Chemistry:Prajmaline
 | |
| Clinical data | |
|---|---|
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
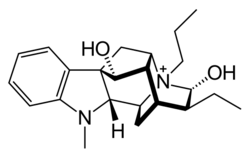
| Formula | C23H33N2O2+ |
| Molar mass | 369.529 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Prajmaline (Neo-gilurythmal)[1] is a class Ia antiarrhythmic agent[2] which has been available since the 1970s.[3] Class Ia drugs increase the time one action potential lasts in the heart.[4] Prajmaline is a semi-synthetic propyl derivative of ajmaline, with a higher bioavailability than its predecessor.[5] It acts to stop arrhythmias of the heart through a frequency-dependent block of cardiac sodium channels.[2]
Mechanism
Prajmaline causes a resting block in the heart.[6] A resting block is the depression of a person's Vmax after a resting period. This effect is seen more in the atrium than the ventricle.[6] The effects of some Class I antiarrhythmics are only seen in a patient who has a normal heart rate (~1 Hz).[7] This is due to the effect of a phenomenon called reverse use dependence.[7] The higher the heart rate, the less effect Prajmaline will have.
Uses
The drug Prajmaline has been used to treat a number of cardiac disorders. These include: coronary artery disease,[8][9] angina,[8][9] paroxysmal tachycardia and Wolff–Parkinson–White syndrome.[1] Prajmaline has been indicated in the treatment of certain disorders where other antiarrhythmic drugs were not effective.[1]
Administration
Prajmaline can be administered orally,[9] parenterally[8] or intravenously.[8] Three days after the last dose, a limited effect has been observed. Therefore, it has been suggested that treatment of arrhythmias with Prajmaline must be continuous to see acceptable results.[1]
Pharmacokinetics
The main metabolites of Prajmaline are: 21-carboxyprajmaline and hydroxyprajmaline. Twenty percent of the drug is excreted in the urine unchanged.
Daily therapeutic dose is 40–80 mg. Distribution half-life is 10 minutes. Plasma protein binding is 60%. Oral bioavailability is 80%. Elimination half-life is 6 hours. Volume of distribution is 4-5 L/kg. [3]
Side Effects
There are no significant adverse side-effects of Prajmaline when taken alone and with a proper dosage.[1][8][9] Patients who are taking other treatments for their symptoms (e.g. beta blockers and nifedipine) have developed minor transient conduction defects when given Prajmaline.[8]
Overdose
An overdose of Prajmaline is possible. The range of symptoms seen during a Prajmaline overdose include: no symptoms, nausea/vomiting, bradycardia, tachycardia, hypotension, and death.[3]
Other Potential Uses
Due to Prajmaline's sodium channel-blocking properties, it has been shown to protect rat white matter from anoxia (82 +/- 15%).[10][11] The concentration used causes little suppression of the preanoxic response.[10][11]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 "[Antiarrhythmic effects of prajmaline (Neo-Gilurythmal) in stable angina pectoris in light of Holter electrocardiographic monitoring]" (in Polish). Przegląd Lekarski 52 (10): 485–491. 1995. PMID 8834838.
- ↑ 2.0 2.1 "Differential analysis of the frequency-dependent effects of class 1 antiarrhythmic drugs according to periodical ligand binding: implications for antiarrhythmic and proarrhythmic efficacy". Journal of Cardiovascular Pharmacology 15 (6): 998–1009. June 1990. doi:10.1097/00005344-199006000-00019. PMID 1694924.
- ↑ 3.0 3.1 3.2 "Clinical course and outcome in class IC antiarrhythmic overdose". Clinical Toxicology 28 (4): 433–44. 1990. doi:10.3109/15563659009038586. PMID 2176700.
- ↑ "Class 1 antiarrhythmic drugs--characteristic electrocardiographic differences when assessed by atrial and ventricular pacing". European Heart Journal 5 (2): 99–107. February 1984. doi:10.1093/oxfordjournals.eurheartj.a061633. PMID 6723689.
- ↑ "The structure of the ring-opened N beta-propyl-ajmaline (Neo-Gilurytmal) at physiological pH is obviously responsible for its better absorption and bioavailability when compared with ajmaline (Gilurytmal)". Die Pharmazie 55 (7): 531–2. July 2000. PMID 10944783.
- ↑ 6.0 6.1 "Comparative analysis of the action of class I antiarrhythmic drugs (lidocaine, quinidine, and prajmaline) in rabbit atrial and ventricular myocardium". Journal of Cardiovascular Pharmacology 15 (2): 338–45. February 1990. doi:10.1097/00005344-199002000-00023. PMID 1689432.
- ↑ 7.0 7.1 "Reverse use dependence of antiarrhythmic class Ia, Ib, and Ic: effects of drugs on the action potential duration?". Pacing and Clinical Electrophysiology 15 (11 Pt 2): 2097–102. November 1992. doi:10.1111/j.1540-8159.1992.tb03028.x. PMID 1279606.
- ↑ 8.0 8.1 8.2 8.3 8.4 8.5 "Acute haemodynamic effects of ajmaline and prajmaline in patients with coronary heart disease". European Journal of Clinical Pharmacology 26 (2): 147–50. 1984. doi:10.1007/bf00630278. PMID 6723753.
- ↑ 9.0 9.1 9.2 9.3 "Effects of oral prajmaline bitartrate on exercise test responses in patients with coronary artery disease". European Journal of Clinical Pharmacology 28 (4): 371–4. 1985. doi:10.1007/bf00544352. PMID 4029242.
- ↑ 10.0 10.1 Stys PK (May 1995). "Protective effects of antiarrhythmic agents against anoxic injury in CNS white matter". Journal of Cerebral Blood Flow and Metabolism 15 (3): 425–32. doi:10.1038/jcbfm.1995.53. PMID 7714000.
- ↑ 11.0 11.1 "Differential effects of Na-K-ATPase pump inhibition, chemical anoxia, and glycolytic blockade on membrane potential of rat optic nerve". Brain Research 1037 (1–2): 171–9. March 2005. doi:10.1016/j.brainres.2005.01.003. PMID 15777766.
 |
