Biology:Von Hippel–Lindau tumor suppressor
 Generic protein structure example |
The Von Hippel–Lindau tumor suppressor also known as pVHL is a protein that, in humans, is encoded by the VHL gene. Mutations of the VHL gene are associated with Von Hippel–Lindau disease, which is characterized by hemangioblastomas of the brain, spinal cord and retina. It is also associated with kidney and pancreatic lesions. [1]
Function
The protein encoded by the VHL gene is the substrate recognition component of a protein complex that includes elongin B, elongin C, and cullin-2, and possesses E3 ubiquitin ligase activity. This complex is involved in the ubiquitination and subsequent degradation of hypoxia-inducible factors (HIFs), which are transcription factors that play a central role regulating gene expression in response to changing oxygen levels. RNA polymerase II subunit POLR2G/RPB7 is also reported to be a target of this protein. Alternatively spliced transcript variants encoding distinct isoforms have been observed.[2]
The resultant protein is produced in two forms, an 18 kDa and a 30 kDa protein that functions as a tumor suppressor. The main action of the VHL protein is thought to be its E3 ubiquitin ligase activity that results in specific target proteins being 'marked' for degradation.
The most researched of these targets is hypoxia inducible factor 1a (HIF1a), a transcription factor that induces the expression of a number of angiogenesis related factors.[3]
HIFs are necessary for tumor growth because most cancers demand high metabolic activity and are only supplied by structurally or functionally inadequate vasculature. Activation of HIFs allow for enhanced angiogenesis, which in turn allow for increased glucose uptake. While HIFs are mostly active in hypoxic conditions, VHL-defective renal carcinoma cells show constitutive activation of HIF even in oxygenated environments.
It is clear that VHL and HIFs interact closely. Firstly, all renal cell carcinoma mutations in VHL that have been tested affect the protein's ability to modify HIF. Additionally, HIF activation can be detected in the earliest events in tumorigenesis in patients with VHL syndrome. In normal cells in hypoxic conditions, HIF1A is activated with little activation of HIF2A. However, in tumors the balance of HIF1A and HIF2A is tipped towards HIF2A. While HIF1A serves as a pro-apoptotic factor, HIF2A interacts with cyclin D1. This leads to increased survival due to lower rates of apoptosis and increased proliferation due to the activation of cyclin D1.[4] Recent genome-wide analysis (GWAS) of HIF binding in kidney cancer showed that HIF1A binds upstream of majorly good prognosis genes, while HIF2A binds upstream to majorly poor prognosis genes. This indicates that the HIF transcription factor distribution in kidney cancer is of major importance in determining the outcome of the patients.[5]
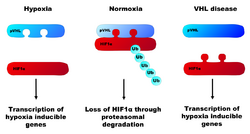
In the normal cell with active VHL protein, HIF alpha is regulated by hydroxylation in the presence of oxygen. When iron, 2-oxoglutarate and oxygen are present, HIF is inactivated by HIF hydroxylases. Hydroxylation of HIF creates a binding site for pVHL (the protein product of the VHL gene).[6] pVHL directs the polyubiquitylation of HIF1A, ensuring that this protein will be degraded by the proteasome. In hypoxic conditions, HIF1A subunits accumulate and bind to HIFB. This heterodimer of HIF is a transcription factor that activates genes that encode for proteins such as vascular endothelial growth factor (VEGF) and erythropoietin, proteins that are both involved in angiogenesis. Cells with abnormal pVHL are unable to disrupt the formation of these dimers, and therefore behave like they are hypoxic even in oxygenated environments.
