Chemistry:Sodium cyanide
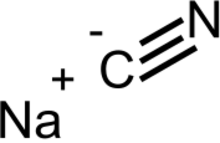
| |
| |

| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1689 |
| |
| |
| Properties | |
| NaCN | |
| Molar mass | 49.0072 g/mol |
| Appearance | white solid |
| Odor | faint bitter almond-like |
| Density | 1.5955 g/cm3 |
| Melting point | 563.7 °C (1,046.7 °F; 836.9 K) |
| Boiling point | 1,496 °C (2,725 °F; 1,769 K) |
| 48.15 g/100 mL (10 °C) 63.7 g/100 mL (25 °C) | |
| Solubility | soluble in ammonia, methanol, ethanol very slightly soluble in dimethylformamide, SO2 insoluble in dimethyl sulfoxide |
Refractive index (nD)
|
1.452 |
| Thermochemistry[1] | |
Heat capacity (C)
|
70.4 J·mol−1·K−1 |
Std molar
entropy (S |
115.6 J·mol−1·K−1 |
Std enthalpy of
formation (ΔfH⦵298) |
−87.5 kJ·mol−1 |
Gibbs free energy (ΔfG˚)
|
−76.4 kJ·mol−1 |
Enthalpy of fusion (ΔfH⦵fus)
|
8.79 kJ·mol−1 |
| Hazards | |
| Safety data sheet | ICSC 1118 |
| GHS pictograms |  
|
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
4 mg/kg (sheep, oral) 15 mg/kg (mammal, oral) 8 mg/kg (rat, oral)[3] |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 5 mg/m3[2] |
REL (Recommended)
|
C 5 mg/m3 (4.7 ppm) [10-minute][2] |
IDLH (Immediate danger)
|
25 mg/m3 (as CN)[2] |
| Related compounds | |
Other cations
|
Potassium cyanide |
Related compounds
|
Hydrogen cyanide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Sodium cyanide is a compound with the formula NaCN and the structure Na+
−
C≡N. It is a white, water-soluble solid. Cyanide has a high affinity for metals, which leads to the high toxicity of this salt. Its main application, in gold mining, also exploits its high reactivity toward metals. It is a moderately strong base.
Production and chemical properties
Sodium cyanide is produced by treating hydrogen cyanide with sodium hydroxide:[4]
- HCN + NaOH → NaCN + H
2O
Worldwide production was estimated at 500,000 tons in the year 2006. Formerly it was prepared by the Castner process involving the reaction of sodium amide with carbon at elevated temperatures.
- NaNH
2 + C → NaCN + H
2
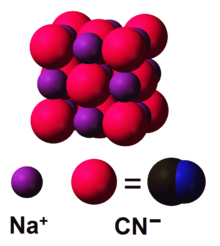
The structure of solid NaCN is related to that of sodium chloride.[5] The anions and cations are each six-coordinate. Potassium cyanide (KCN) adopts a similar structure.[6]
When treated with acid, it forms the toxic gas hydrogen cyanide:
- NaCN + H+
→ HCN + Na+
Because the salt is derived from a weak acid, sodium cyanide readily reverts to HCN by hydrolysis; the moist solid emits small amounts of hydrogen cyanide, which is thought to smell like bitter almonds (not everyone can smell it—the ability thereof is due to a genetic trait[7]). Sodium cyanide reacts rapidly with strong acids to release hydrogen cyanide. This dangerous process represents a significant risk associated with cyanide salts. It is detoxified most efficiently with hydrogen peroxide (H
2O
2) to produce sodium cyanate (NaOCN) and water:[4]
- NaCN + H
2O
2 → NaOCN + H
2O
Applications
Cyanide mining
Gold cyanidation (also known as the cyanide process) is the dominant technique for extracting gold, much of which is obtained from low-grade ore. More than 70% of cyanide consumption globally is used for this purpose. The application exploits the high affinity of gold(I) for cyanide, which induces gold metal to oxidize and dissolve in the presence of air (oxygen) and water, producing the salt sodium dicyanoaurate (or sodium gold cyanide) (NaAu(CN)
2):[4]
- 4 Au + 8 NaCN + O
2 + 2 H
2O → 4 Na[Au(CN)
2] + 4 NaOH
A similar process uses potassium cyanide (KCN, a close relative of sodium cyanide) to produce potassium dicyanoaurate (KAu(CN)
2).
These soluble forms of gold (and silver) can be converted back to the metals by electrolysis.
