Chemistry:Samarium
Samarium is a chemical element; it has symbol Sm and atomic number 62. It is a moderately hard silvery metal that slowly oxidizes in air. Being a typical member of the lanthanide series, samarium usually has the oxidation state +3. Compounds of samarium(II) are also known, most notably the monoxide SmO, monochalcogenides SmS, SmSe and SmTe, as well as samarium(II) iodide.
Discovered in 1879 by French chemist Paul-Émile Lecoq de Boisbaudran, samarium was named after the mineral samarskite from which it was isolated. The mineral itself was named after a Russian mine official, Colonel Vassili Samarsky-Bykhovets, who thus became the first person to have a chemical element named after him, though the name was indirect.
Samarium occurs in concentration up to 2.8% in several minerals including cerite, gadolinite, samarskite, monazite and bastnäsite, the last two being the most common commercial sources of the element. These minerals are mostly found in China, the United States, Brazil, India, Sri Lanka and Australia; China is by far the world leader in samarium mining and production.
The main commercial use of samarium is in samarium–cobalt magnets, which have permanent magnetization second only to neodymium magnets; however, samarium compounds can withstand significantly higher temperatures, above 700 °C (1,292 °F), without losing their permanent magnetic properties. The radioisotope samarium-153 is the active component of the drug samarium (153Sm) lexidronam (Quadramet), which kills cancer cells in lung cancer, prostate cancer, breast cancer and osteosarcoma. Another isotope, samarium-149, is a strong neutron absorber and so is added to control rods of nuclear reactors. It also forms as a decay product during reactor operation and is one of the important factors considered in reactor design and operation. Other uses of samarium include catalysis of chemical reactions, radioactive dating and X-ray lasers. Samarium(II) iodide, in particular, is a common reducing agent in chemical synthesis.
Samarium has no biological role; some samarium salts are slightly toxic.[1]
Physical properties
Samarium is a rare earth element with a hardness and density similar to zinc. With a boiling point of 1,794 °C (3,261 °F), samarium is the third most volatile lanthanide after ytterbium and europium and comparable in this respect to lead and barium; this helps separation of samarium from its ores.[2][3] When freshly prepared, samarium has a silvery lustre, and takes on a duller appearance when oxidized in air. Samarium is calculated to have one of the largest atomic radii of the elements; with a radius of 238 pm, only potassium, praseodymium, barium, rubidium and caesium are larger.[4]
In ambient conditions, samarium has a rhombohedral structure (α form). Upon heating to 731 °C (1,348 °F), its crystal symmetry changes to hexagonal close-packed (hcp),; it has actual transition temperature depending on metal purity. Further heating to 922 °C (1,692 °F) transforms the metal into a body-centered cubic (bcc) phase. Heating to 300 °C (572 °F) plus compression to 40 kbar results in a double-hexagonally close-packed structure (dhcp). Higher pressure of the order of hundreds or thousands of kilobars induces a series of phase transformations, in particular with a tetragonal phase appearing at about 900 kbar.[5] In one study, the dhcp phase could be produced without compression, using a nonequilibrium annealing regime with a rapid temperature change between about 400 °C (752 °F) and 700 °C (1,292 °F), confirming the transient character of this samarium phase. Thin films of samarium obtained by vapor deposition may contain the hcp or dhcp phases in ambient conditions.[5]
Samarium and its sesquioxide are paramagnetic at room temperature. Their corresponding effective magnetic moments, below 2 bohr magnetons, are the third-lowest among lanthanides (and their oxides) after lanthanum and lutetium. The metal transforms to an antiferromagnetic state upon cooling to 14.8 K.[6][7] Individual samarium atoms can be isolated by encapsulating them into fullerene molecules.[8] They can also be intercalated into the interstices of the bulk C60 to form a solid solution of nominal composition Sm3C60, which is superconductive at a temperature of 8 K.[9] Samarium doping of iron-based superconductors – a class of high-temperature superconductor – increases their transition to normal conductivity temperature up to 56 K, the highest value achieved so far in this series.[10]
Chemical properties
In air, samarium slowly oxidizes at room temperature and spontaneously ignites at 150 °C (302 °F).[1][3] Even when stored under mineral oil, samarium gradually oxidizes and develops a grayish-yellow powder of the oxide-hydroxide mixture at the surface. The metallic appearance of a sample can be preserved by sealing it under an inert gas such as argon.
Samarium is quite electropositive and reacts slowly with cold water and rapidly with hot water to form samarium hydroxide:[11]
- 2Sm
(s) + 6H
2O
(l) → 2Sm(OH)
3
(aq) + 3H
2
(g)
Samarium dissolves readily in dilute sulfuric acid to form solutions containing the yellow[12] to pale green Sm(III) ions, which exist as [Sm(OH
2)
9]3+ complexes:[11]
- 2Sm
(s) + 3H
2SO
4
(aq) → 2Sm3+
(aq) + 3SO2−
4
(aq) + 3H
2
(g)
Samarium is one of the few lanthanides with a relatively accessible +2 oxidation state, alongside Eu and Yb.[13] Sm2+ ions are blood-red in aqueous solution.[14]
Compounds
| Formula | Color | Symmetry | Space group | No. | Pearson symbol | a (pm) | b (pm) | c (pm) | Z | Density, g/cm3 |
|---|---|---|---|---|---|---|---|---|---|---|
| Sm | silvery | trigonal[5] | R3m | 166 | hR9 | 362.9 | 362.9 | 2621.3 | 9 | 7.52 |
| Sm | silvery | hexagonal[5] | P63/mmc | 194 | hP4 | 362 | 362 | 1168 | 4 | 7.54 |
| Sm | silvery | tetragonal[15] | I4/mmm | 139 | tI2 | 240.2 | 240.2 | 423.1 | 2 | 20.46 |
| SmO | golden | cubic[16] | Fm3m | 225 | cF8 | 494.3 | 494.3 | 494.3 | 4 | 9.15 |
| Sm2O3 | trigonal[17] | P3m1 | 164 | hP5 | 377.8 | 377.8 | 594 | 1 | 7.89 | |
| Sm2O3 | monoclinic[17] | C2/m | 12 | mS30 | 1418 | 362.4 | 885.5 | 6 | 7.76 | |
| Sm2O3 | cubic[18] | Ia3 | 206 | cI80 | 1093 | 1093 | 1093 | 16 | 7.1 | |
| SmH2 | cubic[19] | Fm3m | 225 | cF12 | 537.73 | 537.73 | 537.73 | 4 | 6.51 | |
| SmH3 | hexagonal[20] | P3c1 | 165 | hP24 | 377.1 | 377.1 | 667.2 | 6 | ||
| Sm2B5 | gray | monoclinic[21] | P21/c | 14 | mP28 | 717.9 | 718 | 720.5 | 4 | 6.49 |
| SmB2 | hexagonal[22] | P6/mmm | 191 | hP3 | 331 | 331 | 401.9 | 1 | 7.49 | |
| SmB4 | tetragonal[23] | P4/mbm | 127 | tP20 | 717.9 | 717.9 | 406.7 | 4 | 6.14 | |
| SmB6 | cubic[24] | Pm3m | 221 | cP7 | 413.4 | 413.4 | 413.4 | 1 | 5.06 | |
| SmB66 | cubic[25] | Fm3c | 226 | cF1936 | 2348.7 | 2348.7 | 2348.7 | 24 | 2.66 | |
| Sm2C3 | cubic[26] | I43d | 220 | cI40 | 839.89 | 839.89 | 839.89 | 8 | 7.55 | |
| SmC2 | tetragonal[26] | I4/mmm | 139 | tI6 | 377 | 377 | 633.1 | 2 | 6.44 | |
| SmF2 | purple[27] | cubic[28] | Fm3m | 225 | cF12 | 587.1 | 587.1 | 587.1 | 4 | 6.18 |
| SmF3 | white[27] | orthorhombic[28] | Pnma | 62 | oP16 | 667.22 | 705.85 | 440.43 | 4 | 6.64 |
| SmCl2 | brown[27] | orthorhombic[29] | Pnma | 62 | oP12 | 756.28 | 450.77 | 901.09 | 4 | 4.79 |
| SmCl3 | yellow[27] | hexagonal[28] | P63/m | 176 | hP8 | 737.33 | 737.33 | 416.84 | 2 | 4.35 |
| SmBr2 | brown[27] | orthorhombic[30] | Pnma | 62 | oP12 | 797.7 | 475.4 | 950.6 | 4 | 5.72 |
| SmBr3 | yellow[27] | orthorhombic[31] | Cmcm | 63 | oS16 | 404 | 1265 | 908 | 2 | 5.58 |
| SmI2 | green[27] | monoclinic | P21/c | 14 | mP12 | |||||
| SmI3 | orange[27] | trigonal[32] | R3 | 63 | hR24 | 749 | 749 | 2080 | 6 | 5.24 |
| SmN | cubic[33] | Fm3m | 225 | cF8 | 357 | 357 | 357 | 4 | 8.48 | |
