Biology:Insect
Insects (from Latin insectum) are hexapod invertebrates of the class Insecta. They are the largest group within the arthropod phylum. Insects have a chitinous exoskeleton, a three-part body (head, thorax and abdomen), three pairs of jointed legs, compound eyes, and a pair of antennae. Insects are the most diverse group of animals, with more than a million described species; they represent more than half of all animal species.
The insect nervous system consists of a brain and a ventral nerve cord. Most insects reproduce by laying eggs. Insects breathe air through a system of paired openings along their sides, connected to small tubes that take air directly to the tissues. The blood therefore does not carry oxygen; it is only partly contained in vessels, and some circulates in an open hemocoel. Insect vision is mainly through their compound eyes, with additional small ocelli. Many insects can hear, using tympanal organs, which may be on the legs or other parts of the body. Their sense of smell is via receptors, usually on the antennae and the mouthparts.
Nearly all insects hatch from eggs. Insect growth is constrained by the inelastic exoskeleton, so development involves a series of molts. The immature stages often differ from the adults in structure, habit, and habitat. Groups that undergo four-stage metamorphosis often have a nearly immobile pupa. Insects that undergo three-stage metamorphosis lack a pupa, developing through a series of increasingly adult-like nymphal stages. The higher level relationship of the insects is unclear. Fossilized insects of enormous size have been found from the Paleozoic Era, including giant dragonfly-like insects with wingspans of 55 to 70 cm (22 to 28 in). The most diverse insect groups appear to have coevolved with flowering plants.
Adult insects typically move about by walking and flying; some can swim. Insects are the only invertebrates that can achieve sustained powered flight; insect flight evolved just once. Many insects are at least partly aquatic, and have larvae with gills; in some species, the adults too are aquatic. Some species, such as water striders, can walk on the surface of water. Insects are mostly solitary, but some, such as bees, ants and termites, are social and live in large, well-organized colonies. Others, such as earwigs, provide maternal care, guarding their eggs and young. Insects can communicate with each other in a variety of ways. Male moths can sense the pheromones of female moths over great distances. Other species communicate with sounds: crickets stridulate, or rub their wings together, to attract a mate and repel other males. Lampyrid beetles communicate with light.
Humans regard many insects as pests, especially those that damage crops, and attempt to control them using insecticides and other techniques. Others are parasitic, and may act as vectors of diseases. Insect pollinators are essential to the reproduction of many flowering plants and so to their ecosystems. Many insects are ecologically beneficial as predators of pest insects, while a few provide direct economic benefit. Two species in particular are economically important and were domesticated many centuries ago: silkworms for silk and honey bees for honey. Insects are consumed as food in 80% of the world's nations, by people in roughly 3,000 ethnic groups. Human activities are having serious effects on insect biodiversity.
Etymology
The word insect comes from the Latin word insectum from in + sĕco, "cut up",[1] as insects appear to be cut into three parts. The Latin word was introduced by Pliny the Elder who calqued the Ancient Greek word ἔντομον éntomon "insect" (as in entomology) from ἔντομος éntomos "cut in pieces";[2] this was Aristotle's term for this class of life in his biology, also in reference to their notched bodies. The English word insect first appears in 1601 in Philemon Holland's translation of Pliny.[3][4]
Insects and other bugs
Distinguishing features
In common speech, insects and other terrestrial arthropods are often called bugs [lower-alpha 1] or creepy crawlies. Entomologists to some extent reserve the name "bugs" for a narrow category of "true bugs", insects of the order Hemiptera, such as cicadas and shield bugs.[6] Other terrestrial arthropods, such as centipedes, millipedes, woodlice, spiders, mites and scorpions, are sometimes confused with insects, since they have a jointed exoskeleton.[7] Adult insects are the only arthropods that ever have wings, with up to two pairs on the thorax. Whether winged or not, adult insects can be distinguished by their three-part body plan, with head, thorax, and abdomen; they have three pairs of legs on the thorax.[8]
- Insects and other 'bugs' that could be confused with them
-
Insect: Six legs, three-part body
(head, thorax, abdomen),
up to two pairs of wings -
Spider: eight legs,
two-part body -
Woodlouse: seven pairs of legs, seven body segments (plus head and tail)
-
Centipede: many legs,
one pair per segment -
Millipede: many legs,
two pairs per segment
Diversity
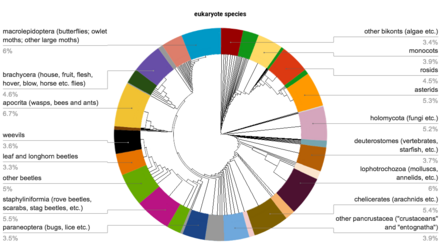
Estimates of the total number of insect species vary considerably, suggesting that there are perhaps some 5.5 million insect species in existence, of which about one million have been described and named.[9] These constitute around half of all eukaryote species, including animals, plants, and fungi.[10] The most diverse insect orders are the Hemiptera (true bugs), Lepidoptera (butterflies and moths), Diptera (true flies), Hymenoptera (wasps, ants, and bees), and Coleoptera (beetles), each with more than 100,000 described species.[9]
- Insects are extremely diverse. Five groups each have over 100,000 described species.
-
True bugs
(Hemiptera) -
Butterflies and moths
(Lepidoptera) -
Flies
(Diptera) -
Wasps
(Hymenoptera) -
Beetles
(Coleoptera)
Distribution and habitats
- Insects occur in habitats as varied as snow, freshwater, the tropics, desert, and even the sea.
-
The snow scorpionfly Boreus hyemalis on snow
-
The great diving beetle Dytiscus marginalis larva in a pond
-
The green orchid bee Euglossa dilemma of Central America
-
The desert locust Schistocerca gregaria laying eggs in sand
-
Sea skater Halobates on a Hawaii beach
Insects are distributed over every continent and almost every terrestrial habitat. There are many more species in the tropics, especially in rainforests, than in temperate zones.[11] The world's regions have received widely differing amounts of attention from entomologists. The British Isles have been thoroughly surveyed, so that Gullan and Cranston 2014 state that the total of around 22,500 species is probably within 5% of the actual number there; they comment that Canada's list of 30,000 described species is surely over half of the actual total. They add that the 3,000 species of the American Arctic must be broadly accurate. In contrast, a large majority of the insect species of the tropics and the southern hemisphere are probably undescribed.[11] Some 30–40,000 species inhabit freshwater; very few insects, perhaps a hundred species, are marine.[12] Insects such as snow scorpionflies flourish in cold habitats including the Arctic and at high altitude.[13] Insects such as desert locusts, ants, beetles, and termites are adapted to some of the hottest and driest environments on earth, such as the Sonoran Desert.[14]
Phylogeny and evolution
External phylogeny
Insects form a clade, a natural group with a common ancestor, among the arthropods.[15] A phylogenetic analysis by Kjer et al. (2016) places the insects among the Hexapoda, six-legged animals with segmented bodies; their closest relatives are the Diplura (bristletails).[16]
| Hexapoda |
| ||||||||||||||||||
Internal phylogeny
The internal phylogeny is based on the works of Wipfler et al. 2019 for the Polyneoptera,[17] Johnson et al. 2018 for the Paraneoptera,[18] and Kjer et al. 2016 for the Holometabola.[19] The numbers of described extant species (boldface for groups with over 100,000 species) are from Stork 2018.[9]
| Insecta |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Taxonomy
Early
| Diagram of Linnaeus's key to his seven orders of insect, 1758[20] | |||||||||||||||||||||||||||||||||||||||
|
Aristotle was the first to describe the insects as a distinct group. He placed them as the second-lowest level of animals on his scala naturae, above the spontaneously generating sponges and worms, but below the hard-shelled marine snails. His classification remained in use for many centuries.[21]
In 1758, in his Systema Naturae,[22] Carl Linnaeus divided the animal kingdom into six classes including Insecta. He created seven orders of insect according to the structure of their wings. These were the wingless Aptera, the two-winged Diptera, and five four-winged orders: the Coleoptera with fully-hardened forewings; the Hemiptera with partly-hardened forewings; the Lepidoptera with scaly wings; the Neuroptera with membranous wings but no sting; and the Hymenoptera, with membranous wings and a sting.[20]
Jean-Baptiste de Lamarck, in his 1809 Philosophie Zoologique, treated the insects as one of nine invertebrate phyla.[23] In his 1817 Le Règne Animal, Georges Cuvier grouped all animals into four embranchements ("branches" with different body plans), one of which was the articulated animals, containing arthropods and annelids.[24] This arrangement was followed by the embryologist Karl Ernst von Baer in 1828, the zoologist Louis Agassiz in 1857, and the comparative anatomist Richard Owen in 1860.[25] In 1874, Ernst Haeckel divided the animal kingdom into two subkingdoms, one of which was Metazoa for the multicellular animals. It had five phyla, including the articulates.[26][25]
Modern
Traditional morphology-based systematics have usually given the Hexapoda the rank of superclass,[27] and identified four groups within it: insects (Ectognatha), Collembola, Protura, and Diplura, the latter three being grouped together as the Entognatha on the basis of internalized mouth parts.[28]
The use of phylogenetic data has brought about numerous changes in relationships above the level of orders.[28] Insects can be divided into two groups historically treated as subclasses: wingless insects or Apterygota, and winged insects or Pterygota. The Apterygota traditionally consisted of the primitively wingless orders Archaeognatha (jumping bristletails) and Zygentoma (silverfish). However, Apterygota is not monophyletic, as Archaeognatha are sister to all other insects, based on the arrangement of their mandibles, while the Pterygota, the winged insects, emerged from within the Dicondylia, alongside the Zygentoma.[29]
The Pterygota (Palaeoptera and Neoptera) are winged and have hardened plates on the outside of their body segments; the Neoptera have muscles that allow their wings to fold flat over the abdomen. Neoptera can be divided into groups with incomplete metamorphosis (Polyneoptera and Paraneoptera) and those with complete metamorphosis (Holometabola). The molecular finding that the traditional louse orders Mallophaga and Anoplura are within Psocoptera has led to the new taxon Psocodea.[30] Phasmatodea and Embiidina have been suggested to form the Eukinolabia.[31] Mantodea, Blattodea, and Isoptera form a monophyletic group, Dictyoptera.[32] Fleas are now thought to be closely related to boreid mecopterans.[33]
Evolutionary history
The oldest fossil that may be a primitive wingless insect is Leverhulmia from the Early Devonian Windyfield chert.[34] The oldest known flying insects are from the mid-Carboniferous, around 328–324 million years ago. The group subsequently underwent a rapid explosive diversification. Claims that they originated substantially earlier, during the Silurian or Devonian (some 400 million years ago) based on molecular clock estimates, are unlikely to be correct, given the fossil record.[35]
Four large-scale radiations of insects have occurred: beetles (from about 300 million years ago), flies (from about 250 million years ago), moths and wasps (both from about 150 million years ago).[36]
The remarkably successful Hymenoptera (wasps, bees, and ants) appeared some 200 million years ago in the Triassic Period, but achieved their wide diversity more recently in the Cenozoic era, which began 66 million years ago. Some highly successful insect groups evolved in conjunction with flowering plants, a powerful illustration of coevolution. Insects were among the earliest terrestrial herbivores and acted as major selection agents on plants.[37] Plants evolved chemical defenses against this herbivory and the insects, in turn, evolved mechanisms to deal with plant toxins. Many insects make use of these toxins to protect themselves from their predators. Such insects often advertise their toxicity using warning colors.[38]
-
The giant dragonfly-like insect Meganeura monyi grew to wingspans of 75 cm (2 ft 6 in) in the late Carboniferous, around 300 million years ago.[39]
Morphology and physiology
External
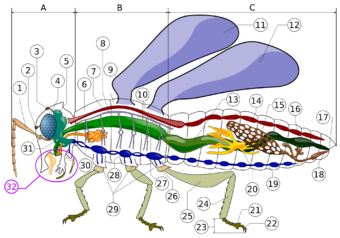
A- Head B- Thorax C- Abdomen
- antenna
- ocellus (lower)
- ocellus (upper)
- compound eye
- brain (cerebral ganglia)
- prothorax
- dorsal blood vessel
- tracheal tubes (trunk with spiracle)
- mesothorax
- metathorax
- forewing
- hindwing
- midgut (stomach)
- dorsal tube (heart)
- ovary
- hindgut (intestine, rectum, anus)
- anus
- oviduct
- nerve cord (abdominal ganglia)
- Malpighian tubules
- tarsal pads
- claws
- tarsus
- tibia
- femur
- trochanter
- foregut (crop, gizzard)
- thoracic ganglion
- coxa
- salivary gland
- subesophageal ganglion
- mouthparts
Three-part body
Insects have a segmented body supported by an exoskeleton, the hard outer covering made mostly of chitin. The body is organized into three interconnected units: the head, thorax and abdomen. The head supports a pair of sensory antennae, a pair of compound eyes, zero to three simple eyes (or ocelli) and three sets of variously modified appendages that form the mouthparts. The thorax carries the three pairs of legs and up to two pairs of wings. The abdomen contains most of the digestive, respiratory, excretory and reproductive structures.[8]
Segmentation
The head is enclosed in a hard, heavily sclerotized, unsegmented head capsule, which contains most of the sensing organs, including the antennae, compound eyes, ocelli, and mouthparts.[40] The thorax is composed of three sections named (from front to back) the prothorax, mesothorax and metathorax. The prothorax carries the first pair of legs. The mesothorax carries the second pair of legs and the front wings. The metathorax carries the third pair of legs and the hind wings.[8][40] The abdomen is the largest part of the insect, typically with 11–12 segments, and is less strongly sclerotized than the head or thorax. Each segment of the abdomen has sclerotized upper and lower plates (the tergum and sternum), connected to adjacent sclerotized parts by membranes. Each segment carries a pair of spiracles.[40]
Exoskeleton
The outer skeleton, the cuticle, is made up of two layers: the epicuticle, a thin and waxy water-resistant outer layer without chitin, and a lower layer, the thick chitinous procuticle. The procuticle has two layers: an outer exocuticle and an inner endocuticle. The tough and flexible endocuticle is built from numerous layers of fibrous chitin and proteins, criss-crossing each other in a sandwich pattern, while the exocuticle is rigid and sclerotized.[41][42] As an adaptation to life on land, insects have an enzyme that uses atmospheric oxygen to harden their cuticle, unlike crustaceans which use heavy calcium compounds for the same purpose. This makes the insect exoskeleton a lightweight material.[43]
Internal systems
Nervous
The nervous system of an insect consists of a brain and a ventral nerve cord. The head capsule is made up of six fused segments, each with either a pair of ganglia, or a cluster of nerve cells outside of the brain. The first three pairs of ganglia are fused into the brain, while the three following pairs are fused into a structure of three pairs of ganglia under the insect's esophagus, called the subesophageal ganglion.[44] The thoracic segments have one ganglion on each side, connected into a pair per segment. This arrangement is also seen in the first eight segments of the abdomen. Many insects have fewer ganglia than this.[45] Insects are capable of learning.[46]
Digestive
An insect uses its digestive system to extract nutrients and other substances from the food it consumes.[47] There is extensive variation among different orders, life stages, and even castes in the digestive system of insects.[48] The gut runs lengthwise through the body. It has three sections, with paired salivary glands and salivary reservoirs.[49] By moving its mouthparts the insect mixes its food with saliva.[50][51] Some insects, like flies, expel digestive enzymes onto their food to break it down, but most insects digest their food in the gut.[52] The foregut is lined with cuticule as protection from tough food. It includes the mouth, pharynx, and crop which stores food.[53] Digestion starts in the mouth with enzymes in the saliva. Strong muscles in the pharynx pump fluid into the mouth, lubricating the food, and enabling certain insects to feed on blood or from the xylem and phloem transport vessels of plants.[54] Once food leaves the crop, it passes to the midgut, where the majority of digestion takes place. Microscopic projections, microvilli, increase the surface area of the wall to absorb nutrients.[55] In the hindgut, undigested food particles are joined by uric acid to form fecal pellets; most of the water is absorbed, leaving a dry pellet to be eliminated. Insects may have one to hundreds of Malpighian tubules. These remove nitrogenous wastes from the hemolymph of the insect and regulate osmotic balance. Wastes and solutes are emptied directly into the alimentary canal, at the junction between the midgut and hindgut.[56]
Reproductive
The reproductive system of female insects consist of a pair of ovaries, accessory glands, one or more spermathecae to store sperm, and ducts connecting these parts. The ovaries are made up of a variable number of egg tubes, ovarioles. Female insects make eggs, receive and store sperm, manipulate sperm from different males, and lay eggs. Accessory glands produce substances to maintain sperm and to protect the eggs. They can produce glue and protective substances for coating eggs, or tough coverings for a batch of eggs called oothecae.[57]
For males, the reproductive system consists of one or two testes, suspended in the body cavity by tracheae. The testes contain sperm tubes or follicles in a membranous sac. These connect to a duct that leads to the outside. The terminal portion of the duct may be sclerotized to form the intromittent organ, the aedeagus.[58]
Respiratory
Insect respiration is accomplished without lungs. Instead, insects have a system of internal tubes and sacs through which gases either diffuse or are actively pumped, delivering oxygen directly to tissues that need it via their tracheae and tracheoles. In most insects, air is taken in through paired spiracles, openings on the sides of the abdomen and thorax. The respiratory system limits the size of insects. As insects get larger, gas exchange via spiracles becomes less efficient, and thus the heaviest insect currently weighs less than 100 g. However, with increased atmospheric oxygen levels, as were present in the late Paleozoic, larger insects were possible, such as dragonflies with wingspans of more than two feet (60 cm).[59] Gas exchange patterns in insects range from continuous and diffusive ventilation, to discontinuous.[60][61][62][63]
Circulatory
Because oxygen is delivered directly to tissues via tracheoles, the circulatory system is not used to carry oxygen, and is therefore greatly reduced. The insect circulatory system is open; it has no veins or arteries, and instead consists of little more than a single, perforated dorsal tube that pulses peristaltically. This dorsal blood vessel is divided into two sections: the heart and aorta. The dorsal blood vessel circulates the hemolymph, arthropods' fluid analog of blood, from the rear of the body cavity forward.[64][65] Hemolymph is composed of plasma in which hemocytes are suspended. Nutrients, hormones, wastes, and other substances are transported throughout the insect body in the hemolymph. Hemocytes include many types of cells that are important for immune responses, wound healing, and other functions. Hemolymph pressure may be increased by muscle contractions or by swallowing air into the digestive system to aid in molting.[66]
Sensory