Chemical feedstock
Some commercially significant chemicals are derived from cyanide: cyanuric chloride, cyanogen chloride, and cyanogen.[4]
Several specialty nitriles may be produced by alkylation of cyanide, a strong nucleophile. Illustrative is the synthesis of benzyl cyanide by the reaction of benzyl chloride and sodium cyanide:[8]
- C
6H
5CH
2Cl + NaCN → C
6H
5CH
2CN + NaCl
Electroplating
Cyanide baths (solutions) of metal ions are used to electroplate many metals, including platinum, gold, and silver. Cadmium plating gives excellent corrosion resistance and is achieved using cadmium oxide and sodium cyanide.[9] Copper 'strikes' (thin high quality layers) are traditionally formed using cyanide electroplating, these thin interstitial layers allow adhesion between metals that are usually incompatible, such as chromium and aluminium.[citation needed]
Niche uses
Being highly toxic, sodium cyanide is used to kill or stun rapidly such as in collecting jars used by entomologists and in widely illegal cyanide fishing.
It was used as an insecticide, rodenticide and antibacterial, but these uses were cancelled by the EPA in 1987.[10]
Toxicity
Sodium cyanide, like other soluble cyanide salts, is among the most rapidly acting of all known poisons. NaCN is a potent inhibitor of cellular respiration, acting on mitochondrial cytochrome oxidase and hence blocking electron transport. This results in decreased oxidative metabolism and oxygen utilization. Lactic acidosis then occurs as a consequence of anaerobic metabolism. An oral dosage as small as 200–300 mg can be fatal.
Disposal
Due to toxicity considerations, the disposal of cyanide is subject to stringent regulations. Industrial cyanide effluent is typically destroyed by oxidation using peroxysulfuric acid, hydrogen peroxide, sulfur dioxide/copper salts ("Inco process") or all three ("Combiox Process"). Use of sodium hypochlorite, traditional for laboratory-scale wastes, is impractical on a commercial scale. Hydrolysis at higher temperatures is highly effective, but requires specialized equipment. Lastly, cyanide wastes can be acidified for recovery of hydrogen cyanide.[4]
References
- ↑ CRC handbook of chemistry and physics : a ready-reference book of chemical and physical data.. William M. Haynes, David R. Lide, Thomas J. Bruno (2016-2017, 97th ed.). Boca Raton, Florida. 2016. ISBN 978-1-4987-5428-6. OCLC 930681942.
- ↑ 2.0 2.1 2.2 NIOSH Pocket Guide to Chemical Hazards. "#0562". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0562.html.
- ↑ "Cyanides (as CN)". Immediately Dangerous to Life and Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/idlh/cyanides.html.
- ↑ 4.0 4.1 4.2 4.3 4.4 Gail, Ernst; Gos, Stephen; Kulzer, Rupprecht; Lorösch, Jürgen; Rubo, Andreas; Sauer, Manfred; Kellens, Raf; Reddy, Jay et al. (2011). "Cyano Compounds, Inorganic". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a08_159.pub3. ISBN 978-3-527-30385-4.
- ↑ Wells, A.F. (1984) Structural Inorganic Chemistry, Oxford: Clarendon Press. ISBN 0-19-855370-6.
- ↑ H. T. Stokes; D. L. Decker; H. M. Nelson; J. D. Jorgensen (1993). "Structure of potassium cyanide at low temperature and high pressure determined by neutron diffraction". Phys. Rev. B 47 (17): 11082–11092. doi:10.1103/PhysRevB.47.11082. PMID 10005242. Bibcode: 1993PhRvB..4711082S. https://zenodo.org/record/1233725.
- ↑ Online Mendelian Inheritance in Man (OMIM) 304300
- ↑ Adams, Roger; Thal, A. F. (1922). "Benzyl cyanide". Organic Syntheses 2: 9. doi:10.15227/orgsyn.002.0009.
- ↑ Schulte-Schrepping, Karl-Heinz; Piscator, Magnus (2000). "Cadmium and Cadmium Compounds". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a04_499. ISBN 978-3-527-30385-4.
- ↑ "Reregistration Eligibility Decision (R.E.D. Facts) Sodium cyanide". September 1994. https://www3.epa.gov/pesticides/chem_search/reg_actions/reregistration/fs_PC-074002_1-Sep-94.pdf.
External links
- Institut national de recherche et de sécurité (INRS), "Cyanure de sodium. Cyanure de potassium", Fiche toxicologique n° 111, Paris, 2006, 6 pp. (PDF file, in French)
- International Chemical Safety Card 1118
- Hydrogen cyanide and cyanides (CICAD 61)
- National Pollutant Inventory - Cyanide compounds fact sheet
- NIOSH Pocket Guide to Chemical Hazards
- EINECS number 205-599-4
- CID {{{1}}} from PubChem
- CSST (Canada)
- Sodium cyanide hazards to fish and other wildlife from gold
 |


