Chemistry:Barium carbide
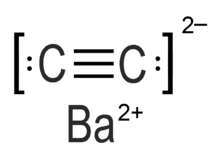
| |
| Names | |
|---|---|
| IUPAC name
Barium ethynediide
| |
| Other names
Barium acetylide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
| |
| |
| Properties | |
| BaC 2 | |
| Molar mass | 161.349 g·mol−1 |
| Appearance | black crystalline solid |
| Density | 3.75 g/cm3 |
| Related compounds | |
Other cations
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Barium carbide (also referred to as barium ethynediide or barium acetylide)[1] is a chemical compound in the carbide family having the chemical formula BaC
2.[2] It consists of barium cations Ba2+ and acetylide anions −
C≡C−
.
Preparation
Barium carbide (BaC2) can be synthesized by several high-temperature reduction methods. An impure form of the compound is obtained by reducing powdered barium carbonate with metallic magnesium in the presence of carbon.[3]
Another laboratory-scale method involves the direct reduction of carbon dioxide with heated metallic barium at approximately 600 °C, producing barium carbide as an intermediate in studies of acetylene formation.[4]
Barium carbide may also be prepared by heating a mixture of a barium amalgam and powdered carbon under a stream of hydrogen, which facilitates the removal of mercury and promotes carbide formation.
The pure compound is typically obtained by carbothermal reduction of barium oxide with carbon at elevated temperatures, a method analogous to the industrial preparation of other alkaline earth metal carbides.[5]
Properties
Barium carbide reacts similarly to calcium carbide,[6] but it's more fusible. When exposed to extreme heat, the barium will evaporate leaving behind crystals of graphite. It can also absorb the carbon in a solution at high temperature.[7]
Hazards
Barium carbide can cause damage to the GI tract and irritation in the skin and eyes.[1]
References
- ↑ 1.0 1.1 "Barium acetylide | C2Ba | ChemSpider". https://www.chemspider.com/Chemical-Structure.13785937.html.
- ↑ "Barium Carbide". https://www.americanelements.com/barium-carbide-50813-65-5.
- ↑ Mishin, V. I.; Georgievskij, S. S.; Eksel', L. M.; Koval', A. I.; Afanas'eva, L. A.; Puchkov, L. D.; Ulybin, V. B. (1989-12-07). "Method for preparation of barium carbide labelled by carbon 14" (in ru). https://inis.iaea.org/Search/search.aspx?orig_q=RN:22022002.
- ↑ Arrol, W. J.; Glascock, R. (1948). "The conversion of carbon dioxide into acetylene on the scale of 2–20 micromoles". Journal of the Chemical Society: 1534–1537. doi:10.1039/JR9480001534. PMID 18101450.
- ↑ "Barium Carbide, BaC2". http://barium.atomistry.com/barium_carbide.html.
- ↑ "Carbide". https://www.infoplease.com/encyclopedia/science/chemistry/elements/carbide.
- ↑ "Barium Carbide, BaC2". http://barium.atomistry.com/barium_carbide.html.
 |