Many insects possess numerous specialized sensory organs able to detect stimuli including limb position (proprioception) by campaniform sensilla, light, water, chemicals (senses of taste and smell), sound, and heat.[67] Some insects such as bees can perceive ultraviolet wavelengths, or detect polarized light, while the antennae of male moths can detect the pheromones of female moths over distances of over a kilometer.[68] There is a trade-off between visual acuity and chemical or tactile acuity, such that most insects with well-developed eyes have reduced or simple antennae, and vice versa. Insects perceive sound by different mechanisms, such as thin vibrating membranes (tympana).[69] Insects were the earliest organisms to produce and sense sounds. Hearing has evolved independently at least 19 times in different insect groups.[70]
Most insects, except some cave crickets, are able to perceive light and dark. Many have acute vision capable of detecting small and rapid movements. The eyes may include simple eyes or ocelli as well as larger compound eyes. Many species can detect light in the infrared, ultraviolet and visible light wavelengths, with color vision. Phylogenetic analysis suggests that UV-green-blue trichromacy existed from at least the Devonian Period, some 400 million years ago.[71]
The individual lenses in compound eyes are immobile, but fruit flies have photoreceptor cells underneath each lens which move rapidly in and out of focus, in a series of movements called photoreceptor microsaccades. This gives them, and possibly many other insects, a much clearer image of the world than previously assumed.[72]
An insect's sense of smell is via chemical receptors, usually on the antennae and the mouthparts. These detect both airborne volatile compounds and odorants on surfaces, including pheromones from other insects and compounds released by food plants. Insects use olfaction to locate mating partners, food, and places to lay eggs, and to avoid predators. It is thus an extremely important sense, enabling insects to discriminate between thousands of volatile compounds.[73]
Some insects are capable of magnetoreception; ants and bees navigate using it both locally (near their nests) and when migrating.[74] The Brazilian stingless bee detects magnetic fields using the hair-like sensilla on its antennae.[75][76]
Reproduction and development
Life-cycles