HIF has also been linked to mTOR, a central controller of growth decisions. It has recently been shown that HIF activation can inactivate mTOR.[7]
HIF can help explain the organ-specific nature of VHL syndrome. It has been theorized that constitutively activating HIF in any cell could lead to cancer, but that there are redundant regulators of HIF in organs not affected by VHL syndrome. This theory has been disproved multiple times since in all cell types loss of VHL function leads to constitutive activation of HIF and its downstream effects. Another theory holds that although in all cells loss of VHL leads to activation of HIF, in most cells this leads to no advantage in proliferation or survival. Additionally, the nature of the mutation in the VHL protein leads to phenotypic manifestations in the pattern of cancer that develops. Nonsense or deletion mutations of VHL protein have been linked to type 1 VHL with a low risk of pheochromocytoma (adrenal gland tumors). Type 2 VHL has been linked to missense mutations and is linked to a high risk of pheochromocytoma. Type 2 has also been further subdivided based on risks of renal cell carcinoma. In types 1, 2A and 2B the mutant pVHL is defective in HIF regulation, while type 2C mutant are defective in protein kinase C regulation.[6] These genotype–phenotype correlations suggest that missense mutations of pVHL lead to a 'gain of function' protein.[8]
The involvement in VHL in renal cell cancer can be rationalized via multiple characteristics of renal cells. First, they are more sensitive to the effects of growth factors created downstream of HIF activation than other cells. Secondly, the link to Cyclin D1 (as mentioned above) is only seen in renal cells. Finally, many cells in the kidney normally operate under hypoxic conditions. This may give them a proliferative advantage over other cells while in hypoxic environments.[6]
In addition to its interaction with HIF the VHL protein can also associate with tubulin.[9] It is then capable to stabilize and thus elongate microtubules. This function plays a key role in the stabilisation of the spindle during mitosis. Deletion of VHL causes a drastic increase of misorientated and rotating spindles during mitosis. Through a not-yet-known mechanism, VHL also increases the concentration of MAD2, an important protein of the spindle checkpoint. Thus VHL loss leads to a weakened checkpoint and subsequently chromosome missegregation and aneuploidy.[10]
Pathology
Von Hippel–Lindau syndrome (VHL) is a dominantly inherited hereditary cancer syndrome predisposing to a variety of malignant and benign tumors of the eye, brain, spinal cord, kidney, pancreas, and adrenal glands. A germline mutation of this gene is the basis of familial inheritance of VHL syndrome. Individuals with VHL syndrome inherit one mutation in the VHL protein that causes the protein's normal function to be lost or altered. Over time, sporadic mutation in the second copy of the VHL protein can lead to carcinomas, in particular hemangioblastomas affecting the liver and kidneys, renal (and vaginal) clear cell adenocarcinomas.
The loss of VHL protein activity results in an increased amount of HIF1a, and thus increased levels of angiogenic factors, including VEGF and PDGF. In turn, this leads to unregulated blood vessel growth, one of the prerequisites of a tumor. Additionally, VHL has been implicated in maintaining the differentiated phenotype in renal cells.[4] Furthermore, cell culture experiments with VHL -/- cells have shown that the addition of pVHL can induce a mesenchymal to epithelial transition. This evidence suggests that VHL has a central role in maintaining a differentiated phenotype in the cell.[6]
Additionally, pVHL is important for extracellular matrix formation.[8] This protein may also be important in inhibition of matrix metalloproteinases. These ideas are extremely important in the metastasis of VHL-deficient cells. In classical VHL disease a single wild-type allele in VHL appears to be sufficient to maintain normal cardiopulmonary function.[11]
Treatment
Suggested targets for VHL-related cancers include targets of the HIF pathway, such as VEGF. Inhibitors of VEGF receptor sorafenib, sunitinib, pazopanib, and recently axitinib have been approved by the FDA.[6] The mTOR inhibitor rapamycin[12] analogs everolimus and temsirolimus or VEGF monoclonal antibody bevacizumab may also be an option.
Since iron, 2-oxoglutarate and oxygen are necessary for the inactivation of HIF, it has been theorized that a lack of these cofactors could reduce the ability of hydroxylases in inactivating HIF. A recent study has shown that in cells with a high activation of HIF even in oxygenated environments was reversed by supplying the cells with ascorbate.[13] Thus, Vitamin C may be a potential treatment for HIF induced tumors.
