Chemistry:Oxone
Oxone is the triple salt 2KHSO
5 · KHSO
4 · K
2SO
4. For almost all applications, the active ingredient in this compound is potassium peroxymonosulfate, KHSO
5.[1] The triple salt has a longer shelf-life than potassium peroxymonosulfate, but releases the same peroxymonosulfate anion upon dissolution.
One advantage of oxone from an industrial point of view is that it's dangerous goods classification tends to be Corrosive (Class 8) rather than Oxidising (Class 5). This makes it easier and cheaper to transport compared to other persulfate salts.
Synthesis and structure
The triple salt is produced from peroxysulfuric acid,[citation needed] which is generated in situ by combining fuming sulfuric acid (oleum) and hydrogen peroxide.[citation needed] Careful neutralization of this solution with potassium hydroxide allows the crystallization of the triple salt.[citation needed] X-ray crystallography confirms the triple salt formulation, revealing hydrogen-bonding network that entraps the persulfate anion. The O-O distance is 1.458(2) Å, as found in H2O2.[2]
The purity of Oxone can be determined by iodometric titration. Heavy metal salts catalyze the decomposition of the title compound, based on reporting on its triple salt formulation.[1] An estimated 43-45% of it, by weight, of which 5.2% active oxygen is theoretically possible, and 4.7% was typically observed.[3] In 2012, a review was reporting the KHSO
5 estimate to be "about 50% per mole" of triple salt.[1]) The stability advantage notwithstanding (see following), methods were developed to deliver a forms of the title compound that required smaller amounts in reactions, and this was achieved on large scale in 2002 via preparations of purified KHSO
5·H
2O.[1][4][needs update]
Uses
Underlying the uses of Oxone is the highly positive oxidation potential for peroxymonosulfate, which is +1.81 V.
Cleaning
Oxone-type products are used for oxidative processes that result in decomposition of organic contaminants, and therefore in cleaning, whitening, and disinfection.[5] For instance, it can be used to whiten materials used in dental health practices, to clean materials in the manufacture of microelectronics, and decontaminate recreational water pools.[6][5][7][8][9] Use of formulations containing the title compound in pool water quality management can interfere with determinations of chlorination assay, using a standard ferrous ammonium sulfate, N,N′-diethyl-p-phenylenediamine (FAS-DPD) method, if added reagents and steps are not followed to neutralise the KMPS (potassium monopersulfate / peroxymonosulfate).[10][11]
Preparative chemistry
Oxone is a versatile oxidant in organic chemistry.[12][1][13] It oxidizes terminal alkenes to epoxides. It converts internal alkenes into two equivalents of carboxylic acid. Oxone convert aldehydes to carboxylic acids. When such reactions are conducted in the presence of alcoholic solvents, the corresponding esters may be obtained.[14]
Oxone converts ketones to dioxiranes, which can be used for diverse oxidations in organic synthesis.[15] and in the oxidation of other unsaturated functionalities, heteroatoms, and even some alkane C-H bonds.[16]
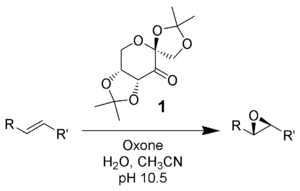
Oxone is used in the production of some organic periodinanes, notably the oxidation of 2-iodobenzoic acid to 2-iodoxybenzoic acid (IBX).[17][non-primary source needed]