The majority of insects hatch from eggs. The fertilization and development takes place inside the egg, enclosed by a shell (chorion) that consists of maternal tissue. In contrast to eggs of other arthropods, most insect eggs are drought resistant. This is because inside the chorion two additional membranes develop from embryonic tissue, the amnion and the serosa. This serosa secretes a cuticle rich in chitin that protects the embryo against desiccation.[77] Some species of insects, like aphids and tsetse flies, are ovoviviparous: their eggs develop entirely inside the female, and then hatch immediately upon being laid.[78] Some other species, such as in the cockroach genus Diploptera, are viviparous, gestating inside the mother and born alive.[79] Some insects, like parasitoid wasps, are polyembryonic, meaning that a single fertilized egg divides into many separate embryos.[80] Insects may be univoltine, bivoltine or multivoltine, having one, two or many broods in a year.[81]

Other developmental and reproductive variations include haplodiploidy, polymorphism, paedomorphosis or peramorphosis, sexual dimorphism, parthenogenesis, and more rarely hermaphroditism.[82][83] In haplodiploidy, which is a type of sex-determination system, the offspring's sex is determined by the number of sets of chromosomes an individual receives. This system is typical in bees and wasps.[84]
Some insects are parthenogenetic, meaning that the female can reproduce and give birth without having the eggs fertilized by a male. Many aphids undergo a cyclical form of parthenogenesis in which they alternate between one or many generations of asexual and sexual reproduction.[85][86] In summer, aphids are generally female and parthenogenetic; in the autumn, males may be produced for sexual reproduction. Other insects produced by parthenogenesis are bees, wasps and ants; in their haplodiploid system, diploid females spawn many females and a few haploid males.[78]
Metamorphosis
Metamorphosis in insects is the process of development that converts young to adults. There are two forms of metamorphosis: incomplete and complete.
Incomplete

Hemimetabolous insects, those with incomplete metamorphosis, change gradually after hatching from the egg by undergoing a series of molts through stages called instars, until the final, adult, stage is reached. An insect molts when it outgrows its exoskeleton, which does not stretch and would otherwise restrict the insect's growth. The molting process begins as the insect's epidermis secretes a new epicuticle inside the old one. After this new epicuticle is secreted, the epidermis releases a mixture of enzymes that digests the endocuticle and thus detaches the old cuticle. When this stage is complete, the insect makes its body swell by taking in a large quantity of water or air; this makes the old cuticle split along predefined weaknesses where it was thinnest.[87][88]
Complete

Holometabolism, or complete metamorphosis, is where the insect changes in four stages, an egg or embryo, a larva, a pupa and the adult or imago. In these species, an egg hatches to produce a larva, which is generally worm-like in form. This can be eruciform (caterpillar-like), scarabaeiform (grub-like), campodeiform (elongated, flattened and active), elateriform (wireworm-like) or vermiform (maggot-like). The larva grows and eventually becomes a pupa, a stage marked by reduced movement. There are three types of pupae: obtect, exarate or coarctate. Obtect pupae are compact, with the legs and other appendages enclosed. Exarate pupae have their legs and other appendages free and extended. Coarctate pupae develop inside the larval skin.[89] Insects undergo considerable change in form during the pupal stage, and emerge as adults. Butterflies are well-known for undergoing complete metamorphosis; most insects use this life cycle. Some insects have evolved this system to hypermetamorphosis. Complete metamorphosis is a trait of the most diverse insect group, the Endopterygota.[82]
Communication
Insects that produce sound can generally hear it. Most insects can hear only a narrow range of frequencies related to the frequency of the sounds they can produce. Mosquitoes can hear up to 2 kilohertz.[90] Certain predatory and parasitic insects can detect the characteristic sounds made by their prey or hosts, respectively. Likewise, some nocturnal moths can perceive the ultrasonic emissions of bats, which helps them avoid predation.[91]
Light production
A few insects, such as Mycetophilidae (Diptera) and the beetle families Lampyridae, Phengodidae, Elateridae and Staphylinidae are bioluminescent. The most familiar group are the fireflies, beetles of the family Lampyridae. Some species are able to control this light generation to produce flashes. The function varies with some species using them to attract mates, while others use them to lure prey. Cave dwelling larvae of Arachnocampa (Mycetophilidae, fungus gnats) glow to lure small flying insects into sticky strands of silk.[92] Some fireflies of the genus Photuris mimic the flashing of female Photinus species to attract males of that species, which are then captured and devoured.[93] The colors of emitted light vary from dull blue (Orfelia fultoni, Mycetophilidae) to the familiar greens and the rare reds (Phrixothrix tiemanni, Phengodidae).[94]
Sound production
Insects make sounds mostly by mechanical action of appendages. In grasshoppers and crickets, this is achieved by stridulation. Cicadas make the loudest sounds among the insects by producing and amplifying sounds with special modifications to their body to form tymbals and associated musculature. The African cicada Brevisana brevis has been measured at 106.7 decibels at a distance of 50 cm (20 in).[95] Some insects, such as the Helicoverpa zea moths, hawk moths and Hedylid butterflies, can hear ultrasound and take evasive action when they sense that they have been detected by bats.[96][97] Some moths produce ultrasonic clicks that warn predatory bats of their unpalatability (acoustic aposematism),[98] while some palatable moths have evolved to mimic these calls (acoustic Batesian mimicry).[99] The claim that some moths can jam bat sonar has been revisited. Ultrasonic recording and high-speed infrared videography of bat-moth interactions suggest the palatable tiger moth really does defend against attacking big brown bats using ultrasonic clicks that jam bat sonar.[100]
Very low sounds are produced in various species of Coleoptera, Hymenoptera, Lepidoptera, Mantodea and Neuroptera. These low sounds are produced by the insect's movement, amplified by stridulatory structures on the insect's muscles and joints; these sounds can be used to warn or communicate with other insects. Most sound-making insects also have tympanal organs that can perceive airborne sounds. Some hemipterans, such as the water boatmen, communicate via underwater sounds.[101]
Communication using surface-borne vibrational signals is more widespread among insects because of size constraints in producing air-borne sounds.[102] Insects cannot effectively produce low-frequency sounds, and high-frequency sounds tend to disperse more in a dense environment (such as foliage), so insects living in such environments communicate primarily using substrate-borne vibrations.[103]
Some species use vibrations for communicating, such as to attract mates as in the songs of the shield bug Nezara viridula.[104] Vibrations can also be used to communicate between species; lycaenid caterpillars, which form a mutualistic association with ants communicate with ants in this way.[105] The Madagascar hissing cockroach has the ability to press air through its spiracles to make a hissing noise as a sign of aggression;[106] the death's-head hawkmoth makes a squeaking noise by forcing air out of their pharynx when agitated, which may also reduce aggressive worker honey bee behavior when the two are close.[107]
Chemical communication