| SmP | cubic[34] | Fm3m | 225 | cF8 | 576 | 576 | 576 | 4 | 6.3 | |
| SmAs | cubic[35] | Fm3m | 225 | cF8 | 591.5 | 591.5 | 591.5 | 4 | 7.23 |
Oxides
The most stable oxide of samarium is the sesquioxide Sm2O3. Like many samarium compounds, it exists in several crystalline phases. The trigonal form is obtained by slow cooling from the melt. The melting point of Sm2O3 is high (2345 °C), so it is usually melted not by direct heating, but with induction heating, through a radio-frequency coil. Sm2O3 crystals of monoclinic symmetry can be grown by the flame fusion method (Verneuil process) from Sm2O3 powder, that yields cylindrical boules up to several centimeters long and about one centimeter in diameter. The boules are transparent when pure and defect-free and are orange otherwise. Heating the metastable trigonal Sm2O3 to 1,900 °C (3,450 °F) converts it to the more stable monoclinic phase.[17] Cubic Sm2O3 has also been described.[18]
Samarium is one of the few lanthanides that form a monoxide, SmO. This lustrous golden-yellow compound was obtained by reducing Sm2O3 with samarium metal at high temperature (1000 °C) and a pressure above 50 kbar; lowering the pressure resulted in incomplete reaction. SmO has cubic rock-salt lattice structure.[16][36]
Chalcogenides
Samarium forms a trivalent sulfide, selenide and telluride. Divalent chalcogenides SmS, SmSe and SmTe with a cubic rock-salt crystal structure are known. These chalcogenides convert from a semiconducting to metallic state at room temperature upon application of pressure.[37] Whereas the transition is continuous and occurs at about 20–30 kbar in SmSe and SmTe, it is abrupt in SmS and requires only 6.5 kbar. This effect results in a spectacular color change in SmS from black to golden yellow when its crystals of films are scratched or polished. The transition does not change the lattice symmetry, but there is a sharp decrease (~15%) in the crystal volume.[38] It exhibits hysteresis, i.e., when the pressure is released, SmS returns to the semiconducting state at a much lower pressure of about 0.4 kbar.[1][39]
Halides

Samarium metal reacts with all the halogens, forming trihalides:[40]
- 2 Sm (s) + 3 X2 (g) → 2 SmX3 (s) (X = F, Cl, Br or I)
Their further reduction with samarium, lithium or sodium metals at elevated temperatures (about 700–900 °C) yields the dihalides.[29] The diiodide can also be prepared by heating SmI3, or by reacting the metal with 1,2-diiodoethane in anhydrous tetrahydrofuran at room temperature:[41]
- Sm (s) + ICH2-CH2I → SmI2 + CH2=CH2.
In addition to dihalides, the reduction also produces many non-stoichiometric samarium halides with a well-defined crystal structure, such as Sm3F7, Sm14F33, Sm27F64,[28] Sm11Br24, Sm5Br11 and Sm6Br13.[42]
Samarium halides change their crystal structures when one type of halide anion is substituted for another, which is an uncommon behavior for most elements (e.g. actinides). Many halides have two major crystal phases for one composition, one being significantly more stable and another being metastable. The latter is formed upon compression or heating, followed by quenching to ambient conditions. For example, compressing the usual monoclinic samarium diiodide and releasing the pressure results in a PbCl2-type orthorhombic structure (density 5.90 g/cm3),[43] and similar treatment results in a new phase of samarium triiodide (density 5.97 g/cm3).[44]
Borides
Sintering powders of samarium oxide and boron, in a vacuum, yields a powder containing several samarium boride phases; the ratio between these phases can be controlled through the mixing proportion.[45] The powder can be converted into larger crystals of samarium borides using arc melting or zone melting techniques, relying on the different melting/crystallization temperature of SmB6 (2580 °C), SmB4 (about 2300 °C) and SmB66 (2150 °C). All these materials are hard, brittle, dark-gray solids with the hardness increasing with the boron content.[24] Samarium diboride is too volatile to be produced with these methods and requires high pressure (about 65 kbar) and low temperatures between 1140 and 1240 °C to stabilize its growth. Increasing the temperature results in the preferential formation of SmB6.[22]
Samarium hexaboride
Samarium hexaboride is a typical intermediate-valence compound where samarium is present both as Sm2+ and Sm3+ ions in a 3:7 ratio.[45] It belongs to a class of Kondo insulators; at temperatures above 50 K, its properties are typical of a Kondo metal, with metallic electrical conductivity characterized by strong electron scattering, whereas at lower temperatures, it behaves as a non-magnetic insulator with a narrow band gap of about 4–14 meV.[46] The cooling-induced metal-insulator transition in SmB6 is accompanied by a sharp increase in the thermal conductivity, peaking at about 15 K. The reason for this increase is that electrons themselves do not contribute to the thermal conductivity at low temperatures, which is dominated by phonons, but the decrease in electron concentration reduces the rate of electron-phonon scattering.[47]
Other inorganic compounds

Samarium carbides are prepared by melting a graphite-metal mixture in an inert atmosphere. After the synthesis, they are unstable in air and need to be studied under an inert atmosphere.[26] Samarium monophosphide SmP is a semiconductor with a bandgap of 1.10 eV, the same as in silicon, and electrical conductivity of n-type. It can be prepared by annealing at 1,100 °C (2,010 °F) an evacuated quartz ampoule containing mixed powders of phosphorus and samarium. Phosphorus is highly volatile at high temperatures and may explode, thus the heating rate has to be kept well below 1 °C/min.[34] A similar procedure is adopted for the monarsenide SmAs, but the synthesis temperature is higher at 1,800 °C (3,270 °F).[35]
Numerous crystalline binary compounds are known for samarium and one of the group 14, 15, or 16 elements X, where X is Si, Ge, Sn, Pb, Sb or Te, and metallic alloys of samarium form another large group. They are all prepared by annealing mixed powders of the corresponding elements. Many of the resulting compounds are non-stoichiometric and have nominal compositions SmaXb, where the b/a ratio varies between 0.5 and 3.[48][49]
Organometallic compounds
Samarium forms a cyclopentadienide Sm(C
5H
5)
3 and its chloroderivatives Sm(C
5H
5)
2Cl and Sm(C
5H
5)Cl
2. They are prepared by reacting samarium trichloride with NaC
5H
5 in tetrahydrofuran. Contrary to cyclopentadienides of most other lanthanides, in Sm(C
5H
5)
3 some C
5H
5 rings bridge each other by forming ring vertexes η1 or edges η2 toward another neighboring samarium, thus creating polymeric chains.[14] The chloroderivative Sm(C
5H
5)
2Cl has a dimer structure, which is more accurately expressed as (η(
5)–C
5H
5)
2Sm(–Cl)
2(η(
5)–C
5H
5)
2. There, the chlorine bridges can be replaced, for instance, by iodine, hydrogen or nitrogen atoms or by CN groups.[50]
The (C
5H
5)− ion in samarium cyclopentadienides can be replaced by the indenide (C
9H
7)− or cyclooctatetraenide (C
8H
8)2− ring, resulting in Sm(C
9H
7)
3 or KSm(η(
8)–C
8H
8)
2. The latter compound has a structure similar to uranocene. There is also a cyclopentadienide of divalent samarium, Sm(C
5H
5)
2 a solid that sublimates at about 85 °C (185 °F). Contrary to ferrocene, the C
5H
5 rings in Sm(C
5H
5)
2 are not parallel but are tilted by 40°.[50][51]
A metathesis reaction in tetrahydrofuran or ether gives alkyls and aryls of samarium:[50]
- SmCl
3 + 3LiR → SmR
3 + 3LiCl - Sm(OR)
3 + 3LiCH(SiMe
3)
2 → Sm{CH(SiMe
3)
2}
3 + 3LiOR
Here R is a hydrocarbon group and Me = methyl.
