10.1039/c7mt00241f. PMID 29120476.
- ↑ "A lipocalin mediates unidirectional heme biomineralization in malaria parasites". Proceedings of the National Academy of Sciences of the United States of America 117 (28): 16546–16556. July 2020. doi:10.1073/pnas.2001153117. PMID 32601225. Bibcode: 2020PNAS..11716546M.
- ↑ 5.0 5.1 "Homology and structure-function correlations between alpha 1-acid glycoprotein and serum retinol-binding protein and its relatives". FASEB Journal 1 (3): 209–214. September 1987. doi:10.1096/fasebj.1.3.3622999. PMID 3622999.
- ↑ "Structural organization of the gene for prostaglandin D synthase in the rat brain". Proceedings of the National Academy of Sciences of the United States of America 89 (12): 5376–5380. June 1992. doi:10.1073/pnas.89.12.5376. PMID 1608945. Bibcode: 1992PNAS...89.5376I.
- ↑ 7.0 7.1 "Crystallographic refinement of human serum retinol binding protein at 2A resolution". Proteins 8 (1): 44–61. 1990. doi:10.1002/prot.340080108. PMID 2217163.
- ↑ 8.0 8.1 8.2 "Structure and sequence relationships in the lipocalins and related proteins". Protein Science 2 (5): 753–761. May 1993. doi:10.1002/pro.5560020507. PMID 7684291.
- ↑ "The structural motif of beta-lactoglobulin and retinol-binding protein: a basic framework for binding and transport of small hydrophobic molecules?". Trends in Biochemical Sciences 13 (2): 64–66. February 1988. doi:10.1016/0968-0004(88)90031-X. PMID 3238752.
- ↑ "Neutrophil gelatinase-associated lipocalin as an immunomodulator in endocrine hypertension". Front Endocrinol (Lausanne) 13. 2022. doi:10.3389/fendo.2022.1006790. PMID 36387895.
- ↑ "Mouse oncogene protein 24p3 is a member of the lipocalin protein family". Biochemical and Biophysical Research Communications 180 (1): 69–74. October 1991. doi:10.1016/S0006-291X(05)81256-2. PMID 1834059.
- ↑ "Drug binding to human alpha-1-acid glycoprotein in health and disease". Pharmacological Reviews 40 (1): 1–47. March 1988. doi:10.1016/S0031-6997(25)00015-8. PMID 3064105.
- ↑ "Structural and functional characterization of complement C8 gamma, a member of the lipocalin protein family". Molecular Immunology 28 (1–2): 123–131. 1991. doi:10.1016/0161-5890(91)90095-2. PMID 1707134.
- ↑ "Complete sequence and model for the A2 subunit of the carotenoid pigment complex, crustacyanin". European Journal of Biochemistry 197 (2): 407–417. April 1991. doi:10.1111/j.1432-1033.1991.tb15925.x. PMID 2026162.
- ↑ "Structure of the epididymal retinoic acid binding protein at 2.1 A resolution". Structure 1 (1): 7–18. September 1993. doi:10.1016/0969-2126(93)90004-Z. PMID 8069623.
- ↑ "The first lipocalin with enzymatic activity". Trends in Biochemical Sciences 16 (10): 363. October 1991. doi:10.1016/0968-0004(91)90149-P. PMID 1723819.
- ↑ "Structural organization of the genes for rat von Ebner's gland proteins 1 and 2 reveals their close relationship to lipocalins". European Journal of Biochemistry 221 (3): 905–916. May 1994. doi:10.1111/j.1432-1033.1994.tb18806.x. PMID 7514123.
- ↑ "LESP, an androgen-regulated lizard epididymal secretory protein family identified as a new member of the lipocalin superfamily". The Journal of Biological Chemistry 268 (14): 10274–10281. May 1993. doi:10.1016/S0021-9258(18)82200-1. PMID 8486691.
- ↑ Flower, D R; North, A C; Sansom, C E (2000-10-01). "The lipocalin protein family: structural and sequence overview". Biochimica et Biophysica Acta (BBA) - Protein Structure and Molecular Enzymology 1482 (1–2): 9–24. doi:10.1016/s0167-4838(00)00148-5. ISSN 1878-2434. PMID 11058743.
- ↑ Achatz, S; Jarasch, A; Skerra, A (2022-01-01). "Structural plasticity in the loop region of engineered lipocalins with novel ligand specificities, so-called Anticalins". Journal of Structural Biology: X 6. doi:10.1016/j.yjsbx.2021.100054. ISSN 2590-1524. PMID 34988429.
- ↑ Roth-Walter, Franziska (2022). "Iron-Deficiency in Atopic Diseases: Innate Immune Priming by Allergens and Siderophores". Frontiers in Allergy 3. doi:10.3389/falgy.2022.859922. ISSN 2673-6101. PMID 35769558.
- ↑ "The major cow milk allergen Bos d 5 manipulates T-helper cells depending on its load with siderophore-bound iron". PLOS ONE 9 (8). August 2014. doi:10.1371/journal.pone.0104803. PMID 25117976. Bibcode: 2014PLoSO...9j4803R.
- ↑ "Identification of protein pheromones that promote aggressive behaviour". Nature 450 (7171): 899–902. December 2007. doi:10.1038/nature05997. PMID 18064011. Bibcode: 2007Natur.450..899C.
- "Aggression protein found in mice". BBC News. 5 December 2007. https://news.bbc.co.uk/1/hi/sci/tech/7129176.stm.
- ↑ [WHO/IUIS Allergen Nomenclature Subcommittee King T.P., Hoffmann D., Loewenstein H., Marsh D.G., Platts-Mills T.A.E., Thomas W. Bull. World Health Organ. 72:797-806(1994)]
- ↑ "Allergen Search Results: Biochemical name includes "Lipocalin"". https://www.allergen.org/search.php?allergenname=&allergensource=&TaxSource=&TaxOrder=&foodallerg=all&bioname=Lipocalin.
Further reading
- "The lipocalin website". Biochimica et Biophysica Acta (BBA) - Protein Structure and Molecular Enzymology 1482 (1–2): 351–352. October 2000. doi:10.1016/S0167-4838(00)00166-7. PMID 11058775.
- "Important animal allergens are lipocalin proteins: why are they allergenic?". International Archives of Allergy and Immunology 120 (4): 247–258. December 1999. doi:10.1159/000024277. PMID 10640908.
- "Lipocalins and cancer". Biochimica et Biophysica Acta (BBA) - Protein Structure and Molecular Enzymology 1482 (1–2): 318–326. October 2000. doi:10.1016/S0167-4838(00)00154-0. PMID 11058772.
- "Identification, expression, and evolutionary analyses of plant lipocalins". Plant Physiology 139 (4): 2017–2028. December 2005. doi:10.1104/pp.105.070466. PMID 16306142.
- "Pheromones, binding proteins and receptor responses in rodents". Biochemical Society Transactions 31 (Pt 1): 117–122. February 2003. doi:10.1042/BST0310117. PMID 12546667.
External links
- Lipocalins in SCOP database
- UMich Orientation of Proteins in Membranes families/superfamily-52 - Calculated spatial positions of some Lipocalins in membranes
 |