Many insects have evolved chemical means for communication. These semiochemicals are often derived from plant metabolites including those meant to attract, repel and provide other kinds of information. Pheromones are used for attracting mates of the opposite sex, for aggregating conspecific individuals of both sexes, for deterring other individuals from approaching, to mark a trail, and to trigger aggression in nearby individuals. Allomones benefit their producer by the effect they have upon the receiver. Kairomones benefit their receiver instead of their producer. Synomones benefit the producer and the receiver. While some chemicals are targeted at individuals of the same species, others are used for communication across species. The use of scents is especially well-developed in social insects.[108] Cuticular hydrocarbons are nonstructural materials produced and secreted to the cuticle surface to fight desiccation and pathogens. They are important, too, as pheromones, especially in social insects.[109]
Social behavior
Social insects, such as termites, ants and many bees and wasps, are eusocial.[110] They live together in such large well-organized colonies of genetically similar individuals that they are sometimes considered superorganisms. In particular, reproduction is largely limited to a queen caste; other females are workers, prevented from reproducing by worker policing. Honey bees have evolved a system of abstract symbolic communication where a behavior is used to represent and convey specific information about the environment. In this communication system, called dance language, the angle at which a bee dances represents a direction relative to the sun, and the length of the dance represents the distance to be flown.[111] Bumblebees too have some social communication behaviors. Bombus terrestris, for example, more rapidly learns about visiting unfamiliar, yet rewarding flowers, when they can see a conspecific foraging on the same species.[112]
Only insects that live in nests or colonies possess fine-scale spatial orientation. Some can navigate unerringly to a single hole a few millimeters in diameter among thousands of similar holes, after a trip of several kilometers. In philopatry, insects that hibernate are able to recall a specific location up to a year after last viewing the area of interest.[113] A few insects seasonally migrate large distances between different geographic regions, as in the continent-wide monarch butterfly migration.[114]
Care of young
Eusocial insects build nests, guard eggs, and provide food for offspring full-time. Most insects, however, lead short lives as adults, and rarely interact with one another except to mate or compete for mates. A small number provide parental care, where they at least guard their eggs, and sometimes guard their offspring until adulthood, possibly even feeding them. Many wasps and bees construct a nest or burrow, store provisions in it, and lay an egg upon those provisions, providing no further care.[115]
Locomotion
Flight

Insects are the only group of invertebrates to have developed flight. The ancient groups of insects in the Palaeoptera, the dragonflies, damselflies and mayflies, operate their wings directly by paired muscles attached to points on each wing base that raise and lower them. This can only be done at a relatively slow rate. All other insects, the Neoptera, have indirect flight, in which the flight muscles cause rapid oscillation of the thorax: there can be more wingbeats than nerve impulses commanding the muscles. One pair of flight muscles is aligned vertically, contracting to pull the top of the thorax down, and the wings up. The other pair runs longitudinally, contracting to force the top of the thorax up and the wings down.[116][117] Most insects gain aerodynamic lift by creating a spiralling vortex at the leading edge of the wings.[118] Small insects like thrips with tiny feathery wings gain lift using the clap and fling mechanism; the wings are clapped together and pulled apart, flinging vortices into the air at the leading edges and at the wingtips.[119][120]
The evolution of insect wings has been a subject of debate; it has been suggested they came from modified gills, flaps on the spiracles, or an appendage, the epicoxa, at the base of the legs.[121] More recently, entomologists have favored evolution of wings from lobes of the notum, of the pleuron, or more likely both.[122] In the Carboniferous age, the dragonfly-like Meganeura had as much as a 50 cm (20 in) wide wingspan. The appearance of gigantic insects is consistent with high atmospheric oxygen at that time, as the respiratory system of insects constrains their size.[123] The largest flying insects today are much smaller, with the largest wingspan belonging to the white witch moth (Thysania agrippina), at approximately 28 cm (11 in).[124]
Unlike birds, small insects are swept along by the prevailing winds[125] although many larger insects migrate. Aphids are transported long distances by low-level jet streams.[126]
Walking
File:Alternating Tripod Gait.webm
Many adult insects use six legs for walking, with an alternating tripod gait. This allows for rapid walking with a stable stance; it has been studied extensively in cockroaches and ants. For the first step, the middle right leg and the front and rear left legs are in contact with the ground and move the insect forward, while the front and rear right leg and the middle left leg are lifted and moved forward to a new position. When they touch the ground to form a new stable triangle, the other legs can be lifted and brought forward in turn.[127] The purest form of the tripedal gait is seen in insects moving at high speeds. However, this type of locomotion is not rigid and insects can adapt a variety of gaits. For example, when moving slowly, turning, avoiding obstacles, climbing or slippery surfaces, four (tetrapodal) or more feet (wave-gait) may be touching the ground.[128] Cockroaches are among the fastest insect runners and, at full speed, adopt a bipedal run. More sedate locomotion is seen in the well-camouflaged stick insects (Phasmatodea). A small number of species such as Water striders can move on the surface of water; their claws are recessed in a special groove, preventing the claws from piercing the water's surface film.[62] The ocean-skaters in the genus Halobates even live on the surface of open oceans, a habitat that has few insect species.[129]
Swimming

A large number of insects live either part or the whole of their lives underwater. In many of the more primitive orders of insect, the immature stages are aquatic. In some groups, such as water beetles, the adults too are aquatic.[62]
Many of these species are adapted for under-water locomotion. Water beetles and water bugs have legs adapted into paddle-like structures. Dragonfly naiads use jet propulsion, forcibly expelling water out of their rectal chamber.[130] Other insects such as the rove beetle Stenus emit pygidial gland surfactant secretions that reduce surface tension; this enables them to move on the surface of water by Marangoni propulsion.[131][132]
Ecology
Insects play many critical roles in ecosystems, including soil turning and aeration, dung burial, pest control, pollination and wildlife nutrition.[133] For instance, termites modify the environment around their nests, encouraging grass growth;[134] many beetles are scavengers; dung beetles recycle biological materials into forms useful to other organisms.[135][136] Insects are responsible for much of the process by which topsoil is created.[137]
Defense

Insects are mostly small, soft bodied, and fragile compared to larger lifeforms. The immature stages are small, move slowly or are immobile, and so all stages are exposed to predation and parasitism. Insects accordingly employ multiple defensive strategies, including camouflage, mimicry, toxicity and active defense.[138] Many insects rely on camouflage to avoid being noticed by their predators or prey.[139] It is common among leaf beetles and weevils that feed on wood or vegetation.[138] Stick insects mimic the forms of sticks and leaves.[140] Many insects use mimicry to deceive predators into avoiding them. In Batesian mimicry, edible species, such as of hoverflies (the mimics), gain a survival advantage by resembling inedible species (the models).[138][141] In Müllerian mimicry, inedible species, such as of wasps and bees, resemble each other so as to reduce the sampling rate by predators who need to learn that those insects are inedible. Heliconius butterflies, many of which are toxic, form Müllerian complexes, advertising their inedibility.[142] Chemical defense is common among Coleoptera and Lepidoptera, usually being advertised by bright warning colors (aposematism), as in the monarch butterfly. As larvae, they obtain their toxicity by sequestering chemicals from the plants they eat into their own tissues. Some manufacture their own toxins. Predators that eat poisonous butterflies and moths may vomit violently, learning not to eat insects with similar markings; this is the basis of Müllerian mimicry.[143] Some ground beetles of the family Carabidae actively defend themselves, spraying chemicals from their abdomen with great accuracy, to repel predators.[138]
Pollination

Pollination is the process by which pollen is transferred in the reproduction of plants, thereby enabling fertilisation and sexual reproduction.[144] Most flowering plants require an animal to do the transportation. The majority of pollination is by insects.[145] Because insects usually receive benefit for the pollination in the form of energy rich nectar it is a mutualism. The various flower traits, such as bright colors and pheromones that coevolved with their pollinators, have been called pollination syndromes, though around one third of flowers cannot be assigned to a single syndrome.[146]
Parasitism
Many insects are parasitic. The largest group, with over 100,000 species[147] and perhaps over a million,[148] consists of a single clade of parasitoid wasps among the Hymenoptera.[149] These are parasites of other insects, eventually killing their hosts.[147] Some are hyper-parasites, as their hosts are other parasitoid wasps.[147][150] Several groups of insects can be considered as either micropredators or external parasites;[151][152] for example, many hemipteran bugs have piercing and sucking mouthparts, adapted for feeding on plant sap,[153][154] while species in groups such as fleas, lice, and mosquitoes are hematophagous, feeding on the blood of animals.[152]
-
Plant parasite or micropredator: a coreid bug sucking plant sap
-
Human head-lice are directly transmitted obligate ectoparasites.
Relationship to humans
As pests

Many insects are considered pests by humans. These include parasites of people and livestock, such as lice and bed bugs; mosquitoes act as vectors of several diseases. Other pests include insects like termites that damage wooden structures; herbivorous insects such as locusts, aphids, and thrips that destroy agricultural crops, or like wheat weevils damage stored agricultural produce. Farmers have often attempted to control insects with chemical insecticides, but increasingly rely on biological pest control. This uses one organism to reduce the population density of a pest organism; it is a key element of integrated pest management.[156][157] Biological control is favored because insecticides can cause harm to ecosystems far beyond the intended pest targets.[158][159]
In beneficial roles