Interactions
Von Hippel–Lindau tumor suppressor has been shown to interact with:
- CCDC82,
- CUL2,[14][15][16][17][18]
- Filamin,[19][20]
- HIF1AN,[21]
- HIF1A,[21][22][23][24][25][26][27][28][29][30]
- Nerve Growth factor IB,[22]
- PHF17,[19]
- PSMC3,[24]
- SAP130,[14]
- TCEB1,[15][16][17][20][22][23][31]
- TCEB2,[14][15][16][17][23] and
- USP33.[31]
See also
References
- ↑ "Von Hippel–Lindau Syndrome". Hormone Research in Paediatrics 84 (3): 145–52. 2015. doi:10.1159/000431323. PMID 26279462.
- ↑ "Entrez Gene: VHL von Hippel–Lindau tumor suppressor". https://www.ncbi.nlm.nih.gov/sites/entrez?Db=gene&Cmd=ShowDetailView&TermToSearch=7428.
- ↑ "von Hippel–Lindau tumor suppressor: not only HIF's executioner". Trends in Molecular Medicine 10 (4): 146–9. April 2004. doi:10.1016/j.molmed.2004.02.004. PMID 15162797.
- ↑ 4.0 4.1 Maxwell, 2005
- ↑ "Heterogeneous Effects of Direct Hypoxia Pathway Activation in Kidney Cancer". PLOS ONE 10 (8): e0134645. 2015. doi:10.1371/journal.pone.0134645. PMID 26262842. Bibcode: 2015PLoSO..1034645S.
- ↑ 6.0 6.1 6.2 6.3 6.4 "The von Hippel–Lindau tumor suppressor protein and clear cell renal carcinoma". Clinical Cancer Research 13 (2 Pt 2): 680s–684s. January 2007. doi:10.1158/1078-0432.CCR-06-1865. PMID 17255293.
- ↑ "Regulation of mTOR function in response to hypoxia by REDD1 and the TSC1/TSC2 tumor suppressor complex". Genes & Development 18 (23): 2893–904. December 2004. doi:10.1101/gad.1256804. PMID 15545625.
- ↑ 8.0 8.1 "Molecular basis of the VHL hereditary cancer syndrome". Nature Reviews. Cancer 2 (9): 673–82. September 2002. doi:10.1038/nrc885. PMID 12209156.
- ↑ "The von Hippel–Lindau tumor suppressor protein influences microtubule dynamics at the cell periphery". Experimental Cell Research 301 (2): 139–46. December 2004. doi:10.1016/j.yexcr.2004.07.016. PMID 15530850.
- ↑ "VHL loss causes spindle disorientation and chromosome instability". Nature Cell Biology 11 (8): 994–1001. 2009. doi:10.1038/ncb1912. PMID 19620968.
- ↑ "Cardiopulmonary function in two human disorders of the hypoxia-inducible factor (HIF) pathway: von Hippel–Lindau disease and HIF-2alpha gain-of-function mutation". FASEB Journal 25 (6): 2001–11. June 2011. doi:10.1096/fj.10-177378. PMID 21389259.
- ↑ "The von Hippel–Lindau tumor suppressor gene and kidney cancer". Clinical Cancer Research 10 (18 Pt 2): 6290S–5S. September 2004. doi:10.1158/1078-0432.CCR-sup-040025. PMID 15448019.
- ↑ "Effect of ascorbate on the activity of hypoxia-inducible factor in cancer cells". Cancer Research 63 (8): 1764–8. April 2003. PMID 12702559.
- ↑ 14.0 14.1 14.2 "Association of SAP130/SF3b-3 with Cullin-RING ubiquitin ligase complexes and its regulation by the COP9 signalosome". BMC Biochemistry 9: 1. 2008. doi:10.1186/1471-2091-9-1. PMID 18173839.
- ↑ 15.0 15.1 15.2 "Large-scale mapping of human protein-protein interactions by mass spectrometry". Molecular Systems Biology 3: 89. 2007. doi:10.1038/msb4100134. PMID 17353931.
