Chemistry:Rubidium nitrate
| |
 Unit cell of rubidium nitrate
| |
| Names | |
|---|---|
| IUPAC name
Rubidium nitrate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| |
| |
| Properties | |
| RbNO3 | |
| Molar mass | 147.473 g/mol |
| Appearance | White hygroscopic solid |
| Density | 3.11 g/cm3 |
| Melting point | 310 °C (590 °F; 583 K) decomposes |
| Boiling point | 578 °C (1,072 °F; 851 K) |
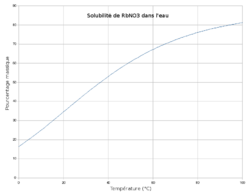
| 44.28 g/100 mL (16 °C) 65.0 g/100 mL (25 °C)[1] | |
| Solubility in acetone | very slightly soluble |
| −41.0·10−6 cm3/mol | |
Refractive index (nD)
|
1.524 |
| Structure[2] | |
| trigonal | |
| P31 | |
a = 10.474 Å, c = 7.443 Å
| |
Lattice volume (V)
|
707.2 Å3 |
| Hazards | |
| Main hazards | Oxidant |
| GHS pictograms |  
|
| GHS Signal word | Danger |
| HH272Script error: No such module "Preview warning".Category:GHS errors, HH315Script error: No such module "Preview warning".Category:GHS errors, HH319Script error: No such module "Preview warning".Category:GHS errors, HH335Script error: No such module "Preview warning".Category:GHS errors | |
| PP210Script error: No such module "Preview warning".Category:GHS errors, PP220Script error: No such module "Preview warning".Category:GHS errors, PP261Script error: No such module "Preview warning".Category:GHS errors, PP264Script error: No such module "Preview warning".Category:GHS errors, PP264+P265Script error: No such module "Preview warning".Category:GHS errors, PP271Script error: No such module "Preview warning".Category:GHS errors, PP280Script error: No such module "Preview warning".Category:GHS errors, PP302+P352Script error: No such module "Preview warning".Category:GHS errors, PP304+P340Script error: No such module "Preview warning".Category:GHS errors, PP305+P351+P338Script error: No such module "Preview warning".Category:GHS errors, PP319Script error: No such module "Preview warning".Category:GHS errors, PP321Script error: No such module "Preview warning".Category:GHS errors, PP332+P317Script error: No such module "Preview warning".Category:GHS errors, PP337+P317Script error: No such module "Preview warning".Category:GHS errors, PP362+P364Script error: No such module "Preview warning".Category:GHS errors, PP370+P378Script error: No such module "Preview warning".Category:GHS errors, PP403+P233Script error: No such module "Preview warning".Category:GHS errors, PP405Script error: No such module "Preview warning".Category:GHS errors, PP501Script error: No such module "Preview warning".Category:GHS errors | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
4625 mg/kg (rat, oral) |
| Related compounds | |
Other anions
|
Rubidium sulfate Rubidium chloride |
Other cations
|
Lithium nitrate Sodium nitrate Potassium nitrate Caesium nitrate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Rubidium nitrate is an inorganic compound with the formula RbNO3. This alkali metal nitrate salt is a white hygroscopic solid that is highly soluble in water.
Properties
In a flame test, RbNO3 gives a mauve/light purple colour.
Production
RbNO3 can be prepared either by dissolving rubidium metal, its hydroxide or carbonate in nitric acid.
- RbOH + HNO
3 → RbNO
3 + H
2O - Rb
2CO
3 + 2 HNO
3 → 2 RbNO
3 + CO
2 + H
2O - 2 Rb + 2 HNO
3 → 2 RbNO
3 + H
2
Uses
Rubidium compounds have very few applications.[1] Like caesium nitrate, it is used in infrared radiation optics, in pyrotechnic compositions as a pyrotechnic colorant and as an oxidizer, e.g. in decoys and illumination flares although it is rarely used in fireworks to produce a red-violet colour. It is also used as a raw material for preparation of other rubidium compounds and rubidium metal, for manufacture of catalysts and in scintillation counters.
References
- ↑ 1.0 1.1 W. Lenk, H. Prinz, A. Steinmetz,"Rubidium and Rubidium Compounds" in Ullmann's Encyclopedia of Industrial Chemistry, 2010 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. doi:10.1002/14356007.a23_473.pub2
- ↑ Jutta Pohl, Dieter Pohl, Gunadi Adiwidjaja (1992). "Phase Transition in Rubidium Nitrate at 346 K and Structure at 296, 372, 413 and 437 K". Acta Crystallographica Section B B48 (2): 160–166. doi:10.1107/S0108768191013459.
- ↑ PubChem. "Rubidium nitrate" (in en). https://pubchem.ncbi.nlm.nih.gov/compound/25731.
| HNO3 | He | ||||||||||||||||
| LiNO3 | Be(NO3)2 | B(NO3)−4 | C | NO−3, NH4NO3 |
O | FNO3 | Ne | ||||||||||
| NaNO3 | Mg(NO3)2 | Al(NO3)3 | Si | P | S | ClONO2 | Ar | ||||||||||
| KNO3 | Ca(NO3)2 | Sc(NO3)3 | Ti(NO3)4 | VO(NO3)3 | Cr(NO3)3 | Mn(NO3)2 | Fe(NO3)3, Fe(NO3)2 |
Co(NO3)2, Co(NO3)3 |
Ni(NO3)2 | Cu(NO3)2 | Zn(NO3)2 | Ga(NO3)3 | Ge | As | Se | Br | Kr |
| RbNO3 | Sr(NO3)2 | Y(NO3)3 | Zr(NO3)4 | Nb | Mo | Tc | Ru | Rh | Pd(NO3)2 | AgNO3 | Cd(NO3)2 | In | Sn | Sb(NO3)3 | Te | I | Xe(NO3)2 |
| CsNO3 | Ba(NO3)2 | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg2(NO3)2, Hg(NO3)2 |
Tl(NO3)3, TlNO3 |
Pb(NO3)2 | Bi(NO3)3 BiO(NO3) |
Po | At | Rn | |
| FrNO3 | Ra(NO3)2 | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |
| ↓ | |||||||||||||||||
| La(NO3)3 | Ce(NO3)3, Ce(NO3)4 |
Pr | Nd(NO3)3 | Pm | Sm | Eu(NO3)3 | Gd(NO3)3 | Tb(NO3)3 | Dy | Ho | Er | Tm | Yb | Lu | |||
| Ac(NO3)3 | Th(NO3)4 | Pa | UO2(NO3)2 | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | |||
 |