Pollination of flowering plants by insects including bees, butterflies, flies, and beetles, is economically important.[162] The value of insect pollination of crops and fruit trees was estimated in 2021 to be about $34 billion in the US alone.[163]
Insects produce useful substances such as honey,[164] wax,[165][166] lacquer[167] and silk.[168] Honey bees have been cultured by humans for thousands of years for honey.[169] Beekeeping in pottery vessels began about 9,000 years ago in North Africa.[170] The silkworm has greatly affected human history, as silk-driven trade established relationships between China and the rest of the world.[171][172]
Insects that feed on or parasitise other insects are beneficial to humans if they thereby reduce damage to agriculture and human structures. For example, aphids feed on crops, causing economic loss, but ladybugs feed on aphids, and can be used to control them. Insects account for the vast majority of insect consumption.[173][174][175]
Fly larvae (maggots) were formerly used to treat wounds to prevent or stop gangrene, as they would only consume dead flesh. This treatment is finding modern usage in some hospitals. Insects have gained attention as potential sources of drugs and other medicinal substances.[176] Adult insects, such as crickets and insect larvae of various kinds, are commonly used as fishing bait.[177]
Population declines
At least 66 insect species extinctions have been recorded since 1500, many of them on oceanic islands.[178] Declines in insect abundance have been attributed to human activity in the form of artificial lighting,[179] land use changes such as urbanization or farming,[180][181] pesticide use,[182] and invasive species.[183][184] A 2019 research review suggested that a large proportion of insect species is threatened with extinction in the 21st century,[185] though the details have been disputed.[186] A larger 2020 meta-study, analyzing data from 166 long-term surveys, suggested that populations of terrestrial insects are indeed decreasing rapidly, by about 9% per decade.[187][188]
In research

Insects play important roles in biological research. For example, because of its small size, short generation time and high fecundity, the common fruit fly Drosophila melanogaster is a model organism for studies in the genetics of eukaryotes, including genetic linkage, interactions between genes, chromosomal genetics, development, behavior and evolution. Because genetic systems are well conserved among eukaryotes, understanding basic cellular processes like DNA replication or transcription in fruit flies can help to understand those processes in other eukaryotes, including humans.[189] The genome of D. melanogaster was sequenced in 2000, reflecting the organism's important role in biological research. It was found that 70% of the fly genome is similar to the human genome, supporting the theory of evolution.[190]
As food

Insects are consumed as food in 80% of the world's nations, by people in roughly 3,000 ethnic groups.[192][193] In Africa, locally abundant species of locusts and termites are a common traditional human food source.[194] Some, especially deep-fried cicadas, are considered to be delicacies. Insects have a high protein content for their mass, and some authors suggest their potential as a major source of protein in human nutrition.[195] In most first-world countries, however, entomophagy (the eating of insects), is taboo.[196] They are also recommended by armed forces as a survival food for troops in adversity.[194] Because of the abundance of insects and a worldwide concern of food shortages, the Food and Agriculture Organization of the United Nations considers that people throughout the world may have to eat insects as a food staple. Insects are noted for their nutrients, having a high content of protein, minerals and fats and are already regularly eaten by one-third of the world's population.[197]
In other products
Black soldier fly larvae can provide protein and fats for use in cosmetics.[198] Insect cooking oil, insect butter and fatty alcohols can be made from such insects as the superworm (Zophobas morio).[199] Insect species including the black soldier fly or the housefly in their maggot forms, and beetle larvae such as mealworms, can be processed and used as feed for farmed animals including chicken, fish and pigs.[200] Many species of insects are sold and kept as pets.[201]
In religion and folklore