- ↑ 16.0 16.1 16.2 "Synthetic peptides define critical contacts between elongin C, elongin B, and the von Hippel–Lindau protein". The Journal of Clinical Investigation 104 (11): 1583–91. December 1999. doi:10.1172/JCI8161. PMID 10587522.
- ↑ 17.0 17.1 17.2 Zhang, Baohong, ed (2008). "VHL type 2B mutations retain VBC complex form and function". PLOS ONE 3 (11): e3801. doi:10.1371/journal.pone.0003801. PMID 19030229. Bibcode: 2008PLoSO...3.3801H.
- ↑ "Muf1, a novel Elongin BC-interacting leucine-rich repeat protein that can assemble with Cul5 and Rbx1 to reconstitute a ubiquitin ligase". The Journal of Biological Chemistry 276 (32): 29748–53. August 2001. doi:10.1074/jbc.M103093200. PMID 11384984.
- ↑ 19.0 19.1 "The von Hippel–Lindau tumor suppressor stabilizes novel plant homeodomain protein Jade-1". The Journal of Biological Chemistry 277 (42): 39887–98. October 2002. doi:10.1074/jbc.M205040200. PMID 12169691.
- ↑ 20.0 20.1 "Identification of a novel protein (VBP-1) binding to the von Hippel–Lindau (VHL) tumor suppressor gene product". Cancer Research 56 (13): 2881–5. July 1996. PMID 8674032.
- ↑ 21.0 21.1 "FIH-1: a novel protein that interacts with HIF-1alpha and VHL to mediate repression of HIF-1 transcriptional activity". Genes & Development 15 (20): 2675–86. October 2001. doi:10.1101/gad.924501. PMID 11641274.
- ↑ 22.0 22.1 22.2 "Nur77 upregulates HIF-alpha by inhibiting pVHL-mediated degradation". Experimental & Molecular Medicine 40 (1): 71–83. February 2008. doi:10.3858/emm.2008.40.1.71. PMID 18305400.
- ↑ 23.0 23.1 23.2 "Structure of an HIF-1alpha -pVHL complex: hydroxyproline recognition in signaling". Science 296 (5574): 1886–9. June 2002. doi:10.1126/science.1073440. PMID 12004076. Bibcode: 2002Sci...296.1886M.
- ↑ 24.0 24.1 "Tat-binding protein-1, a component of the 26S proteasome, contributes to the E3 ubiquitin ligase function of the von Hippel–Lindau protein". Nature Genetics 35 (3): 229–37. November 2003. doi:10.1038/ng1254. PMID 14556007.
- ↑ "The VHL protein recruits a novel KRAB-A domain protein to repress HIF-1alpha transcriptional activity". The EMBO Journal 22 (8): 1857–67. April 2003. doi:10.1093/emboj/cdg173. PMID 12682018.
- ↑ "Mechanism of regulation of the hypoxia-inducible factor-1 alpha by the von Hippel–Lindau tumor suppressor protein". The EMBO Journal 19 (16): 4298–309. August 2000. doi:10.1093/emboj/19.16.4298. PMID 10944113.
- ↑ "HIF-1alpha binding to VHL is regulated by stimulus-sensitive proline hydroxylation". Proceedings of the National Academy of Sciences of the United States of America 98 (17): 9630–5. August 2001. doi:10.1073/pnas.181341498. PMID 11504942. Bibcode: 2001PNAS...98.9630Y.
- ↑ "STAT3 inhibits the degradation of HIF-1alpha by pVHL-mediated ubiquitination". Experimental & Molecular Medicine 40 (5): 479–85. October 2008. doi:10.3858/emm.2008.40.5.479. PMID 18985005.
- ↑ "Identification of an alternative mechanism of degradation of the hypoxia-inducible factor-1alpha". The Journal of Biological Chemistry 283 (43): 29375–84. October 2008. doi:10.1074/jbc.M805919200. PMID 18694926.
- ↑ "Nitric oxide donor, (+/-)-S-nitroso-N-acetylpenicillamine, stabilizes transactive hypoxia-inducible factor-1alpha by inhibiting von Hippel–Lindau recruitment and asparagine hydroxylation". Molecular Pharmacology 74 (1): 236–45. July 2008. doi:10.1124/mol.108.045278. PMID 18426857.
