Astronomy:Neutronium
Neutronium (sometimes shortened to neutrium,[1] also referred to as neutrite[2]) is a hypothetical substance composed purely of neutrons. The word was coined by scientist Andreas von Antropoff in 1926 (before the 1932 discovery of the neutron) for the hypothetical "element of atomic number zero" (with zero protons in its nucleus) that he placed at the head of the periodic table (denoted by -).[3][4] However, the meaning of the term has changed over time, and from the last half of the 20th century onward it has been also used to refer to extremely dense substances resembling the neutron-degenerate matter theorized to exist in the cores of neutron stars; hereinafter "degenerate neutronium" will refer to this.
In neutron stars
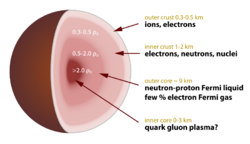
Neutronium is used in popular physics literature[1][2] to refer to the material present in the cores of neutron stars (stars which are too massive to be supported by electron degeneracy pressure and which collapse into a denser phase of matter). In scientific literature the term "neutron-degenerate matter"[5] or simply neutron matter is used for this material.[6]
Hypothetical multi-neutrons
The term "neutronium" was coined in 1926 by Andreas von Antropoff for a conjectured form of matter made up of neutrons with no protons or electrons, which he placed as the chemical element of atomic number zero at the head of his new version of the periodic table.[3] It was subsequently placed in the middle of several spiral representations of the periodic system for classifying the chemical elements, such as those of Charles Janet (1928), Edgar Emerson (1944),[7][8] and John D. Clark (1950).
The term is not used in the scientific literature either for a condensed form of matter, or as an element, and theoretical analysis expects no bound forms of neutrons without protons.[9] If neutronium were considered to be an element, then these neutron clusters could be considered to be the isotopes of that element. However, these reports have not been further substantiated.
- Neutron: An isolated neutron undergoes beta decay with a mean lifetime of approximately 15 minutes (half-life of approximately 10 minutes), becoming a proton (the nucleus of hydrogen), an electron, and an antineutrino.
- Dineutron: The dineutron, containing two neutrons, is not a stable bound particle, but had been proposed as an extremely short-lived resonance state produced by nuclear reactions involving tritium. The resonance was unambiguously observed in 2012 in the decay of beryllium-16.[10][11] It has been suggested to have a transitory existence in nuclear reactions produced by helions (completely ionized helium-3 nuclei) that result in the formation of a proton and a nucleus having the same atomic number as the target nucleus but a mass number two units greater. The dineutron hypothesis had been used in nuclear reactions with exotic nuclei for a long time.[12] Several applications of the dineutron in nuclear reactions can be found in review papers.[13] Its existence has been proven to be relevant for nuclear structure of exotic nuclei.[14] A system made up of only two neutrons is not bound, though the attraction between them is very nearly enough to make them so.[15] This has some consequences on nucleosynthesis and the abundance of the chemical elements.[13][16]
- Trineutron: A trineutron state consisting of three bound neutrons has not been detected, and is not expected to exist[citation needed] even for a short time.
- Tetraneutron: A tetraneutron is a hypothetical particle consisting of four bound neutrons. Reports of its existence have not been replicated.[17][18]
- Pentaneutron: Calculations indicate that the hypothetical pentaneutron state, consisting of a cluster of five neutrons, would not be bound.[19]
See also
- Compact star
- Hydrogen – in its most common form, has a nucleus with only a proton
References
- ↑ 1.0 1.1 Inglis-Arkell, Esther (2012-04-14). "Neutrium: The Most Neutral Hypothetical State of Matter Ever". io9.com. http://io9.com/5899961/neutrium-the-most-neutral-hypothetical-state-of-matter-ever.
- ↑ 2.0 2.1 Zhuravleva, Valentina (2005). Ballad of the Stars: Stories of Science Fiction, Ultraimagination, and TRIZ. Technical Innovation Center, Inc.. p. 75. ISBN 978-0-9640740-6-4. https://books.google.com/books?id=HpttCzNiB6wC&pg=PA75. Retrieved 2019-04-25.
- ↑ 3.0 3.1 von Antropoff, A. (1926). "Eine neue Form des periodischen Systems der Elementen". Zeitschrift für Angewandte Chemie 39 (23): 722–725. doi:10.1002/ange.19260392303. Bibcode: 1926AngCh..39..722V.
- ↑ Stewart, P. J. (2007). "A century on from Dmitrii Mendeleev: Tables and spirals, noble gases and Nobel prizes". Foundations of Chemistry 9 (3): 235–245. doi:10.1007/s10698-007-9038-x.