Scarab beetles held religious and cultural symbolism in ancient Egypt, Greece and some shamanistic Old World cultures. The ancient Chinese regarded cicadas as symbols of rebirth or immortality. In Mesopotamian literature, the epic poem of Gilgamesh has allusions to Odonata that signify the impossibility of immortality. In the case of the 'San' bush-men of the Kalahari, it is the praying mantis that holds much cultural significance including creation and zen-like patience in waiting.[202]
See also
- Entomology
- Ethnoentomology
- Flying and gliding animals
- Insect-borne diseases
Notes
- ↑ The Museum of New Zealand notes that "in everyday conversation", bug "refers to land arthropods with at least six legs, such as insects, spiders, and centipedes".[5] In a chapter on "Bugs That Are Not Insects", entomologist Gilbert Walbauer specifies centipedes, millipedes, arachnids (spiders, daddy longlegs, scorpions, mites, chiggers and ticks) as well as the few terrestrial crustaceans (sowbugs and pillbugs).[6]
References
- ↑ Lewis, Charlton T.; Short, Charles (1879). "insĕco". insĕco. Perseus Digital Library. https://www.perseus.tufts.edu/hopper/text?doc=Perseus%3Atext%3A1999.04.0059%3Aentry%3Dinseco1.
- ↑ Liddell, Henry George; Scott, Robert (1940). "ἔντομος". A Greek-English Lexicon. Perseus Digital Library. https://www.perseus.tufts.edu/hopper/text?doc=Perseus:text:1999.04.0057:entry=e)/ntomos.
- ↑ Harper, Douglas; McCormack, Dan (November 2001). "Online Etymological Dictionary". LogoBee.com. p. 1. http://www.etymonline.com/index.php?term=insect&allowed_in_frame=0.
- ↑ "insect translations". https://www.ezglot.com/words.php?w=insect&l=eng.
- ↑ "What is a bug? Insects, arachnids, and myriapods" at Museum of New Zealand Te Papa Tongarewa website. Accessed 10 March 2022.
- ↑ 6.0 6.1 Waldbauer, Gilbert (1998). The Handy Bug Answer Book. Visible Ink. pp. 1, 5–26. ISBN 978-1-57859-049-0. https://archive.org/details/handybuganswerbo00wald/page/5/mode/2up.
- ↑ Chinery, Michael (1993). "Introduction". Insects of Britain & Northern Europe (3rd ed.). London: HarperCollins. pp. 11–13. ISBN 978-0-00-219918-6.
- ↑ 8.0 8.1 8.2 Gullan & Cranston 2005, pp. 22–48.
- ↑ 9.0 9.1 9.2 Stork, Nigel E. (7 January 2018). "How Many Species of Insects and Other Terrestrial Arthropods Are There on Earth?". Annual Review of Entomology 63 (1): 31–45. doi:10.1146/annurev-ento-020117-043348. PMID 28938083.
- ↑ Erwin, Terry L. (1982). "Tropical forests: their richness in Coleoptera and other arthropod species". The Coleopterists Bulletin 36: 74–75. https://repository.si.edu/bitstream/handle/10088/4383/Classic_papers_in_Foundations.pdf?sequence=1&isAllowed=y. Retrieved 16 September 2018.
- ↑ 11.0 11.1 Gullan & Cranston 2014, p. 8.
- ↑ Crook, Glynis. "Marine insects: small but significant". Deutsche Welle. https://www.dw.com/en/why-are-there-so-few-insects-at-sea/a-44388348.
- ↑ Hågvar, Sigmund (2010). "A review of Fennoscandian arthropods living on and in snow". European Journal of Entomology 107 (3): 281–298. doi:10.14411/eje.2010.037. Bibcode: 2010EJEnt.107..281H. http://www.eje.cz/pdfs/eje/2010/03/01.pdf.
- ↑ "Invertebrates: A Vertebrate Looks at Arthropods". Arizona-Sonora Desert Museum. http://www.desertmuseum.org/books/nhsd_inverts.php.
- ↑ Misof, Bernhard (7 November 2014). "Phylogenomics resolves the timing and pattern of insect evolution". Science 346 (6210): 763–767. doi:10.1126/science.1257570. PMID 25378627. Bibcode: 2014Sci...346..763M. https://www.science.org/. Retrieved 17 October 2009.
- ↑ Kjer, Karl M.; Simon, Chris; Yavorskaya, Margarita; Beutel, Rolf G. (2016). "Progress, pitfalls and parallel universes: a history of insect phylogenetics". Journal of the Royal Society Interface 13 (121): 121. doi:10.1098/rsif.2016.0363. PMID 27558853.
- ↑ Wipfler, Benjamin; Letsch, Harald; Frandsen, Paul B.; Kapli, Paschalia; Mayer, Christoph et al. (February 2019). "Evolutionary history of Polyneoptera and its implications for our understanding of early winged insects". Proceedings of the National Academy of Sciences 116 (8): 3024–3029. doi:10.1073/pnas.1817794116. PMID 30642969. Bibcode: 2019PNAS..116.3024W.
- ↑ Johnson, Kevin P.; Dietrich, Christopher H.; Friedrich, Frank; Beutel, Rolf G.; Wipfler, Benjamin et al. (26 November 2018). "Phylogenomics and the evolution of hemipteroid insects". Proceedings of the National Academy of Sciences 115 (50): 12775–12780. doi:10.1073/pnas.1815820115. ISSN 0027-8424. PMID 30478043. Bibcode: 2018PNAS..11512775J.
- ↑ Kjer, Karl M.; Simon, Chris; Yavorskaya, Margarita; Beutel, Rolf G. (2016). "Progress, pitfalls and parallel universes: a history of insect phylogenetics". Journal of the Royal Society Interface 13 (121): 121. doi:10.1098/rsif.2016.0363. PMID 27558853.
- ↑ 20.0 20.1 Winsor, Mary P. (1976). "The development of Linnaean insect classification". Taxon 25 (1): 57–67. doi:10.2307/1220406. Bibcode: 1976Taxon..25...57W.
- ↑ Leroi, Armand Marie (2014). The Lagoon: How Aristotle Invented Science. Bloomsbury. pp. 111–119. ISBN 978-1-4088-3622-4.
- ↑ Linnaeus, Carl (1758) (in la). Systema naturae per regna tria naturae :secundum classes, ordines, genera, species, cum characteribus, differentiis, synonymis, locis. (10th ed.). Holmiae (Laurentii Salvii). https://www.biodiversitylibrary.org/bibliography/542. Retrieved 22 September 2008.
- ↑ Gould, Stephen Jay (2011). The Lying Stones of Marrakech. Harvard University Press. pp. 130–134. ISBN 978-0-674-06167-5. https://books.google.com/books?id=wApMpVmi-5gC&pg=PA130.
- ↑ De Wit, Hendrik C. D. (1994). Histoire du Développement de la Biologie, Volume III. Presses Polytechniques et Universitaires Romandes. pp. 94–96. ISBN 978-2-88074-264-5.
- ↑ 25.0 25.1 Valentine, James W. (2004). On the Origin of Phyla. University of Chicago Press. pp. 7–8. ISBN 978-0-226-84548-7. https://books.google.com/books?id=DMBkmHm5fe4C&pg=PA8.
- ↑ Haeckel, Ernst (1874) (in de). Anthropogenie oder Entwickelungsgeschichte des menschen. W. Engelmann. p. 202. https://archive.org/details/anthropogenieod05haecgoog.
- ↑ Gullan & Cranston 2005, p. 180.
- ↑ 28.0 28.1 Kendall, David A. (2009). "Classification of Bugs". http://www.kendall-bioresearch.co.uk/class.htm.
- ↑ Blanke, Alexander; Machida, Ryuichiro; Szucsich, Nikolaus Urban; Wilde, Fabian; Misoe, Bernhard (15 October 2014). "Mandibles with two joints evolved much earlier in the history of insects: dicondyly is a synapomorphy of bristletails, silverfish and winged insects". Systematic Entomology (Wiley) 40 (2): 357–364. doi:10.1111/syen.12107.
- ↑ Johnson, K. P.; Yoshizawa, K.; Smith, V. S. (2004). "Multiple origins of parasitism in lice". Proceedings of the Royal Society of London 271 (1550): 1771–1776. doi:10.1098/rspb.2004.2798. PMID 15315891. Bibcode: 2004PBioS.271.1771J.
- ↑ Terry, M. D.; Whiting, M. F. (2005). "Mantophasmatodea and phylogeny of the lower neopterous insects". Cladistics 21 (3): 240–257. doi:10.1111/j.1096-0031.2005.00062.x. https://www.researchgate.net/publication/227604832.
- ↑ Lo, Nathan; Tokuda, Gaku; Watanabe, Hirofumi; Rose, Harley; Slaytor, Michael et al. (2000). "Evidence from multiple gene sequences indicates that termites evolved from wood-feeding cockroaches". Current Biology 10 (13): 801–804. doi:10.1016/S0960-9822(00)00561-3. PMID 10898984. Bibcode: 2000CBio...10..801L.
- ↑ Whiting, M. F. (2002). "Mecoptera is paraphyletic: multiple genes and phylogeny of Mecoptera and Siphonaptera". Zoologica Scripta 31 (1): 93–104. doi:10.1046/j.0300-3256.2001.00095.x.
- ↑ Ross, Andrew (August 2022). "Evolution: The origin of insect wings revisited". Current Biology 32 (15): R851–R853. doi:10.1016/j.cub.2022.06.087. PMID 35944489. Bibcode: 2022CBio...32.R851R.
- ↑ Schachat, Sandra R.; Goldstein, Paul Z.; Desalle, Rob; Bobo, Dean M.; Boyce, C. Kevin; Payne, Jonathan L.; Labandeira, Conrad C. (2 February 2023). "Illusion of flight? Absence, evidence and the age of winged insects". Biological Journal of the Linnean Society 138 (2): 143–168. doi:10.1093/biolinnean/blac137. https://academic.oup.com/biolinnean/article/138/2/143/6936511.
- ↑ Wiegmann, Brian M.; Trautwein, Michelle D.; Winkler, Isaac S.; Barr, Norman B.; Kim, Jung-Wook et al. (14 March 2011). "Episodic radiations in the fly tree of life". Proceedings of the National Academy of Sciences 108 (14): 5690–5695. doi:10.1073/pnas.1012675108. PMID 21402926. Bibcode: 2011PNAS..108.5690W.
- ↑ Carter, J. Stein (29 March 2005). "Coevolution and Pollination". University of Cincinnati. http://biology.clc.uc.edu/courses/bio303/coevolution.htm.
