Biology:Francisella tularensis
| Francisella tularensis | |
|---|---|
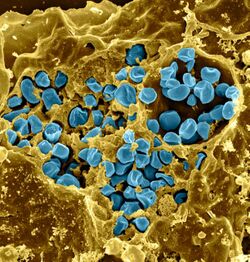
| |
| Francisella tularensis bacteria (blue) infecting a macrophage (yellow) | |
| Scientific classification | |
| Domain: | Bacteria |
| Phylum: | Pseudomonadota |
| Class: | Gammaproteobacteria |
| Order: | Thiotrichales |
| Family: | Francisellaceae |
| Genus: | Francisella |
| Species: | F. tularensis
|
| Binomial name | |
| Francisella tularensis (McCoy and Chapin 1912)
Dorofe'ev 1947 | |
Francisella tularensis is a pathogenic species of Gram-negative coccobacillus, an aerobic bacterium.[1] It is nonspore-forming, nonmotile,[2] and the causative agent of tularemia, the pneumonic form of which is often lethal without treatment. It is a fastidious, facultative intracellular bacterium, which requires cysteine for growth.[3] Due to its low infectious dose, ease of spread by aerosol, and high virulence, F. tularensis is classified as a Tier 1 Select Agent by the U.S. government, along with other potential agents of bioterrorism such as Yersinia pestis, Bacillus anthracis, and Ebola virus. When found in nature, Francisella tularensis can survive for several weeks at low temperatures in animal carcasses, soil, and water. In the laboratory, F. tularensis appears as small rods (0.2 by 0.2 µm), and is grown best at 35–37 °C.[4]
History
This species was discovered in ground squirrels in Tulare County, California in 1911. Bacterium tularense was soon isolated by George Walter McCoy (1876–1952) of the US Plague Lab in San Francisco and reported in 1912. In 1922, Edward Francis (1872–1957), a physician and medical researcher from Ohio, discovered that Bacterium tularense was the causative agent of tularemia, after studying several cases with symptoms of the disease. Later, it became known as Francisella tularensis, in honor of the discovery by Francis.[5][6][7]
The disease was also described in the Fukushima region of Japan by Hachiro Ohara in the 1920s, where it was associated with hunting rabbits.[8] In 1938, Soviet bacteriologist Vladimir Dorofeev (1911–1988) and his team recreated the infectious cycle of the pathogen in humans, and his team was the first to create protection measures. In 1947, Dorofeev independently isolated the pathogen that Francis discovered in 1922. Hence it is commonly known as Francisella dorofeev in former Soviet countries.
Classification
Three subspecies (biovars) of F. tularensis are recognised (as of 2020):[8]
- F. t. tularensis (or type A), found predominantly in North America, is the most virulent of the four known subspecies, and is associated with lethal pulmonary infections. This includes the primary type A laboratory strain, SCHUS4.
- F. t. holarctica (also known as biovar F. t. palearctica or type B) is found predominantly in Europe and Asia, but rarely leads to fatal disease. An attenuated live vaccine strain of subspecies F. t. holarctica has been described, though it is not yet fully licensed by the Food and Drug Administration as a vaccine. This subspecies lacks the citrulline ureidase activity and ability to produce acid from glucose of biovar F. t. palearctica.
- F. t. mediasiatica, is found primarily in central Asia; little is currently known about this subspecies or its ability to infect humans.
Additionally, F. novicida[9] has sometimes previously been classified as F. t. novicida. It was characterized as a relatively nonvirulent Francisella; only two tularemia cases in North America have been attributed to the organism, and these were only in severely immunocompromised individuals.
Pathogenesis
F. tularensis has been reported in invertebrates including insects and ticks, and vertebrates such as birds, amphibians, reptiles,[citation needed] fish[citation needed] and mammals, including humans.[8] Human infection is often caused by vectors, particularly ticks but also mosquitos, deer flies and horse-flies. Direct contact with infected animals or carcasses is another source.[8] Important reservoir hosts include lagomorphs (e.g. rabbits), rodents,[8] galliform birds and deer.[citation needed] Infection via fomites (objects) is also important.[8] Human-to-human transmission has been demonstrated via solid organ transplantation.[10]
F. tularensis can survive for weeks outside a mammalian host[citation needed] and has been found in water,[8] grassland, and haystacks. Aerosols containing the bacteria may be generated by disturbing carcasses due to brush cutting or lawn mowing; as a result, tularemia has been referred to as "lawnmower disease". Epidemiological studies have shown a positive correlation between occupations involving the above activities and infection with F. tularensis.[citation needed]
Human infection with F. tularensis can occur by several routes. Portals of entry are through blood and the respiratory system. The most common occurs via skin contact, yielding an ulceroglandular form of the disease. Inhalation of bacteria,[8] particularly biovar F. t. tularensis,[citation needed] leads to the potentially lethal pneumonic tularemia. While the pulmonary and ulceroglandular forms of tularemia are more common, other routes of inoculation have been described and include oropharyngeal infection due to consumption of contaminated food or water, and conjunctival infection due to inoculation at the eye.[8]
Lifecycle
F. tularensis is a facultative intracellular bacterium that is capable of infecting most cell types, but primarily infects macrophages in the host organism.[citation needed] Entry into the macrophage occurs by phagocytosis and the bacterium is sequestered from the interior of the infected cell by a phagosome.[citation needed] F. tularensis then breaks out of this phagosome into the cytosol and rapidly proliferates. Eventually, the infected cell undergoes apoptosis, and the progeny bacteria are released in a single "burst" event[11] to initiate new rounds of infection.