- ↑ 31.0 31.1 "Ubiquitination of a novel deubiquitinating enzyme requires direct binding to von Hippel–Lindau tumor suppressor protein". The Journal of Biological Chemistry 277 (7): 4656–62. February 2002. doi:10.1074/jbc.M108269200. PMID 11739384.
Further reading
- The von Hippel–Lindau tumor suppressor complex and regulation of hypoxia-inducible transcription. 85. 2003. 1–12. doi:10.1016/S0065-230X(02)85001-1. ISBN 978-0120066858.
- "von Hippel–Lindau tumor suppressor: not only HIF's executioner". Trends in Molecular Medicine 10 (4): 146–9. April 2004. doi:10.1016/j.molmed.2004.02.004. PMID 15162797.
- "Formation of primary cilia in the renal epithelium is regulated by the von Hippel–Lindau tumor suppressor protein". Journal of the American Society of Nephrology 17 (7): 1801–6. July 2006. doi:10.1681/ASN.2006020181. PMID 16775032.
- "The von Hippel–Lindau tumor suppressor gene expression level has prognostic value in neuroblastoma". International Journal of Cancer 119 (3): 624–9. August 2006. doi:10.1002/ijc.21888. PMID 16506218.
- "The von Hippel–Lindau tumor suppressor gene and kidney cancer". Clinical Cancer Research 10 (18 Pt 2): 6290S–5S. September 2004. doi:10.1158/1078-0432.CCR-sup-040025. PMID 15448019.
- "The von Hippel–Lindau tumor suppressor protein and clear cell renal carcinoma". Clinical Cancer Research 13 (2 Pt 2): 680s–684s. January 2007. doi:10.1158/1078-0432.CCR-06-1865. PMID 17255293.
- "Roles of SCF and VHL Ubiquitin Ligases in Regulation of Cell Growth". Protein Degradation in Health and Disease. Progress in Molecular and Subcellular Biology. 29. 2002. pp. 1–15. doi:10.1007/978-3-642-56373-7_1. ISBN 978-3-642-62714-9.
- "Molecular pathogenesis of Philadelphia chromosome negative myeloproliferative disorders". Blood Reviews 19 (1): 1–13. January 2005. doi:10.1016/j.blre.2004.02.002. PMID 15572213.
- "von Hippel–Lindau disease". Lancet 361 (9374): 2059–67. June 2003. doi:10.1016/S0140-6736(03)13643-4. PMID 12814730.
- "Clustering of features of von Hippel–Lindau syndrome: evidence for a complex genetic locus". Lancet 337 (8749): 1052–4. May 1991. doi:10.1016/0140-6736(91)91705-Y. PMID 1673491.
- "The role of VHL in the regulation of E-cadherin: a new connection in an old pathway". Cell Cycle 6 (1): 56–9. January 2007. doi:10.4161/cc.6.1.3668. PMID 17245122.
- "Hypoxia and HIF-1 alpha in chondrogenesis". Seminars in Cell & Developmental Biology 16 (4–5): 539–46. 2006. doi:10.1016/j.semcdb.2005.03.003. PMID 16144691.
- "A novel mutation in the von Hippel–Lindau tumor suppressor gene identified in a Japanese family with pheochromocytoma and hepatic hemangioma". Internal Medicine 45 (5): 265–9. 2006. doi:10.2169/internalmedicine.45.1547. PMID 16595991.
- "The VHL Handbook: What You Need to Know about VHL". VHL Family Alliance 12 (1): 1–56. 2005.
External links
- VHL Alliance
- GeneReviews/NCBI/NIH/UW entry on Von Hippel–Lindau Syndrome or Angiomatosis Retinae, VHL Syndrome, von Hippel–Lindau Disease
- Von+Hippel–Lindau+Tumor+Suppressor+Protein at the US National Library of Medicine Medical Subject Headings (MeSH)
 |