- ↑ "Coevolution and Pollination". University of Cincinnati. http://biology.clc.uc.edu/courses/bio303/coevolution.htm.
- ↑ Taylor, Paul D.; Lewis, David N. (2007). Fossil Invertebrates (repeated ed.). Harvard University Press. p. 160. ISBN 978-0-674-02574-5. https://books.google.com/books?id=7kPwZ2LeSAoC.
- ↑ 40.0 40.1 40.2 Resh & Carde 2009, p. 13.
- ↑ Gullan & Cranston 2005, p. 22–24.
- ↑ Belles, Xavier (14 October 2019). "The innovation of the final moult and the origin of insect metamorphosis". Philosophical Transactions of the Royal Society B: Biological Sciences 374 (1783). doi:10.1098/rstb.2018.0415. PMID 31438822.
- ↑ Asano, Tsunaki; Hashimoto, Kosei; Everroad, R. Craig (2023). "Eco-evolutionary implications for a possible contribution of cuticle hardening system in insect evolution and terrestrialisation". Physiological Entomology 48 (2–3): 55–60. doi:10.1111/phen.12406.
- ↑ Gullan & Cranston 2005, p. 57.
- ↑ Schneiderman, Howard A. (1960). "Discontinuous respiration in insects: role of the spiracles". The Biological Bulletin 119 (3): 494–528. doi:10.2307/1539265. http://www.biolbull.org/cgi/reprint/119/3/494?maxtoshow=&HITS=10&hits=10&RESULTFORMAT=&fulltext=insect+thoracic+spiracle&andorexactfulltext=and&searchid=1&FIRSTINDEX=0&sortspec=relevance&resourcetype=HWCIT. Retrieved 22 May 2009.
- ↑ Dukas, Reuven (1 January 2008). "Evolutionary Biology of Insect Learning". Annual Review of Entomology 53 (1): 145–160. doi:10.1146/annurev.ento.53.103106.093343. PMID 17803459. https://www.annualreviews.org/doi/10.1146/annurev.ento.53.103106.093343.
- ↑ "General Entomology – Digestive and Excritory system". NC state University. http://www.cals.ncsu.edu/course/ent425/library/tutorials/internal_anatomy/digestive.html.
- ↑ Bueno, Odair Correa; Tanaka, Francisco André Ossamu; de Lima Nogueira, Neusa; Fox, Eduardo Gonçalves Paterson; Rossi, Mônica Lanzoni; Solis, Daniel Russ (1 January 2013). "On the morphology of the digestive system of two Monomorium ant species". Journal of Insect Science 13 (1): 70. doi:10.1673/031.013.7001. PMID 24224520.
- ↑ Gullan & Cranston 2005, pp. 70–77.
- ↑ "General Entomology – Digestive and Excretory system". North Carolina State University. http://www.cals.ncsu.edu/course/ent425/library/tutorials/internal_anatomy/digestive.html.
- ↑ Duncan, Carl D. (1939). A Contribution to The Biology of North American Vespine Wasps (1st ed.). Stanford: Stanford University Press. pp. 24–29.
- ↑ Nation 2001, p. 31.
- ↑ Gullan & Cranston 2005, p. 70.
- ↑ Nation 2001, p. 30–31.
- ↑ Nation 2001, p. 32.
- ↑ Gullan & Cranston 2005, pp. 71–72, 78–80.
- ↑ Resh, Carde & 2009, p. 880.
- ↑ Resh & Carde 2009, p. 885.
- ↑ "What Keeps Bugs from Being Bigger?". Argonne National Laboratory. 8 August 2007. https://www1.aps.anl.gov/APS-Science-Highlight/2007/What-Keeps-Bugs-from-Being-Bigger.
- ↑ Gullan & Cranston 2005, pp. 65–68.
- ↑ Chown, S. L.; Nicholson, S. W. (2004). Insect Physiological Ecology. New York: Oxford University Press. ISBN 978-0-19-851549-4.
- ↑ 62.0 62.1 62.2 An Introduction to the Aquatic Insects of North America (4th ed.). Kendall Hunt Publishers. 2007. ISBN 978-0-7575-5049-2.
- ↑ Merritt, R. W.; Cummins, K. W.; Berg, M. B. (2007). An Introduction To The Aquatic Insects Of North America. Kendall Hunt Publishing. ISBN 978-0-7575-4128-5.
- ↑ Gullan & Cranston 2005, pp. 61–65.
- ↑ Meyer, John R. (17 February 2006). "Circulatory System". NC State University: Department of Entomology, NC State University. p. 1. http://www.cals.ncsu.edu/course/ent425/tutorial/circulatory.html.
- ↑ Triplehorn, Charles (2005). Borror and DeLong's introduction to the study of insects. Johnson, Norman F., Borror, Donald J. (7th ed.). Belmont, California: Thompson Brooks/Cole. pp. 27–28. ISBN 978-0-03-096835-8. OCLC 55793895.
- ↑ Gullan & Cranston 2014, pp. 95–124.
- ↑ "Insects". Alien Life Forms. p. 4. http://crazydaz.com/insects.pdf.
- ↑ Gullan & Cranston 2014, pp. 97–103.
- ↑ Warren, Ben; Nowotny, Manuela (11 April 2021). "Bridging the Gap Between Mammal and Insect Ears – A Comparative and Evolutionary View of Sound-Reception". Frontiers in Ecology and Evolution 9. doi:10.3389/fevo.2021.667218. Bibcode: 2021FrEEv...967218W.
- ↑ Briscoe, A. D.; Chittka, L. (2001). "The evolution of color vision in insects". Annual Review of Entomology 46: 471–510. doi:10.1146/annurev.ento.46.1.471. PMID 11112177.
- ↑ Kemppainen, Joni; Scales, Ben; Razban Haghighi, Keivan; Takalo, Jouni; Mansour, Neveen et al. (22 March 2022). "Binocular mirror–symmetric microsaccadic sampling enables Drosophila hyperacute 3D vision". Proceedings of the National Academy of Sciences 119 (12). doi:10.1073/pnas.2109717119. PMID 35298337. Bibcode: 2022PNAS..11909717K.
- ↑ Carraher, Colm; Dalziel, Julie; Jordan, Melissa D.; Christie, David L.; Newcomb, Richard D.; Kralicek, Andrew V. (2015). "Towards an understanding of the structural basis for insect olfaction by odorant receptors". Insect Biochemistry and Molecular Biology 66: 31–41. doi:10.1016/j.ibmb.2015.09.010. PMID 26416146. Bibcode: 2015IBMB...66...31C. https://figshare.com/articles/journal_contribution/22745009.
- ↑ Wajnberg, E.; Acosta-Avalos, D.; Alves, O.C.; de Oliveira, J.F.; Srygley, R.B.; Esquivel, D.M. (2010). "Magnetoreception in eusocial insects: An update". Journal of the Royal Society Interface 7 (Suppl 2): S207–S225. doi:10.1098/rsif.2009.0526.focus. PMID 20106876. Bibcode: 2010JRSI....7focusW.
- ↑ Esquivel, Darci M.S.; Wajnberg, E.; do Nascimento, F.S.; Pinho, M.B.; Lins de Barros, H.G.P.; Eizemberg, R. (2005). "Do Magnetic Storms Change Behavior of the Stingless Bee Guiriçu (Schwarziana quadripunctata)?". Naturwissenschaften 94 (2): 139–142. doi:10.1007/s00114-006-0169-z. PMID 17028885.
- ↑ Lucano, M.J.; Cernicchiaro, G.; Wajnberg, E.; Esquivel, D.M.S. (2005). "Stingless Bee Antennae: A Magnetic Sensory Organ?". BioMetals 19 (3): 295–300. doi:10.1007/s10534-005-0520-4. PMID 16799867.
- ↑ Jacobs, C. G.; Rezende, G. L.; Lamers, G. E.; van der Zee, M. (2013). "The extraembryonic serosa protects the insect egg against desiccation". Proceedings of the Royal Society of London B 280 (1764). doi:10.1098/rspb.2013.1082. PMID 23782888.
- ↑ 78.0 78.1 "insect physiology" McGraw-Hill Encyclopedia of Science and Technology, Ch. 9, p. 233, 2007
- ↑ Gullan & Cranston 2005, pp. 129, 131, 134–135.
- ↑ Gullan & Cranston 2005, pp. 136–137.
- ↑ "Glossary of Lepidopteran and Odonate anatomy". Virginia Department of Conservation and Recreation. 2013. http://www.vararespecies.org/glossary.shtml.
- ↑ 82.0 82.1 Gullan & Cranston 2005, p. 143.
- ↑ Judson, Olivia (14 August 2002). Dr. Tatiana's Sex Advice to All Creation: The Definitive Guide to the Evolutionary Biology of Sex. Macmillan. p. 198. ISBN 978-0-8050-6331-8. https://books.google.com/books?id=HzzdYcX9P54C&pg=PA198.
- ↑ Hughes, William O. H.; Oldroyd, Benjamin P.; Beekman, Madeleine; Ratnieks, Francis L. W. (2008). "Ancestral Monogamy Shows Kin Selection Is Key to the Evolution of Eusociality". Science 320 (5880): 1213–1216. doi:10.1126/science.1156108. PMID 18511689. Bibcode: 2008Sci...320.1213H.
- ↑ Nevo, E.; Coll, M. (2001). "Effect of nitrogen fertilization on Aphis gossypii (Homoptera: Aphididae): variation in size, color, and reproduction". Journal of Economic Entomology 94 (1): 27–32. doi:10.1603/0022-0493-94.1.27. PMID 11233124.
- ↑ Jahn, G. C.; Almazan, L .P.; Pacia, J. (2005). "Effect of nitrogen fertilizer on the intrinsic rate of increase of the rusty plum aphid, Hysteroneura setariae (Thomas) (Homoptera: Aphididae) on rice (Oryza sativa L.)". Environmental Entomology 34 (4): 938–943. doi:10.1603/0046-225X-34.4.938. http://docserver.esa.catchword.org/deliver/cw/pdf/esa/freepdfs/0046225x/v34n4s26.pdf.
- ↑ Gullan & Cranston 2005, p. 142.
- ↑ Ruppert, E. E.; Fox, R. S.; Barnes, R. D. (2004). Invertebrate Zoology (7th ed.). Brooks / Cole. pp. 523–524. ISBN 978-0-03-025982-1. https://archive.org/details/isbn_9780030259821/page/523.
- ↑ Gullan & Cranston 2005, p. 151.
- ↑ Cator, L.J.; Arthur, B.J.; Harrington, L.C.; Hoy, R.R. (2009). "Harmonic convergence in the love songs of the dengue vector mosquito". Science 323 (5917): 1077–1079. doi:10.1126/science.1166541. PMID 19131593. Bibcode: 2009Sci...323.1077C.
- ↑ Gullan & Cranston 2005, pp. 87–94.
- ↑ Pugsley, Chris W. (1983). "Literature review of the New Zealand glowworm Arachnocampa luminosa (Diptera: Keroplatidae) and related cave-dwelling Diptera". New Zealand Entomologist 7 (4): 419–424. doi:10.1080/00779962.1983.9722435. Bibcode: 1983NZEnt...7..419P. http://www.ento.org.nz/nzentomologist/free_issues/NZEnto07_4_1983/Volume%207-4-419-424.pdf.