- ↑ Angelo, J. A. (2006). Encyclopedia of Space and Astronomy. Infobase Publishing. p. 178. ISBN 978-0-8160-5330-8. https://books.google.com/books?id=VUWno1sOwnUC&pg=PA178. Retrieved 2016-10-28.
- ↑ Gandolfi, Stefano; Gezerlis, Alexandros; Carlson, J. (2015-10-19). "Neutron Matter from Low to High Density" (in en). Annual Review of Nuclear and Particle Science 65 (1): 303–328. doi:10.1146/annurev-nucl-102014-021957. ISSN 0163-8998. https://www.annualreviews.org/doi/10.1146/annurev-nucl-102014-021957.
- ↑ Emerson, Edgar I. (1944). "A new spiral form of the periodic table". Journal of Chemical Education 21 (3): 111. doi:10.1021/ed021p111. Bibcode: 1944JChEd..21..111E.
- ↑ Emerson, Edgar I. (1944). "A chart based on atomic numbers showing the electronic structure of the elements". Journal of Chemical Education 21 (5): 254. doi:10.1021/ed021p254. Bibcode: 1944JChEd..21..254E.
- ↑ Timofeyuk, N. K. (2003). "Do multineutrons exist?". Journal of Physics G 29 (2): L9. doi:10.1088/0954-3899/29/2/102. Bibcode: 2003JPhG...29L...9T.
- ↑ Schirber, M. (2012). "Nuclei Emit Paired-up Neutrons". Physics 5: 30. doi:10.1103/Physics.5.30. Bibcode: 2012PhyOJ...5...30S.
- ↑ Spyrou, A.; Kohley, Z.; Baumann, T.; Bazin, D. et al. (2012). "First Observation of Ground State Dineutron Decay: 16Be". Physical Review Letters 108 (10): 102501. doi:10.1103/PhysRevLett.108.102501. PMID 22463404. Bibcode: 2012PhRvL.108j2501S.
- ↑ Bertulani, C. A.; Baur, G. (1986). "Coincidence Cross-sections for the Dissociation of Light Ions in High-energy Collisions". Nuclear Physics A 480 (3–4): 615–628. doi:10.1016/0375-9474(88)90467-8. Bibcode: 1988NuPhA.480..615B. http://faculty.tamu-commerce.edu/cbertulani/cab/papers/NPA480_1988_615.pdf.
- ↑ 13.0 13.1 Bertulani, C. A.; Canto, L. F.; Hussein, M. S. (1993). "The Structure And Reactions Of Neutron-Rich Nuclei". Physics Reports 226 (6): 281–376. doi:10.1016/0370-1573(93)90128-Z. Bibcode: 1993PhR...226..281B. http://www.tamu-commerce.edu/physics/carlos/papers/PRep226_1993_281.pdf.
- ↑ Hagino, K.; Sagawa, H.; Nakamura, T.; Shimoura, S. (2009). "Two-particle correlations in continuum dipole transitions in Borromean nuclei". Physical Review C 80 (3): 1301. doi:10.1103/PhysRevC.80.031301. Bibcode: 2009PhRvC..80c1301H.
- ↑ MacDonald, J.; Mullan, D. J. (2009). "Big Bang Nucleosynthesis: The Strong Nuclear Force meets the Weak Anthropic Principle". Physical Review D 80 (4): 3507. doi:10.1103/PhysRevD.80.043507. Bibcode: 2009PhRvD..80d3507M.
- ↑ Kneller, J. P.; McLaughlin, G. C. (2004). "The Effect of Bound Dineutrons upon BBN". Physical Review D 70 (4): 3512. doi:10.1103/PhysRevD.70.043512. Bibcode: 2004PhRvD..70d3512K.
- ↑ Bertulani, C. A.; Zelevinsky, V. (2003). "Is the tetraneutron a bound dineutron-dineutron molecule?". Journal of Physics G 29 (10): 2431–2437. doi:10.1088/0954-3899/29/10/309. Bibcode: 2003JPhG...29.2431B.
- ↑ "Tetra-Neutron Experiment: Understanding of Nuclear Forces Might Have To Be Significantly Changed". . SciTechDaily, December 12, 2021. Technical University of Munich (TUM)
- ↑ Bevelacqua, J. J. (1981). "Particle stability of the pentaneutron". Physics Letters B 102 (2–3): 79–80. doi:10.1016/0370-2693(81)91033-9. Bibcode: 1981PhLB..102...79B.
 |