Isotopes
Naturally occurring samarium is composed of five stable isotopes: 144Sm, 149Sm, 150Sm, 152Sm and 154Sm, and two extremely long-lived radioisotopes, 147Sm (half-life t1/2 = 1.066×1011 years) and 148Sm (6.3×1015 years), with 152Sm being the most abundant (26.75%).[52] 149Sm is listed by various sources as being stable,[52][53] but some sources state that it is radioactive;[54] the lower bound on its half-life is 2×1015 years.[52] Some observationally stable samarium isotopes are predicted to decay to isotopes of neodymium.[55] The long-lived isotopes 146Sm, 147Sm, and 148Sm (and 149Sm if it decays) undergo alpha decay to neodymium isotopes. Lighter unstable isotopes of samarium mainly decay by electron capture to promethium, while heavier ones beta decay to europium.[52] The known isotopes range from 129Sm to 168Sm.[56] The half-lives of 151Sm and 145Sm are 94.6 years and 340 days, respectively. All remaining radioisotopes have half-lives that are less than 2 days, and most these have half-life less than 48 seconds.[52] Natural samarium has a radioactivity of 127 Bq/g, mostly due to 147Sm,[57] whose alpha decay to 143Nd is used in samarium–neodymium dating.[58][59] 146Sm is an extinct radionuclide, with the half-life of 9.20×107 years.[60] There have been searches of samarium-146 as a primordial nuclide, because its half-life is long enough such that minute quantities of the element should persist today.[61] It can be used in radiometric dating.[62]
Samarium-149 is an observationally stable isotope of samarium and a product of the decay chain from the fission product 149Nd (yield 1.0888%). 149Sm is a significant neutron absorber in nuclear reactors, with a neutron poison effect that is second in importance for reactor design and operation only to 135Xe.[63][64] Its neutron cross section is 41000 barns for thermal neutrons.[65] Because samarium-149 is not radioactive and is not removed by decay, it presents problems somewhat different from those encountered with xenon-135. The equilibrium concentration (and thus the poisoning effect) builds to an equilibrium value during reactor operations in about 500 hours (about three weeks) as the isotope does not measurably vanish except through neutron capture.[66]
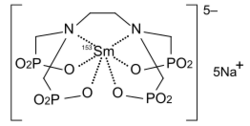
Samarium-153 is a beta emitter with a half-life of 46.285 hours. It is used to kill cancer cells in lung cancer, prostate cancer, breast cancer, and osteosarcoma. For this purpose, samarium-153 is chelated with ethylene diamine tetramethylene phosphonate (EDTMP) and injected intravenously. The chelation prevents accumulation of radioactive samarium in the body that would result in excessive irradiation and generation of new cancer cells.[1] The corresponding drug has several names including samarium (153Sm) lexidronam; its trade name is Quadramet.[67][68][69]
History

Detection of samarium and related elements was announced by several scientists in the second half of the 19th century; however, most sources give priority to French chemist Paul-Émile Lecoq de Boisbaudran.[70][71] Boisbaudran isolated samarium oxide and/or hydroxide in Paris in 1879 from the mineral samarskite ((Y,Ce,U,Fe)
3(Nb,Ta,Ti)
5O
16) and identified a new element in it via sharp optical absorption lines.[3] Swiss chemist Marc Delafontaine announced a new element decipium (from Latin: decipiens meaning "deceptive, misleading") in 1878,[72][73] but later in 1880–1881 demonstrated that it was a mix of several elements, one being identical to Boisbaudran's samarium.[74][75] Though samarskite was first found in the Ural Mountains in Russia, by the late 1870s it had been found in other places, making it available to many researchers. In particular, it was found that the samarium isolated by Boisbaudran was also impure and had a comparable amount of europium. The pure samarium(III) oxide was produced only in 1901 by Eugène-Anatole Demarçay,[76][77][78] and in 1903 Wilhelm Muthmann isolated the element.
Boisbaudran named his element samarium after the mineral samarskite, which in turn honored Vassili Samarsky-Bykhovets (1803–1870). Samarsky-Bykhovets, as the Chief of Staff of the Russian Corps of Mining Engineers, had granted access for two German mineralogists, the brothers Gustav and Heinrich Rose, to study the mineral samples from the Urals.[79][80][81] Samarium was thus the first chemical element to be named after a person.[76][82] The word samaria is sometimes used to mean samarium(III) oxide, by analogy with yttria, zirconia, alumina, ceria, holmia, etc. The symbol Sm was suggested for samarium, but an alternative Sa was often used instead until the 1920s.[76][83]
Before the advent of ion-exchange separation technology in the 1950s, pure samarium had no commercial uses. However, a by-product of fractional crystallization purification of neodymium was a mix of samarium and gadolinium that got the name "Lindsay Mix" after the company that made it, and was used for nuclear control rods in some early nuclear reactors.[84] Nowadays, a similar commodity product has the name "samarium-europium-gadolinium" (SEG) concentrate.[82] It is prepared by solvent extraction from the mixed lanthanides isolated from bastnäsite (or monazite). Since heavier lanthanides have more affinity for the solvent used, they are easily extracted from the bulk using relatively small proportions of solvent. Not all rare-earth producers who process bastnäsite do so on a large enough scale to continue by separating the components of SEG, which typically makes up only 1–2% of the original ore. Such producers therefore make SEG with a view to marketing it to the specialized processors. In this manner, the valuable europium in the ore is rescued for use in making phosphor. Samarium purification follows the removal of the europium. As of 2012[update], being in oversupply, samarium oxide is cheaper on a commercial scale than its relative abundance in the ore might suggest.[85]
Occurrence and production

Samarium concentration in soils varies between 2 and 23 ppm, and oceans contain about 0.5–0.8 parts per trillion.[1] The median value for its abundance in the Earth's crust used by the CRC Handbook is 7 parts per million (ppm)[86] and is the 40th most abundant element.[87] Distribution of samarium in soils strongly depends on its chemical state and is very inhomogeneous: in sandy soils, samarium concentration is about 200 times higher at the surface of soil particles than in the water trapped between them, and this ratio can exceed 1,000 in clays.[88]
Samarium is not found free in nature, but, like other rare earth elements, is contained in many minerals, including monazite, bastnäsite, cerite, gadolinite and samarskite; monazite (in which samarium occurs at concentrations of up to 2.8%)[3] and bastnäsite are mostly used as commercial sources. World resources of samarium are estimated at two million tonnes; they are mostly located in China, US, Brazil, India, Sri Lanka and Australia, and the annual production is about 700 tonnes.[1] Country production reports are usually given for all rare-earth metals combined. By far, China has the largest production with 120,000 tonnes mined per year; it is followed by the US (about 5,000 tonnes)[88] and India (2,700 tonnes).[89] Samarium is usually sold as oxide, which at the price of about US$30/kg is one of the cheapest lanthanide oxides.[85] Whereas mischmetal – a mixture of rare earth metals containing about 1% of samarium – has long been used, relatively pure samarium has been isolated only recently, through ion exchange processes, solvent extraction techniques, and electrochemical deposition. The metal is often prepared by electrolysis of a molten mixture of samarium(III) chloride with sodium chloride or calcium chloride. Samarium can also be obtained by reducing its oxide with lanthanum. The product is then distilled to separate samarium (boiling point 1,794 °C) and lanthanum (b.p. 3,464 °C).[71]
Very few minerals have samarium being the most dominant element. Minerals with essential (dominant) samarium include monazite-(Sm) and florencite-(Sm). These minerals are very rare and are usually found containing other elements, usually cerium or neodymium.[90][91][92][93] It is also made by neutron capture by samarium-149, which is added to the control rods of nuclear reactors. Therefore, 151Sm is present in spent nuclear fuel and radioactive waste.[88]
Separating samarium from minerals involves nearly 100 individual processes and extremely strong acids.[94]
Geopolitics
Western militaries across the world relied on a single samarium production plant in La Rochelle, France from the 1970s until the plant's closure in 1994.[94] The facility sourced its samarium from Australia.[94] A $1 billion United States government effort to re-open a closed rare earths mine in Mountain Pass, California resulted in the facility going bankrupt.[94]
As of 2025[update], China produces all of the world's usable samarium; refining is concentrated in Baotou.[94] The Biden administration signed two contracts for samarium production plants in the United States, but neither materialized.[94] During US president Donald Trump's second-term tariff war, China leveled strict limits on the export of samarium, among other rare earth metals, as part of the long-running rare earths trade dispute (and larger trade war) between the two nations.[94]
Applications
Magnets
An important use of samarium is samarium–cobalt magnets, which are nominally SmCo
5 or Sm
2Co
17.[95] They have high permanent magnetization, about 10,000 times that of iron and second only to neodymium magnets. However, samarium magnets resist demagnetization better; they are stable to temperatures above 700 °C (1,292 °F) (cf. 300–400 °C for neodymium magnets). These magnets are found in small motors, headphones, and high-end magnetic pickups for guitars and related musical instruments.[1] For example, they are used in the motors of a solar-powered electric aircraft, the Solar Challenger, and in the Samarium Cobalt Noiseless electric guitar and bass pickups.