Peroxymonosulfate-driven conversions can be used with sulfides and selenides to prepare sulfones and selenones, with anilines and amino sugars to provide nitro compounds, oximes to provide nitro compounds (in aqueous buffered conditions) or to return the parent carbonyl compounds (in the presence of alumina, with microwave heating), primary and secondary amines to provide hydroxylamines (using adsorbed Oxone) or N-nitrosation products (in the presence of sodium nitrite), pyridines and tertiary amines to provide amine oxides, and phosphorus(III) compounds to provide phosphono-compounds largely retaining configuration at phosphorus (with comparable outcomes when a sulfur or selenium atom replaces the phosphorus(III) lone pair).[1]
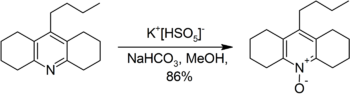
Examples of preparative scale oxidatives of these types are the conversion of an acridine derivative to the corresponding acridine-N-oxide,[18] and the synthesis of fluoromethyl phenyl sulfone, a reagent used in the synthesis of fluoroalkenes.[19]
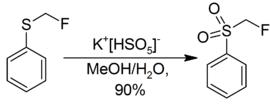
Further reading
- Wu, Mingsong; Xu, Xinyang; Xu, Xun (November 2014). "Algicidal and Bactericidal Effect of Potassium Monopersulfate Compound on Eutrophic Water". Applied Mechanics and Materials 707: 259. doi:10.4028/www.scientific.net/AMM.707.259. https://www.scientific.net/AMM.707.259. Retrieved 3 November 2025.[non-primary source needed]
- Crandall, Jack K.; Shi, Yian; Burke, Christopher P.; Buckley, Benjamin R. (14 September 2012). Paquette, Leo A.. ed (in en). Encyclopedia of Reagents for Organic Synthesis. New York, NY: Wiley & Sons. doi:10.1002/047084289x.rp246.pub3. ISBN 9780470842898. https://onlinelibrary.wiley.com/doi/abs/10.1002/047084289X.rp246.pub3. Retrieved 3 November 2025. The original 1995 print publication of this chapter by Crandall was vol. 3, on pages 295ff.
- DuPont Staff (2008). "DuPont™ Oxone® Monopersulfate Compound / General Technical Attributes". du Pont de Nemours. Wilmington, DE: E.I. du Pont de Nemours & Co.. https://atamankimya.com/Assets/Documents/OXONE_Ataman_Kimya_20201212_185848.pdf.
- Jakob, Harald; Leininger, Stefan; Lehmann, Thomas; Jacobi, Sylvia & Gutewort, Sven (15 July 2007). "Ullmann's Encyclopedia of Industrial Chemistry". in Ley, Claudia. Ullmann's Encyclopedia of Industrial Chemistry. Weinheim, Germany: Wiley-VCH. doi:10.1002/14356007.a19_177.pub2. ISBN 978-3-527-30673-2. https://onlinelibrary.wiley.com/doi/10.1002/14356007.a19_177.pub2. Retrieved 3 November 2025.
- Mundy, B.P.; Ellerd, M.G. & Favaloro Jr., F.G. (2005). "Oxone®". Name Reactions and Reagents in Organic Synthesis (2nd ed.). Hoboken, NJ: John Wiley & Sons. p. 828. ISBN 9780471739869. https://books.google.com/books?id=ahRrPTfeqPEC. Retrieved 3 November 2025.
- Page, P.C.B.; Barros, D.; Buckley, B.R.; Ardakani, A. & Marples, B.A. (2004). "Organocatalysis of Asymmetric Epoxidation Mediated by Iminium Salts under Nonaqueous Conditions". J. Org. Chem. 69 (10): 3595–3597. doi:10.1021/jo035820j. PMID 15132582. https://pubs.acs.org/doi/pdf/10.1021/jo035820j. Retrieved 3 November 2025. Presents an organic-soluble form of Oxone®, tetraphenylphosphonium peroxymonosulfate (Ph4PHSO5), and its successful deployment in the asymmetric epoxidation using peroxymonosulfate-generated oxaziridinium salts.
- Travis, B. R.; Ciaramitaro, B. P. & Borhan, B. (30 September 2002). "Preparation of Purified KHSO5·H2O and nBu4NHSO5 from Oxone by Simple and Efficient Methods". Eur. J. Org. Chem.: 3429–3434. doi:10.1002/1099-0690(200210)2002:20<3429::AID-EJOC3429>3.0.CO;2-D. https://chemistry-europe.onlinelibrary.wiley.com/doi/abs/10.1002/1099-0690%28200210%292002%3A20%3C3429%3A%3AAID-EJOC3429%3E3.0.CO%3B2-D. Retrieved 3 November 2025. The article has been made available by an author, at this link.