Virulence factors

The virulence mechanisms for F. tularensis have not been well characterized. Like other intracellular bacteria that break out of phagosomal compartments to replicate in the cytosol, F. tularensis strains produce different hemolytic agents, which may facilitate degradation of the phagosome.[12] A hemolysin activity, named NlyA, with immunological reactivity to Escherichia coli anti-HlyA antibody, was identified in biovar F. t. novicida.[13] Acid phosphatase AcpA has been found in other bacteria to act as a hemolysin, whereas in Francisella, its role as a virulence factor is under vigorous debate.
F. tularensis contains type VI secretion system (T6SS), also present in some other pathogenic bacteria.[14] It also contains a number of ATP-binding cassette (ABC) proteins that may be linked to the secretion of virulence factors.[15] F. tularensis uses type IV pili to bind to the exterior of a host cell and thus become phagocytosed. Mutant strains lacking pili show severely attenuated pathogenicity.
The expression of a 23-kD protein known as IglC is required for F. tularensis phagosomal breakout and intracellular replication; in its absence, mutant F. tularensis cells die and are degraded by the macrophage. This protein is located in a putative pathogenicity island regulated by the transcription factor MglA.
F. tularensis, in vitro, downregulates the immune response of infected cells, a tactic used by a significant number of pathogenic organisms to ensure their replication is (albeit briefly) unhindered by the host immune system by blocking the warning signals from the infected cells. This downmodulation of the immune response requires the IglC protein, though again the contributions of IglC and other genes are unclear. Several other putative virulence genes exist, but have yet to be characterized for function in F. tularensis pathogenicity.
Genetics
Like many other bacteria, F. tularensis undergoes asexual replication. Bacteria divide into two daughter cells, each of which contains identical genetic information. Genetic variation may be introduced by mutation or horizontal gene transfer.
The genome of F. t. tularensis strain SCHU4 has been sequenced.[16] The studies resulting from the sequencing suggest a number of gene-coding regions in the F. tularensis genome are disrupted by mutations, thus create blocks in a number of metabolic and synthetic pathways required for survival. This indicates F. tularensis has evolved to depend on the host organism for certain nutrients and other processes ordinarily taken care of by these disrupted genes.
The F. tularensis genome contains unusual transposon-like elements resembling counterparts that normally are found in eukaryotic organisms.
Phylogenetics
Much of the known global genetic diversity of F. t. holarctica is present in Sweden.[17] This suggests this subspecies originated in Scandinavia and spread from there to the rest of Eurosiberia.
Use as a biological weapon
When the U.S. biological warfare program ended in 1969, F. tularensis was one of seven standardized biological weapons it had developed as part of German-American cooperation in the 1920s–1930s.[18]
Diagnosis, treatment, and prevention

- Diagnosis
Infection by F. tularensis is diagnosed by clinicians based on symptoms and patient history, imaging, and laboratory studies.
- Treatment
Tularemia is treated with antibiotics, such as aminoglycosides, tetracyclines, or fluoroquinolones. About 15 proteins were suggested that could facilitate drug and vaccine design pipeline.[19]
- Prevention
Preventive measures include preventing bites from ticks, flies, and mosquitos; ensuring that all game is cooked thoroughly; refraining from drinking untreated water and using insect repellents. If working with cultures of F. tularensis, in the lab, wear a gown, impermeable gloves, mask, and eye protection. When dressing game, wear impermeable gloves. A live attenuated vaccine is available for individuals who are at high risk for exposure such, as laboratory personnel.[20]
Genomics
- Francisella Genome Projects (from Genomes OnLine Database)
- Comparative Analysis of Francisella Genomes (at DOE's IMG system)
See also
References
- ↑ "Francisella tularensis". Wadsworth Center: New York State Department of Health. http://www.health.ny.gov/guidance/oph/wadsworth/francisella_tularensis.pdf.
- ↑ "Tularemia (Francisella tularensis)". Michigan Department of Community Health. http://michigan.gov/documents/Healthcare_provider_FAQ-tularemia_08-2004_104331_7.pdf.
- ↑ Ryan KJ, ed (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. pp. 488–90. ISBN 0-8385-8529-9.