Due to their heat resistance, samarium magnets are also used for military applications and are needed to manufacture modern aircraft and missiles.[94] A single F-35 fighter jet contains about 50 pounds (23 kg) of samarium magnets.[94]
Chemical reagent

2
Samarium and its compounds are important as catalysts and chemical reagents. Samarium catalysts help the decomposition of plastics, dechlorination of pollutants such as polychlorinated biphenyls (PCB), as well as dehydration and dehydrogenation of ethanol.[3] Samarium(III) triflate Sm(OTf)
3, that is Sm(CF
3SO
3)
3, is one of the most efficient Lewis acid catalysts for a halogen-promoted Friedel–Crafts reaction with alkenes.[96] Samarium(II) iodide is a very common reducing and coupling agent in organic synthesis, for example in desulfonylation reactions; annulation; Danishefsky, Kuwajima, Mukaiyama and Holton Taxol total syntheses; strychnine total synthesis; Barbier reaction and other reductions with samarium(II) iodide.[97]
In its usual oxidized form, samarium is added to ceramics and glasses where it increases absorption of infrared light. As a (minor) part of mischmetal, samarium is found in the "flint" ignition devices of many lighters and torches.[1][3]
Neutron absorber
Samarium-149 has a high cross section for neutron capture (41,000 barns) and so is used in control rods of nuclear reactors. Its advantage compared to competing materials, such as boron and cadmium, is stability of absorption – most of the fusion products of 149Sm are other isotopes of samarium that are also good neutron absorbers. For example, the cross section of samarium-151 is 15,000 barns, it is on the order of hundreds of barns for 150Sm, 152Sm, and 153Sm, and 6,800 barns for natural (mixed-isotope) samarium.[3][88][98]
Lasers
Samarium-doped calcium fluoride crystals were used as an active medium in one of the first solid-state lasers designed and built by Peter Sorokin (co-inventor of the dye laser) and Mirek Stevenson at IBM research labs in early 1961. This samarium laser gave pulses of red light at 708.5 nm. It had to be cooled by liquid helium and so did not find practical applications.[99][100] Another samarium-based laser became the first saturated X-ray laser operating at wavelengths shorter than 10 nanometers. It gave 50-picosecond pulses at 7.3 and 6.8 nm suitable for uses in holography, high-resolution microscopy of biological specimens, deflectometry, interferometry, and radiography of dense plasmas related to confinement fusion and astrophysics. Saturated operation meant that the maximum possible power was extracted from the lasing medium, resulting in the high peak energy of 0.3 mJ. The active medium was samarium plasma produced by irradiating samarium-coated glass with a pulsed infrared Nd-glass laser (wavelength ~1.05 μm).[101]
Storage phosphor
In 2007 it was shown that nanocrystalline BaFCl:Sm3+ as prepared by co-precipitation can serve as a very efficient X-ray storage phosphor.[102] The co-precipitation leads to nanocrystallites of the order of 100–200 nm in size and their sensitivity as X-ray storage phosphors is increased a remarkable ~500,000 times because of the specific arrangements and density of defect centers in comparison with microcrystalline samples prepared by sintering at high temperature.[103] The mechanism is based on reduction of Sm3+ to Sm2+ by trapping electrons that are created upon exposure to ionizing radiation in the BaFCl host. The 5DJ–7FJ f–f luminescence lines can be very efficiently excited via the parity allowed 4f6→4f55d transition at ~417 nm. The latter wavelength is ideal for efficient excitation by blue-violet laser diodes as the transition is electric dipole allowed and thus relatively intense (400 L/(mol⋅cm)).[104] The phosphor has potential applications in personal dosimetry, dosimetry and imaging in radiotherapy, and medical imaging.[105]
Non-commercial and potential uses
- The change in electrical resistivity in samarium monochalcogenides can be used in a pressure sensor or in a memory device triggered between a low-resistance and high-resistance state by external pressure,[106] and such devices are being developed commercially.[107] Samarium monosulfide also generates electric voltage upon moderate heating to about 150 °C (302 °F) that can be applied in thermoelectric power converters.[108]
- Analysis of relative concentrations of samarium and neodymium isotopes 147Sm, 144Nd, and 143Nd allows determination of the age and origin of rocks and meteorites in samarium–neodymium dating. Both elements are lanthanides and are very similar physically and chemically. Thus, Sm–Nd dating is either insensitive to partitioning of the marker elements during various geologic processes, or such partitioning can well be understood and modeled from the ionic radii of said elements.[109]
- The Sm3+ ion is a potential activator for use in warm-white light emitting diodes. It offers high luminous efficacy due to narrow emission bands; but the generally low quantum efficiency and too little absorption in the UV-A to blue spectral region hinders commercial application.[110]
- Samarium is used for ionosphere testing. A rocket spreads samarium monoxide as a red vapor at high altitude, and researchers test how the atmosphere disperses it and how it impacts radio transmissions.[111][112]
- Samarium hexaboride, SmB
6, has recently been shown to be a topological insulator with potential uses in quantum computing.[113][114][115][116]
Biological role and precautions
| Hazards[117] | |
|---|---|
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H261 | |
| PP231 + P232Script error: No such module "Preview warning".Category:GHS errors, PP280Script error: No such module "Preview warning".Category:GHS errors, PP370 + P378Script error: No such module "Preview warning".Category:GHS errors, PP501Script error: No such module "Preview warning".Category:GHS errors | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Samarium salts stimulate metabolism, but it is unclear whether this is from samarium or other lanthanides present with it. The total amount of samarium in adults is about 50 μg, mostly in liver and kidneys and with ~8 μg/L being dissolved in blood. Samarium is not absorbed by plants to a measurable concentration and so is normally not part of human diet. However, a few plants and vegetables may contain up to 1 part per million of samarium. Insoluble salts of samarium are non-toxic and the soluble ones are only slightly toxic.[1][119] When ingested, only 0.05% of samarium salts are absorbed into the bloodstream and the remainder are excreted. From the blood, 45% goes to the liver and 45% is deposited on the surface of the bones where it remains for 10 years; the remaining 10% is excreted.[88]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 Emsley, John (2001). "Samarium". Nature's Building Blocks: An A–Z Guide to the Elements. Oxford, England, UK: Oxford University Press. pp. 371–374. ISBN 0-19-850340-7. https://books.google.com/books?id=j-Xu07p3cKwC&pg=PA371.