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Crandall, Jack K.; Shi, Yian; Burke, Christopher P.; Buckley, Benjamin R. (14 September 2012). Paquette, Leo A.. ed (in en). Encyclopedia of Reagents for Organic Synthesis. New York, NY: Wiley & Sons. doi:10.1002/047084289x.rp246.pub3. ISBN 9780470842898. https://onlinelibrary.wiley.com/doi/abs/10.1002/047084289X.rp246.pub3. Retrieved 3 November 2025..
- ↑ Ermer, Otto; Röbke, Christof (2003). "Crystal Structure and Chemical Stabilization of the Triple Salt (KHSO5)2⋅KHSO4⋅K2SO4". Helvetica Chimica Acta 86 (8): 2908–2913. doi:10.1002/hlca.200390238.
- ↑ DuPont Staff (2008). "DuPont™ Oxone® Monopersulfate Compound / General Technical Attributes". du Pont de Nemours. Wilmington, DE: E.I. du Pont de Nemours & Co.. https://atamankimya.com/Assets/Documents/OXONE_Ataman_Kimya_20201212_185848.pdf.
- ↑ Travis, B. R.; Ciaramitaro, B. P. & Borhan, B. (30 September 2002). "Preparation of Purified KHSO5·H2O and nBu4NHSO5 from Oxone by Simple and Efficient Methods". Eur. J. Org. Chem.: 3429–3434. doi:10.1002/1099-0690(200210)2002:20<3429::AID-EJOC3429>3.0.CO;2-D. https://chemistry-europe.onlinelibrary.wiley.com/doi/abs/10.1002/1099-0690%28200210%292002%3A20%3C3429%3A%3AAID-EJOC3429%3E3.0.CO%3B2-D. Retrieved 3 November 2025. The article has been made available by an author, at this link.
- ↑ 5.0 5.1 Laxness Staff (2025). "Products and Brands / Brands / Oxone™". Laxness.com. Cologne, Germany: LanXess AG. https://lanxess.com/en/products-and-brands/brands/oxone. "Oxone™ powder contains the active component potassium peroxymonosulfate (KHSO5), also known as potassium monopersulfate. The oxidizing power of Oxone™ is derived from this peracid chemistry, making it one of the strongest oxidants available on the market. / Oxone™ is used worldwide in a variety of consumer and industrial applications. / Applications / Pool and spa / Water treatment / Pulp and paper / Home care / Electronics / Denture cleaners / Odor control / Disinfection."
- ↑ Jakob, Harald; Leininger, Stefan; Lehmann, Thomas; Jacobi, Sylvia & Gutewort, Sven (15 July 2007). "Ullmann's Encyclopedia of Industrial Chemistry". in Ley, Claudia. Ullmann's Encyclopedia of Industrial Chemistry. Weinheim, Germany: Wiley-VCH. doi:10.1002/14356007.a19_177.pub2. ISBN 978-3-527-30673-2. https://onlinelibrary.wiley.com/doi/10.1002/14356007.a19_177.pub2. Retrieved 3 November 2025.
- ↑ Peroxy Compounds Human Health and Ecological Draft Risk Assessment DP 455445, 455446 (Report). United States Environmental Protection Agency. 2020-03-11. p. 9-10. https://www.regulations.gov/search?filter=EPA-HQ-OPP-2017-0354-0006. Retrieved 2021-09-24.
- ↑ Wacławek, Stanisław; Lutze, Holger V.; Grübel, Klaudiusz; Padil, Vinod V.T.; Černík, Miroslav; Dionysiou, Dionysios. D. (2017-12-15). "Peroxy Compounds Human Health and Ecological Draft Risk Assessment DP 455445, 455446". Chemical Engineering Journal 330: 44–62. doi:10.1016/j.cej.2017.07.132.
- ↑ PHTA Recreational Water Quality Committee (June 11, 2019). "Tech Notes: Potassium Monopersulfate". Aqua Magazine (AquaMagazine.com). Madison, WI: AB Media Inc.. https://www.aquamagazine.com/service/article/15121932/tech-notes-potassium-monopersulfate. "Potassium Monopersulfate (monopersulfate, KMPS or MPS) is a white, granular, free-flowing peroxygen that provides powerful non-chlorine oxidation for a wide variety of uses. It is the active ingredient in most nonchlorine oxidizers used for pool and spa/hot tub oxidation. / Most non-chlorine oxidizers contain 45% of the active ingredient potassium monopersulfate, but blended compositions are also commercially available..."