- ↑ "Francisella tularensis". https://microbewiki.kenyon.edu/index.php/Francisella_tularensis.
- ↑ Tärnvik, A.; Berglund, L. (1 February 2003). "Tularaemia". European Respiratory Journal 21 (2): 361–373. doi:10.1183/09031936.03.00088903. PMID 12608453. https://erj.ersjournals.com/content/21/2/361. Retrieved 20 March 2022.
- ↑ McCoy GW, Chapin CW. Bacterium tularense, the cause of a plaguelike disease of rodents. Public Health Bull 1912;53:17–23.
- ↑ Jeanette Barry, Notable Contributions to Medical Research by Public Health Service Scientists. National Institute of Health, Public Health Service Publication No. 752, 1960, p. 36.
- ↑ 8.0 8.1 8.2 8.3 8.4 8.5 8.6 8.7 8.8 Sam R. Telford III, Heidi K. Goethert (2020). "Ecology of Francisella tularensis", Annual Review of Entomology 65: 351–372 doi:10.1146/annurev-ento-011019-025134
- ↑ Sjöstedt AB. "Genus I. Francisella Dorofe'ev 1947, 176AL". Bergey's Manual of Systematic Bacteriology. 2 (The Proteobacteria), part B (The Gammaproteobacteria) (2nd ed.). New York: Springer. pp. 200–210.
- ↑ Nelson CA, Murua C, Jones JM, Mohler K, Zhang Y, Wiggins L (2019). "Francisella tularensis Transmission by Solid Organ Transplantation, 2017.". Emerg Infect Dis 25 (4): 767–775. doi:10.3201/eid2504.181807. PMID 30730826.
- ↑ Carruthers, Jonathan; Lythe, Grant; López-García, Martín; Gillard, Joseph; Laws, Thomas R.; Lukaszewski, Roman; Molina-París, Carmen (2020-06-01). "Stochastic dynamics of Francisella tularensis infection and replication" (in en). PLOS Computational Biology 16 (6): e1007752. doi:10.1371/journal.pcbi.1007752. ISSN 1553-7358. PMID 32479491. Bibcode: 2020PLSCB..16E7752C.
- ↑ Mohapatra, Nrusingh P.; Soni, Shilpa; Reilly, Thomas J.; Liu, Jirong; Klose, Karl E.; Gunn, John S. (2008). "Combined Deletion of Four Francisella novicida Acid Phosphatases Attenuates Virulence and Macrophage Vacuolar Escape". Infection and Immunity 76 (8): 3690–3699. doi:10.1128/IAI.00262-08. PMID 18490464. PMC 2493204. http://iai.asm.org/cgi/reprint/76/8/3690.
- ↑ "Wiley Online Library". https://onlinelibrary.wiley.com/.
- ↑ Spidlova, Petra; Stulik, Jiri (2017). "Francisella tularensis type VI secretion system comes of age". Virulence 8 (6): 628–631. doi:10.1080/21505594.2016.1278336. ISSN 2150-5594. PMID 28060573.
- ↑ "The identification and evaluation of ATP binding cassette systems in the intracellular bacterium Francisella tularensis". Res Microbiol 157 (6): 593–604. 2006. doi:10.1016/j.resmic.2005.12.004. PMID 16503121.
- ↑ Larsson P; Oyston P; Chain P et al. (2005). "The complete genome sequence of Francisella tularensis, the causative agent of tularemia". Nat Genet 37 (2): 153–9. doi:10.1038/ng1499. PMID 15640799.
- ↑ Karlsson E, Svensson K, Lindgren P, Byström M, Sjödin A, Forsman M, Johansson A (2012) The phylogeographic pattern of Francisella tularensis in Sweden indicates a Scandinavian origin of Eurosiberian tularaemia. Environ Microbiol doi: 10.1111/1462-2920.12052
- ↑ Croddy, Eric C. and Hart, C. Perez-Armendariz J., Chemical and Biological Warfare, (Google Books), Springer, 2002, pp. 30–31, (ISBN:0387950761), accessed October 24, 2008.
- ↑ Francisella tularensis: In silico Identification of Drug and Vaccine Targets by Metabolic Pathway Analysis J Harati, J Fallah The 6th Conference on Bioinformatics
- ↑ "Part IV : Acute Communicable Diseases". http://publichealth.lacounty.gov/acd/procs/b73/B73Part4.pdf.
External links
- Francisella tularensis information from the CDC/National Center for Infectious Diesase:
- BioHealthBase Bioinformatics Resource Center The National Institute of Allergy and Infectious Disease (NIAID) supports a public database describing the molecular genetics of F. tularensis. The website describes the genes, proteins, and cellular characteristics of the pathogen.
- Type strain of Francisella tularensis at BacDive – the Bacterial Diversity Metadatabase
Wikidata ☰ Q1003460 entry
 |