- ↑ J.A. Dean, ed (1999). Lange's Handbook of Chemistry (15th ed.). New York, NY: McGraw-Hill. Section 3; Table 3.2 Physical Constants of Inorganic Compounds. ISBN 978-0-07016384-3.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 Hammond, C. R. (2004-06-29). "The Elements". Handbook of Chemistry and Physics (81st ed.). Boca Raton New York Washington: CRC press. pp. 4–27. ISBN 978-0-8493-0481-1. https://archive.org/details/crchandbookofche81lide.
- ↑ Clementi, E.; Raimond, D. L.; Reinhardt, W. P. (1967). "Atomic Screening Constants from SCF Functions. II. Atoms with 37 to 86 Electrons". Journal of Chemical Physics 47 (4): 1300–1307. doi:10.1063/1.1712084. Bibcode: 1967JChPh..47.1300C.
- ↑ 5.0 5.1 5.2 5.3 Shi, N.; Fort, D. (1985). "Preparation of samarium in the double hexagonal close packed form". Journal of the Less Common Metals 113 (2): 21. doi:10.1016/0022-5088(85)90294-2.
- ↑ Lock, J. M. (1957). "The Magnetic Susceptibilities of Lanthanum, Cerium, Praseodymium, Neodymium and Samarium, from 1.5 K to 300 K". Proceedings of the Physical Society. Series B 70 (6): 566. doi:10.1088/0370-1301/70/6/304. Bibcode: 1957PPSB...70..566L.
- ↑ Huray, P.; Nave, S.; Haire, R. (1983). "Magnetism of the heavy 5f elements". Journal of the Less Common Metals 93 (2): 293. doi:10.1016/0022-5088(83)90175-3.
- ↑ Okazaki, T.; Suenaga, Kazutomo; Hirahara, Kaori et al. (2002). "Electronic and geometric structures of metallofullerene peapods". Physica B 323 (1–4): 97. doi:10.1016/S0921-4526(02)00991-2. Bibcode: 2002PhyB..323...97O.
- ↑ Chen, X.; Roth, G. (1995). "Superconductivity at 8 K in samarium-doped C60". Physical Review B 52 (21): 15534–15536. doi:10.1103/PhysRevB.52.15534. PMID 9980911. Bibcode: 1995PhRvB..5215534C.
- ↑ Wu, G.; Xie, Y. L.; Chen, H. et al. (2008). "Superconductivity at 56 K in Samarium-doped SrFeAsF". Journal of Physics: Condensed Matter 21 (14). doi:10.1088/0953-8984/21/14/142203. PMID 21825317. Bibcode: 2009JPCM...21n2203W.
- ↑ 11.0 11.1 "Chemical reactions of Samarium". Webelements. https://www.webelements.com/samarium/chemistry.html.
- ↑ Greenwood, p. 1243
- ↑ Stephen T. Liddle, ed (2022). The lanthanides and actinides: synthesis, reactivity, properties and applications. London. p. 213. ISBN 978-1-80061-015-6. OCLC 1251740566.
- ↑ 14.0 14.1 Greenwood, p. 1248
- ↑ Vohra, Y.; Akella, Jagannadham; Weir, Sam; Smith, Gordon S. (1991). "A new ultra-high pressure phase in samarium". Physics Letters A 158 (1–2): 89. doi:10.1016/0375-9601(91)90346-A. Bibcode: 1991PhLA..158...89V. https://zenodo.org/record/1258493.
- ↑ 16.0 16.1 Leger, J.; Yacoubi, N.; Loriers, J. (1981). "Synthesis of rare earth monoxides". Journal of Solid State Chemistry 36 (3): 261. doi:10.1016/0022-4596(81)90436-9. Bibcode: 1981JSSCh..36..261L.
- ↑ 17.0 17.1 17.2 Gouteron, J.; Michel, D.; Lejus, A. M.; Zarembowitch, J. (1981). "Raman spectra of lanthanide sesquioxide single crystals: Correlation between A and B-type structures". Journal of Solid State Chemistry 38 (3): 288. doi:10.1016/0022-4596(81)90058-X. Bibcode: 1981JSSCh..38..288G.
- ↑ 18.0 18.1 Taylor, D. (1984). "Thermal Expansion Data: III Sesquioxides, M2O3, with the corundum and the A-, B- and C-M2O3 structures". British Ceramic Transactions and Journal 83 (4): 92–98.
- ↑ Daou, J.; Vajda, P.; Burger, J. (1989). "Low temperature thermal expansion in SmH2+x". Solid State Communications 71 (12): 1145. doi:10.1016/0038-1098(89)90728-X. Bibcode: 1989SSCom..71.1145D.
- ↑ Dolukhanyan, S. (1997). "Synthesis of novel compounds by hydrogen combustion". Journal of Alloys and Compounds 253–254: 10. doi:10.1016/S0925-8388(96)03071-X.
- ↑ Zavalii, L. V.; Kuz'ma, Yu. B.; Mikhalenko, S. I. (1990). "Sm2B5 boride and its structure". Soviet Powder Metallurgy and Metal Ceramics 29 (6): 471. doi:10.1007/BF00795346.
- ↑ 22.0 22.1 Cannon, J.; Cannon, D.; Tracyhall, H. (1977). "High pressure syntheses of SmB2 and GdB12". Journal of the Less Common Metals 56: 83. doi:10.1016/0022-5088(77)90221-1.
- ↑ Etourneau, J.; Mercurio, J.; Berrada, A.; Hagenmuller, P.; Georges, R.; Bourezg, R.; Gianduzzo, J. (1979). "The magnetic and electrical properties of some rare earth tetraborides". Journal of the Less Common Metals 67 (2): 531. doi:10.1016/0022-5088(79)90038-9.
- ↑ 24.0 24.1 Solovyev, G. I.; Spear, K. E. (1972). "Phase Behavior in the Sm-B System". Journal of the American Ceramic Society 55 (9): 475. doi:10.1111/j.1151-2916.1972.tb11344.x.
- ↑ Schwetz, K.; Ettmayer, P.; Kieffer, R.; Lipp, A. (1972). "Über die Hektoboridphasen der Lanthaniden und Aktiniden". Journal of the Less Common Metals 26: 99. doi:10.1016/0022-5088(72)90012-4.
- ↑ 26.0 26.1 26.2 Spedding, F. H.; Gschneidner, K.; Daane, A. H. (1958). "The Crystal Structures of Some of the Rare Earth Carbides". Journal of the American Chemical Society 80 (17): 4499. doi:10.1021/ja01550a017. Bibcode: 1958JAChS..80.4499S.
- ↑ 27.0 27.1 27.2 27.3 27.4 27.5 27.6 27.7 Greenwood, p. 1241
- ↑ 28.0 28.1 28.2 28.3 Greis, O. (1978). "Über neue Verbindungen im system SmF2_SmF3". Journal of Solid State Chemistry 24 (2): 227. doi:10.1016/0022-4596(78)90013-0. Bibcode: 1978JSSCh..24..227G.
- ↑ 29.0 29.1 Meyer, G.; Schleid, T. (1986). "The metallothermic reduction of several rare-earth trichlorides with lithium and sodium". Journal of the Less Common Metals 116: 187. doi:10.1016/0022-5088(86)90228-6.