- ↑ TFP Staff (Anon.) (November 30, 2018). "ABC's of Pool Water Chemistry". Pool School. Unknown location: Trouble Free Pool (TFP). https://books.google.com/books?id=WWBODwAAQBAJ&pg=PT4. Retrieved 3 November 2025. "Potassium monopersulfate (a common non-chlorine shock) will show up on [ferrous ammonium sulfate, N,N′-diethyl-p-phenylenediamine,] FAS-DPD chlorine tests as [combined chlorine,] CC." See also the same content at the organisation's website, linked here.
- ↑ For a literature starting point for the FAS-DPD test, see Moberg, Ludvig & Karlberg, Bo (2000). "An Improved N,N′-diethyl-p-phenylenediamine (DPD) Method for the Determination of Free Chlorine Based on Multiple Wavelength Detection". Analytica Chimica Acta 407 (1–2): 127–133. doi:10.1016/S0003-2670(99)00780-1. ISSN 0003-2670. Bibcode: 2000AcAC..407..127M. https://www.sciencedirect.com/science/article/pii/S0003267099007801. Retrieved 3 November 2025.[non-primary source needed]
- ↑ Hussain, Hidayat; Green, Ivan R.; Ahmed, Ishtiaq (2013). "Journey Describing Applications of Oxone in Synthetic Chemistry". Chemical Reviews 113 (5): 3329–3371. doi:10.1021/cr3004373. PMID 23451713. Bibcode: 2013ChRv..113.3329H.Hussain, Hidayat; Green, Ivan R.; Ahmed, Ishtiaq (2018). "Addition and Correction to Journey Describing Applications of Oxone in Synthetic Chemistry". Chemical Reviews 118 (7): 4114–4115. doi:10.1021/acs.chemrev.8b00117. PMID 29521500.
- ↑ Mundy, B.P.; Ellerd, M.G. & Favaloro Jr., F.G. (2005). "Oxone®". Name Reactions and Reagents in Organic Synthesis (2nd ed.). Hoboken, NJ: John Wiley & Sons. p. 828. ISBN 9780471739869. https://books.google.com/books?id=ahRrPTfeqPEC. Retrieved 3 November 2025.
- ↑ Smith, Michael B.; March, Jerry (2007), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (6th ed.), New York: Wiley-Interscience, p. 1769, ISBN 978-0-471-72091-1, https://books.google.com/books?id=JDR-nZpojeEC&printsec=frontcover
- ↑ Adam, W.; Saha-Moller, C.; Zhao, C.-G. (2004). "Dioxirane Epoxidation of Alkenes". Org. React. 61: 219. doi:10.1002/0471264180.or061.02. ISBN 978-0-471-26418-7.
- ↑ Adam, W.; Zhao, C.-G.; Jakka, K. (2007). "Dioxirane Oxidations of Compounds other than Alkenes". Org. Reactions 69: 1. doi:10.1002/0471264180.or069.01. ISBN 978-0-471-26418-7.
- ↑ Frigerio, M.; Santagostino, M.; Sputore, S. (1999). "A User-Friendly Entry to 2-Iodoxybenzoic Acid (IBX)". J. Org. Chem. 64 (12): 4537–4538. doi:10.1021/jo9824596. Bibcode: 1999JOrgC..64.4537F.[non-primary source needed]
- ↑ Bell, Thomas W.; Cho, Young-Moon; Firestone, Albert; Healy, Karin; Liu, Jia; Ludwig, Richard; Rothenberger, Scott D. (1990). "9-n-Butyl-1,2,3,4,5,6,7,8-Octahydroacridin-4-ol". Organic Syntheses 69: 226. doi:10.15227/orgsyn.069.0226.
- ↑ McCarthy, James R.; Matthews, Donald P.; P. Paolini, John (1995). "Reaction of Sulfoxides with Diethylaminosulfur Trifluoride". Organic Syntheses 72: 209. doi:10.15227/orgsyn.072.0209.
 |