- ↑ Bärnighausen, H. (1973). "Revue für anorganic Chemie". Revue de chimie minérale. 10: 77–92.
- ↑ Zachariasen, W. H. (1948). "Crystal chemical studies of the 5f-series of elements. I. New structure types". Acta Crystallographica 1 (5): 265. doi:10.1107/S0365110X48000703. Bibcode: 1948AcCry...1..265Z.
- ↑ Asprey, L. B.; Keenan, T. K.; Kruse, F. H. (1964). "Preparation and Crystal Data for Lanthanide and Actinide Triiodides". Inorganic Chemistry 3 (8): 1137. doi:10.1021/ic50018a015. https://digital.library.unt.edu/ark:/67531/metadc867868/m2/1/high_res_d/4067674.pdf.
- ↑ Brown, R.; Clark, N. J. (1974). "Composition limits and vaporization behaviour of rare earth nitrides". Journal of Inorganic and Nuclear Chemistry 36 (11): 2507. doi:10.1016/0022-1902(74)80462-8.
- ↑ 34.0 34.1 Meng, J.; Ren, Yufang (1991). "Studies on the electrical properties of rare earth monophosphides". Journal of Solid State Chemistry 95 (2): 346. doi:10.1016/0022-4596(91)90115-X. Bibcode: 1991JSSCh..95..346M.
- ↑ 35.0 35.1 Beeken, R.; Schweitzer, J. (1981). "Intermediate valence in alloys of SmSe with SmAs". Physical Review B 23 (8): 3620. doi:10.1103/PhysRevB.23.3620. Bibcode: 1981PhRvB..23.3620B.
- ↑ Greenwood, p. 1239
- ↑ Bakar, Abu; Afaq, A.; Khan, M. Faizan; ul Aarifeen, Najm; Imran Jamil, M.; Asif, Muhammad (2020-01-01). "Insight into the structural, vibrational and thermodynamic properties of SmX (X = S, Se, Te) chalcogenides: First-principles investigations" (in en). Physica B: Condensed Matter 576. doi:10.1016/j.physb.2019.411715. ISSN 0921-4526. https://www.sciencedirect.com/science/article/pii/S0921452619306209.
- ↑ Beaurepaire, E. (2006). Magnetism: a synchrotron radiation approach. Berlin: Springer. ISBN 978-3-540-33242-8. OCLC 262692720.
- ↑ Jayaraman, A.; Narayanamurti, V.; Bucher, E.; Maines, R. (1970). "Continuous and Discontinuous Semiconductor-Metal Transition in Samarium Monochalcogenides Under Pressure". Physical Review Letters 25 (20): 1430. doi:10.1103/PhysRevLett.25.1430. Bibcode: 1970PhRvL..25.1430J.
- ↑ Greenwood, pp. 1236, 1241
- ↑ Greenwood, p. 1240
- ↑ Baernighausen, H.; Haschke, John M. (1978). "Compositions and crystal structures of the intermediate phases in the samarium-bromine system". Inorganic Chemistry 17: 18. doi:10.1021/ic50179a005.
- ↑ Beck, H. P. (1979). "Hochdruckmodifikationen der Diiodide von Sr., Sm und Eu. Eine neue PbCl2-Variante?". Zeitschrift für anorganische und allgemeine Chemie 459 (1): 81. doi:10.1002/zaac.19794590108. Bibcode: 1979ZAACh.459...81B.
- ↑ Beck, H. P.; Gladrow, E. (1979). "Zur Hochdruckpolymorphie der Seltenerd-Trihalogenide". Zeitschrift für anorganische und allgemeine Chemie 453 (1): 79. doi:10.1002/zaac.19794530610. Bibcode: 1979ZAACh.453...79B.
- ↑ 45.0 45.1 Nickerson, J.; White, R.; Lee, K.; Bachmann, R.; Geballe, T.; Hull, G. (1971). "Physical Properties of SmB6". Physical Review B 3 (6): 2030. doi:10.1103/PhysRevB.3.2030. Bibcode: 1971PhRvB...3.2030N.
- ↑ Nyhus, P.; Cooper, S.; Fisk, Z.; Sarrao, J. (1995). "Light scattering from gap excitations and bound states in SmB6". Physical Review B 52 (20): 14308–14311. doi:10.1103/PhysRevB.52.R14308. PMID 9980746. Bibcode: 1995PhRvB..5214308N.
- ↑ Sera, M.; Kobayashi, S.; Hiroi, M.; Kobayashi, N.; Kunii, S. (1996). "Thermal conductivity of RB6 (R=Ce, Pr, Nd, Sm, Gd) single crystals". Physical Review B 54 (8): R5207–R5210. doi:10.1103/PhysRevB.54.R5207. PMID 9986570. Bibcode: 1996PhRvB..54.5207S.
- ↑ Gladyshevskii, E. I.; Kripyakevich, P. I. (1965). "Monosilicides of rare earth metals and their crystal structures". Journal of Structural Chemistry 5 (6): 789. doi:10.1007/BF00744231. Bibcode: 1965JStCh...5..789G.
- ↑ Smith, G. S.; Tharp, A. G.; Johnson, W. (1967). "Rare earth–germanium and –silicon compounds at 5:4 and 5:3 compositions". Acta Crystallographica 22 (6): 940. doi:10.1107/S0365110X67001902. Bibcode: 1967AcCry..22..940S.
- ↑ 50.0 50.1 50.2 Greenwood, p. 1249
- ↑ Evans, William J.; Hughes, Laura A.; Hanusa, Timothy P. (1986). "Synthesis and x-ray crystal structure of bis(pentamethylcyclopentadienyl) complexes of samarium and europium: (C5Me5)2Sm and (C5Me5)2Eu". Organometallics 5 (7): 1285. doi:10.1021/om00138a001.
- ↑ 52.0 52.1 52.2 52.3 52.4 Kondev, F. G.; Wang, M.; Huang, W. J.; Naimi, S.; Audi, G. (2021). "The NUBASE2020 evaluation of nuclear properties". Chinese Physics C 45 (3). doi:10.1088/1674-1137/abddae. https://www-nds.iaea.org/amdc/ame2020/NUBASE2020.pdf.
- ↑ "Chart of the nuclides". Brookhaven National Laboratory. http://www.nndc.bnl.gov/chart/reCenter.jsp?z=62&n=87.
- ↑ Holden, Norman E. "Table of the isotopes" in Lide, D. R., ed (2005). CRC Handbook of Chemistry and Physics (86th ed.). Boca Raton (FL): CRC Press. ISBN 0-8493-0486-5.
- ↑ Belli, P.; Bernabei, R.; Danevich, F. A.; Incicchitti, A.; Tretyak, V. I. (2019). "Experimental searches for rare alpha and beta decays". European Physical Journal A 55 (140): 4–6. doi:10.1140/epja/i2019-12823-2. Bibcode: 2019EPJA...55..140B.
- ↑ Kiss, G. G.Expression error: Unrecognized word "et". (2022). "Measuring the β-decay properties of neutron-rich exotic Pm, Sm, Eu, and Gd isotopes to constrain the nucleosynthesis yields in the rare-earth region". The Astrophysical Journal 936 (107): 107. doi:10.3847/1538-4357/ac80fc. Bibcode: 2022ApJ...936..107K.
- ↑ Radiation Protection and NORM Residue Management in the Production of Rare Earths from Thorium Containing Minerals (Report). Safety Report Series. International Atomic Energy Agency. 2011. p. 174. https://www-pub.iaea.org/MTCD/Publications/PDF/Pub1512_web.pdf. Retrieved 2022-07-25.
- ↑ Depaolo, D. J.; Wasserburg, G. J. (1976). "Nd isotopic variations and petrogenetic models". Geophysical Research Letters 3 (5): 249. doi:10.1029/GL003i005p00249. Bibcode: 1976GeoRL...3..249D. https://authors.library.caltech.edu/41937/1/grl330.pdf.
- ↑ McCulloch, M. T.; Wasserburg, G. J. (1978). "Sm-Nd and Rb-Sr Chronology of Continental Crust Formation". Science 200 (4345): 1003–11. doi:10.1126/science.200.4345.1003. PMID 17740673. Bibcode: 1978Sci...200.1003M. https://resolver.caltech.edu/CaltechAUTHORS:20131107-143832294.
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedChiera2024 - ↑ Macfarlane, Ronald D. (1960). "Natural Occurrence of Samarium-146". Nature 188 (4757): 1180–1181. doi:10.1038/1881180a0. ISSN 0028-0836. Bibcode: 1960Natur.188.1180M.
- ↑ Samir Maji et al. (2006). "Separation of samarium and neodymium: a prerequisite for getting signals from nuclear synthesis". Analyst 131 (12): 1332–1334. doi:10.1039/b608157f. PMID 17124541. Bibcode: 2006Ana...131.1332M.
- ↑ DOE Fundamentals Handbook: Nuclear Physics and Reactor Theory. U.S. Department of Energy. January 1993. pp. 34, 67. http://www.hss.energy.gov/nuclearsafety/ns/techstds/standard/hdbk1019/h1019v2.pdf.
- ↑ K., Khattab (2005). "Comparison of xenon-135 and samarium-149 poisoning in the Miniature Neutron Source Reactor" (in Arabic). https://inis.iaea.org/search/search.aspx?orig_q=RN:36069288.
- ↑ Espinosa, Carlos E.; Bodmann, Bardo E.J.. "Modeling and simulation of nuclear fuel in scenarios with long time scales" (in English). 19. ENFIR: meeting on nuclear reactor physics and thermal hydraulics. https://inis.iaea.org/search/search.aspx?orig_q=RN:46133777.
- ↑ DOE Handbook, pp. 43–47.
- ↑ "Centerwatch About drug Quadramet". http://www.centerwatch.com/patient/drugs/dru267.html.
- ↑ Pattison, John E. (1999). "Finger doses received during 153Sm injections". Health Physics 77 (5): 530–5. doi:10.1097/00004032-199911000-00006. PMID 10524506. Bibcode: 1999HeaPh..77..530P.
- ↑ Finlay, I. G.; Mason, M. D.; Shelley, M. (2005). "Radioisotopes for the palliation of metastatic bone cancer: a systematic review". The Lancet Oncology 6 (6): 392–400. doi:10.1016/S1470-2045(05)70206-0. PMID 15925817.
- ↑ Greenwood, p. 1229
- ↑ 71.0 71.1 Samarium, Encyclopædia Britannica on-line
- ↑ Delafontaine, Marc (1878). "Sur le décepium, métal nouveau de la samarskite" (in fr). Journal de pharmacie et de chimie 28: 540. https://gallica.bnf.fr/ark:/12148/bpt6k78100m.image.r=Decipium.f548.langEN.
- ↑ Delafontaine, Marc (1878). "Sur le décepium, métal nouveau de la samarskite" (in fr). Comptes rendus hebdomadaires des séances de l'Académie des Sciences 87: 632. https://gallica.bnf.fr/ark:/12148/bpt6k3044x.image.r=Decipium.f694.langEN.
- ↑ Template:CIAAW2003
- ↑ Delafontaine, Marc (1881). "Sur le décipium et le samarium" (in fr). Comptes rendus hebdomadaires des séances de l'Académie des Sciences 93: 63. https://gallica.bnf.fr/ark:/12148/bpt6k3049g.image.r=Decipium.f63.langEN.
- ↑ 76.0 76.1 76.2 Samarium: History & Etymology. Elements.vanderkrogt.net. Retrieved on 2013-03-21.
- ↑ Weeks, Mary Elvira (1956). The discovery of the elements (6th ed.). Easton, PA: Journal of Chemical Education. https://archive.org/details/discoveryoftheel002045mbp.
- ↑ Weeks, Mary Elvira (1932). "The discovery of the elements. XIII. Some elements predicted by Mendeleeff". Journal of Chemical Education 9 (9): 1605–1619. doi:10.1021/ed009p1605. Bibcode: 1932JChEd...9.1605W.
- ↑ Samarskite, Great Soviet Encyclopedia (in Russian)
- ↑ Boisbaudran, Lecoq de (1879). "Recherches sur le samarium, radical d'une terre nouvelle extraite de la samarskite". Comptes rendus hebdomadaires des séances de l'Académie des sciences 89: 212–214. https://gallica.bnf.fr/ark:/12148/bpt6k3046j/f214.pagination.
- ↑ Shipley, Joseph Twadell. The Origins of English Words: A Discursive Dictionary of Indo-European Roots, JHU Press, 2001, p.90. ISBN 0-8018-6784-3
- ↑ 82.0 82.1 "Chemistry in Its Element - Samarium". http://www.rsc.org/chemistryworld/podcast/Interactive_Periodic_Table_Transcripts/Samarium.asp.
- ↑ Coplen, T. B.; Peiser, H. S. (1998). "History of the recommended atomic-weight values from 1882 to 1997: A comparison of differences from current values to the estimated uncertainties of earlier values (Technical Report)". Pure and Applied Chemistry 70: 237. doi:10.1351/pac199870010237. https://zenodo.org/record/1236255.
- ↑ Ghaemi, Arezoo; Tavakkoli, Haman; Rajabi, Negar (2015-06-01). "Solvent influence upon complex formation between 4,13-didecyl-1,7,10,16-tetraoxa-4,13-diazacyclooctadecane with samarium(III) metal cation in binary mixed non-aqueous solvents" (in en). Russian Journal of Applied Chemistry 88 (6): 977–984. doi:10.1134/S1070427215060130. ISSN 1608-3296.
- ↑ 85.0 85.1 What are their prices?, Lynas corp.
- ↑ ABUNDANCE OF ELEMENTS IN THE EARTH'S CRUST AND IN THE SEA, CRC Handbook of Chemistry and Physics, 97th edition (2016–2017), p. 14-17
- ↑ Emsley, John (2011-08-25) (in en). Nature's Building Blocks: An A-Z Guide to the Elements. OUP Oxford. ISBN 978-0-19-960563-7. https://books.google.com/books?id=2EfYXzwPo3UC&dq=%2240th+most+abundant+element%22&pg=PA466.
- ↑ 88.0 88.1 88.2 88.3 88.4 Human Health Fact Sheet on Samarium , Los Alamos National Laboratory
- ↑ "Rare Earths". United States Geological Survey. January 2010. https://minerals.usgs.gov/minerals/pubs/commodity/rare_earths/mcs-2010-raree.pdf.
- ↑ Masau, M.; Cerny, P.; Cooper, M. A.; Chapman, R.; Grice, J. D. (2002-12-01). "MONAZITE-(Sm), A NEW MEMBER OF THE MONAZITE GROUP FROM THE ANNIE CLAIM #3 GRANITIC PEGMATITE, SOUTHEASTERN MANITOBA" (in en). The Canadian Mineralogist 40 (6): 1649–1655. doi:10.2113/gscanmin.40.6.1649. ISSN 0008-4476. Bibcode: 2002CaMin..40.1649M. http://www.canmin.org/cgi/doi/10.2113/gscanmin.40.6.1649.
- ↑ Repina, S. A.; Popova, V. I.; Churin, E. I.; Belogub, E. V.; Khiller, V. V. (December 2011). "Florencite-(Sm)—(Sm,Nd)Al3(PO4)2(OH)6: a new mineral species of the alunite–jarosite group from the subpolar Urals" (in en). Geology of Ore Deposits 53 (7): 564–574. doi:10.1134/S1075701511070191. ISSN 1075-7015. Bibcode: 2011GeoOD..53..564R. https://link.springer.com/10.1134/S1075701511070191.
- ↑ "Monazite-(Sm): Monazite-(Sm) mineral information and data". http://www.mindat.org/min-11438.html.
- ↑ "Florencite-(Sm): Florencite-(Sm) mineral information and data". http://www.mindat.org/min-42495.html.
- ↑ 94.0 94.1 94.2 94.3 94.4 94.5 94.6 94.7 94.8 Bradsher, Keith (2025-06-09). "China's Chokehold on This Obscure Mineral Threatens the West's Militaries". https://www.nytimes.com/2025/06/09/business/china-rare-earth-samarium-fighter-jets.html.
- ↑ Marchio, Cathy (Apr 16, 2024). "Two Grades of Samarium Cobalt Magnets: SmCo5 & Sm2Co17". https://www.stanfordmagnets.com/two-grades-of-samarium-cobalt-magnets-smco5-sm2co17.html.
- ↑ Hajra, S.; Maji, B.; Bar, S. (2007). "Samarium Triflate-Catalyzed Halogen-Promoted Friedel-Crafts Alkylation with Alkenes". Org. Lett. 9 (15): 2783–2786. doi:10.1021/ol070813t. PMID 17585769.
- ↑ Cotton, F. Albert; Wilkinson, Geoffrey; Murillo, Carlos A.; Bochmann, Manfred (2007). Advanced inorganic chemistry (6th ed.). New Delhi, India: Wiley. p. 1128. ISBN 978-81-265-1338-3. https://books.google.com/books?id=U3MWRONWAmMC&pg=PA1128.
- ↑ Thermal neutron capture cross sections and resonance integrals – Fission product nuclear data. ipen.br
- ↑ Bud, Robert and Gummett, Philip Cold War, Hot Science: Applied Research in Britain's Defence Laboratories, 1945–1990, NMSI Trading Ltd, 2002 ISBN 1-900747-47-2 p. 268
- ↑ Sorokin, P. P. (1979). "Contributions of IBM to Laser Science—1960 to the Present". IBM Journal of Research and Development 23 (5): 476. doi:10.1147/rd.235.0476. Bibcode: 1979IBMJ...23..476S.
- ↑ Zhang, J. (1997). "A Saturated X-ray Laser Beam at 7 Nanometers". Science 276 (5315): 1097. doi:10.1126/science.276.5315.1097.
- ↑ Riesen, Hans; Kaczmarek, Wieslaw (2007-08-02). "Efficient X-ray Generation of Sm2+ in Nanocrystalline BaFCl/Sm3+: a Photoluminescent X-ray Storage Phosphor". Inorganic Chemistry 46 (18): 7235–7. doi:10.1021/ic062455g. PMID 17672448.
- ↑ Liu, Zhiqiang; Stevens-Kalceff, Marion; Riesen, Hans (2012-03-16). "Photoluminescence and Cathodoluminescence Properties of Nanocrystalline BaFCl:Sm3+ X-ray Storage Phosphor". Journal of Physical Chemistry C 116 (14): 8322–8331. doi:10.1021/jp301338b.
- ↑ Wang, Xianglei; Liu, Zhiqiang; Stevens-Kalceff, Marion; Riesen, Hans (August 12, 2014). "Mechanochemical Preparation of Nanocrystalline BaFCl Doped with Samarium in the 2+ Oxidation State". Inorganic Chemistry 53 (17): 8839–8841. doi:10.1021/ic500712b. PMID 25113662. http://handle.unsw.edu.au/1959.4/unsworks_34984.
- ↑ "Dosimetry&Imaging Pty Ltd". http://www.oelimaging.com.
- ↑ , Bruce G.; Lia Krusin-Elbaum & Xiao Hu Liu et al."Piezo-Driven Non-Volatile Memory Cell with Hysteretic Resistance" patent 20100073997, issued 2010-03-25
- ↑ "About us". https://tenzo-sms.ru/en/about.
- ↑ Kaminskii, V. V.; Solov'ev, S. M.; Golubkov, A. V. (2002). "Electromotive Force Generation in Homogeneously Heated Semiconducting Samarium Monosulfide". Technical Physics Letters 28 (3): 229. doi:10.1134/1.1467284. Bibcode: 2002TePhL..28..229K. http://www.tenzo-sms.ru/en/articles/5.
- ↑ Bowen, Robert and Attendorn, H -G Isotopes in the Earth Sciences, Springer, 1988, ISBN 0-412-53710-9, pp. 270 ff
- ↑ Baur, F.; Katelnikovas, A.; Sazirnakovas, S.; Jüstel, T. (2014). "Synthesis and Optical Properties of Li3Ba2La3(MoO4)8:Sm3+". Zeitschrift für Naturforschung B 69 (2): 183–192. doi:10.5560/ZNB.2014-3279.
- ↑ Caton, Ronald G.; Pedersen, Todd R.; Groves, Keith M.; Hines, Jack; Cannon, Paul S.; Jackson-Booth, Natasha; Parris, Richard T.; Holmes, Jeffrey M. et al. (May 2017). "Artificial ionospheric modification: The Metal Oxide Space Cloud experiment". Radio Science 52 (5): 539–558. doi:10.1002/2016rs005988. Bibcode: 2017RaSc...52..539C. https://pure-oai.bham.ac.uk/ws/files/40897676/Caton_et_al_2017_Radio_Science.pdf.
- ↑ Zell, Holly (2013-06-07). "First of Four Sounding Rockets Launched from the Marshall Islands" (in en). https://www.nasa.gov/mission_pages/sounding-rockets/news/mosc.html.
- ↑ Li, G.; Xiang, Z.; Yu, F.; Asaba, T.; Lawson, B.; Cai, P.; Tinsman, C.; Berkley, A. et al. (2014-12-05). "Two-dimensional Fermi surfaces in Kondo insulator SmB 6" (in en). Science 346 (6214): 1208–1212. doi:10.1126/science.1250366. ISSN 0036-8075. PMID 25477456. Bibcode: 2014Sci...346.1208L. https://www.science.org/doi/10.1126/science.1250366.
- ↑ Botimer, J.; Kim, D. J.; Thomas, S.; Grant, T.; Fisk, Z.; Jing Xia (2013). "Robust Surface Hall Effect and Nonlocal Transport in SmB6: Indication for an Ideal Topological Insulator". Scientific Reports 3 (3150): 3150. doi:10.1038/srep03150. PMID 24193196. Bibcode: 2013NatSR...3.3150K.
- ↑ Zhang, Xiaohang; Butch, N. P.; Syers, P.; Ziemak, S.; Greene, Richard L.; Paglione, Johnpierre (2013). "Hybridization, Correlation, and In-Gap States in the Kondo Insulator SmB6". Physical Review X 3 (1). doi:10.1103/PhysRevX.3.011011. Bibcode: 2013PhRvX...3a1011Z.
- ↑ Wolgast, Steven; Kurdak, Cagliyan; Sun, Kai et al. (2012). "Low-temperature surface conduction in the Kondo insulator SmB6". Physical Review B 88 (18). doi:10.1103/PhysRevB.88.180405. Bibcode: 2013PhRvB..88r0405W.
- ↑ Sigma-Aldrich Co., Samarium 263184.
- ↑ "Safety Data Sheet". Thermo Fisher Scientific. 2020-02-14. https://www.fishersci.com/store/msds?partNumber=AA40297DH&productDescription=SAMR+RD+6.35MMDIA+99.9+25MM&vendorId=VN00024248&countryCode=US&language=en.
- ↑ Bayouth, J. E.; Macey, D. J.; Kasi, L. P.; Fossella, F. V. (1994). "Dosimetry and toxicity of samarium-153-EDTMP administered for bone pain due to skeletal metastases". Journal of Nuclear Medicine 35 (1): 63–69. PMID 7505819. https://jnm.snmjournals.org/content/35/1/63.long.
Bibliography
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth–Heinemann. ISBN 978-0-08-037941-8.
External links
 |