- ↑ Lloyd, James E. (1984). "Occurrence of Aggressive Mimicry in Fireflies". The Florida Entomologist 67 (3): 368–376. doi:10.2307/3494715.
- ↑ Lloyd, James E.; Gentry, Erin C. (2003). The Encyclopedia of Insects. Academic Press. pp. 115–120. ISBN 978-0-12-586990-4. https://archive.org/details/encyclopediaofin00bada/page/115.
- ↑ "The University of Florida Book of Insect Records". Department of Entomology & Nematology, UF/IFAS. https://entnemdept.ufl.edu/walker/ufbir/index.shtml.
- ↑ Kay, Robert E. (1969). "Acoustic signalling and its possible relationship to assembling and navigation in the moth, Heliothis zea". Journal of Insect Physiology 15 (6): 989–1001. doi:10.1016/0022-1910(69)90139-5. Bibcode: 1969JInsP..15..989K.
- ↑ Spangler, Hayward G. (1988). "Moth hearing, defense, and communication". Annual Review of Entomology 33 (1): 59–81. doi:10.1146/annurev.ento.33.1.59.
- ↑ Hristov, N. I.; Conner, William E. (2005). "Sound strategy: acoustic aposematism in the bat–tiger moth arms race". Naturwissenschaften 92 (4): 164–169. doi:10.1007/s00114-005-0611-7. PMID 15772807. Bibcode: 2005NW.....92..164H.
- ↑ Barber, J. R.; Conner, W. E. (2007). "Acoustic mimicry in a predator–prey interaction". Proceedings of the National Academy of Sciences 104 (22): 9331–9334. doi:10.1073/pnas.0703627104. PMID 17517637. Bibcode: 2007PNAS..104.9331B.
- ↑ Corcoran, Aaron J.; Barber, Jesse R.; Conner, William E. (2009). "Tiger Moth Jams Bat Sonar". Science 325 (5938): 325–327. doi:10.1126/science.1174096. PMID 19608920. Bibcode: 2009Sci...325..325C.
- ↑ Theiss, Joachim (1982). "Generation and radiation of sound by stridulating water insects as exemplified by the corixids". Behavioral Ecology and Sociobiology 10 (3): 225–235. doi:10.1007/BF00299689. Bibcode: 1982BEcoS..10..225T.
- ↑ Virant-Doberlet, M.; Čokl, Andrej (2004). "Vibrational communication in insects". Neotropical Entomology 33 (2): 121–134. doi:10.1590/S1519-566X2004000200001.
- ↑ Bennet-Clark, H. C. (1998). "Size and scale effects as constraints in insect sound communication". Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences 353 (1367): 407–419. doi:10.1098/rstb.1998.0219.
- ↑ Miklas, Nadège; Stritih, Nataša; Čokl, Andrej; Virant-Doberlet, Meta; Renou, Michel (2001). "The Influence of Substrate on Male Responsiveness to the Female Calling Song in Nezara viridula". Journal of Insect Behavior 14 (3): 313–332. doi:10.1023/A:1011115111592. Bibcode: 2001JIBeh..14..313M.
- ↑ DeVries, P. J. (1990). "Enhancement of symbiosis between butterfly caterpillars and ants by vibrational communication". Science 248 (4959): 1104–1106. doi:10.1126/science.248.4959.1104. PMID 17733373. Bibcode: 1990Sci...248.1104D.
- ↑ Nelson, Margaret C.; Fraser, Jean (1980). "Sound production in the cockroach, Gromphadorhina portentosa: evidence for communication by hissing". Behavioral Ecology and Sociobiology 6 (4): 305–314. doi:10.1007/BF00292773. Bibcode: 1980BEcoS...6..305N.
- ↑ Moritz, R. F. A.; Kirchner, W. H.; Crewe, R. M. (1991). "Chemical camouflage of the death's head hawkmoth (Acherontia atropos L.) in honeybee colonies". Naturwissenschaften 78 (4): 179–182. doi:10.1007/BF01136209. Bibcode: 1991NW.....78..179M.
- ↑ 108.0 108.1 Gullan & Cranston 2005, pp. 96–105.
- ↑ Yan, Hua; Liebig, Jürgen (1 April 2021). "Genetic basis of chemical communication in eusocial insects". Genes & Development (Cold Spring Harbor Laboratory Press & The Genetics Society) 35 (7–8): 470–482. doi:10.1101/gad.346965.120. PMID 33861721.
- ↑ Brewer, Gary. "Social insects". North Dakota State University. http://www.ndsu.nodak.edu/entomology/topics/societies.htm.
- ↑ Gullan & Cranston 2005, pp. 309–311.
- ↑ Leadbeater, E.; Chittka, L. (2007). "The dynamics of social learning in an insect model, the bumblebee (Bombus terrestris)". Behavioral Ecology and Sociobiology 61 (11): 1789–1796. doi:10.1007/s00265-007-0412-4. Bibcode: 2007BEcoS..61.1789L.
- ↑ Salt, R. W. (1961). "Principles of Insect Cold-Hardiness". Annual Review of Entomology 6: 55–74. doi:10.1146/annurev.en.06.010161.000415. https://archive.org/details/sim_annual-review-of-entomology_1961_6/page/54.
- ↑ Gullan & Cranston 2005, p. 14.
- ↑ "Social Insects". North Dakota State University. http://www.ndsu.nodak.edu/entomology/topics/societies.htm.
- ↑ Chapman, A. D. (2006). Numbers of living species in Australia and the World. Canberra: Australian Biological Resources Study. ISBN 978-0-642-56850-2. http://www.deh.gov.au/biodiversity/abrs/publications/other/species-numbers/index.html.
- ↑ Smith, D. S. (1965). "Flight muscles of insects". Scientific American 212 (6): 76–88. doi:10.1038/scientificamerican0665-76. PMID 14327957. Bibcode: 1965SciAm.212f..76S.
- ↑ Sane, Sanjay P. (2003). "The aerodynamics of insect flight". Journal of Experimental Biology 206 (23): 4191–4208. doi:10.1242/jeb.00663. PMID 14581590. Bibcode: 2003JExpB.206.4191S. http://www.cs.washington.edu/homes/diorio/MURI2003/Publications/sane_review.pdf.
- ↑ Weis-Fogh, Torkel (1973). "Quick estimates of flight fitness in hovering animals, including novel mechanisms of lift production". Journal of Experimental Biology 59 (1): 169–230. doi:10.1242/jeb.59.1.169. Bibcode: 1973JExpB..59..169W.
- ↑ Bennett, L. (1977). "Clap and fling aerodynamics- an experimental evaluation". Journal of Experimental Biology 69 (1): 261–272. doi:10.1242/jeb.69.1.261. Bibcode: 1977JExpB..69..261B.
- ↑ Jockusch, E. L.; Ober, K. A. (September 2004). "Hypothesis testing in evolutionary developmental biology: a case study from insect wings". Journal of Heredity 95 (5): 382–396. doi:10.1093/jhered/esh064. PMID 15388766.
- ↑ Grimaldi, David A. (2023). The Complete Insect: Anatomy, Physiology, Evolution, and Ecology. Princeton University Press. p. 135. ISBN 978-0-691-24311-5. https://books.google.com/books?id=xGquEAAAQBAJ&dq=insects+wings+notum+pleuron&pg=PA135.
- ↑ Dudley, R. (1998). "Atmospheric oxygen, giant Paleozoic insects and the evolution of aerial locomotor performance". Journal of Experimental Biology 201 (8): 1043–1050. doi:10.1242/jeb.201.8.1043. PMID 9510518. Bibcode: 1998JExpB.201.1043D. http://jeb.biologists.org/content/201/8/1043.full.pdf. Retrieved 8 December 2012.
- ↑ "Chapter 32: Largest Lepidopteran Wing Span | The University of Florida Book of Insect Records | Department of Entomology & Nematology | UF/IFAS". https://entnemdept.ufl.edu/walker/ufbir/chapters/chapter_32.shtml.
- ↑ Yates, Diana. "Birds migrate together at night in dispersed flocks, new study indicates". https://news.illinois.edu/view/6367/206246.
- ↑ Drake, V. A.; Farrow, R. A. (1988). "The Influence of Atmospheric Structure and Motions on Insect Migration". Annual Review of Entomology 33: 183–210. doi:10.1146/annurev.en.33.010188.001151.
- ↑ Biewener, Andrew A. (2003). Animal Locomotion. Oxford University Press. ISBN 978-0-19-850022-3.[page needed]
- ↑ Grabowska, Martyna; Godlewska, Elzbieta; Schmidt, Joachim; Daun-Gruhn, Silvia (2012). "Quadrupedal gaits in hexapod animals – inter-leg coordination in free-walking adult stick insects". Journal of Experimental Biology 215 (24): 4255–4266. doi:10.1242/jeb.073643. PMID 22972892.
- ↑ Ikawa, Terumi; Okabe, Hidehiko; Hoshizaki, Sugihiko; Kamikado, Takahiro; Cheng, Lanna (2004). "Distribution of the oceanic insects Halobates (Hemiptera: Gerridae) off the south coast of Japan". Entomological Science 7 (4): 351–357. doi:10.1111/j.1479-8298.2004.00083.x. https://escholarship.org/uc/item/9r50k828.
- ↑ Mill, P. J.; Pickard, R. S. (1975). "Jet-propulsion in anisopteran dragonfly larvae". Journal of Comparative Physiology A 97 (4): 329–338. doi:10.1007/BF00631969.
- ↑ Linsenmair, K.; Jander, R. (1976). "Das "entspannungsschwimmen" von Velia and Stenus". Naturwissenschaften 50 (6): 231. doi:10.1007/BF00639292. Bibcode: 1963NW.....50..231L. https://nbn-resolving.org/urn:nbn:de:bvb:20-opus-44663.
- ↑ Bush, J. W. M.; Hu, David L. Hu (2006). "Walking on Water: Biolocomotion at the Interface". Annual Review of Fluid Mechanics 38 (1): 339–369. doi:10.1146/annurev.fluid.38.050304.092157. Bibcode: 2006AnRFM..38..339B. http://www.cims.nyu.edu/~dhu/Pubweb/Bush_Hu_06.pdf.
- ↑ Schowalter 2006, pp. 3, 572.
- ↑ Gullan & Cranston 2014, p. 257.
- ↑ Gullan & Cranston 2014, pp. 261–264.
- ↑ Losey, John E.; Vaughan, Mace (2006). "The Economic Value of Ecological Services Provided by Insects". BioScience 56 (4): 311–323(13). doi:10.1641/0006-3568(2006)56[311:TEVOES2.0.CO;2]. http://wordinfo.info/unit/3562?letter=I&spage=3. Retrieved 8 November 2011.
- ↑ Gullan & Cranston 2005, pp. 3, 218–228.
- ↑ 138.0 138.1 138.2 138.3 Evans, Arthur V.; Bellamy, Charles (2000). An Inordinate Fondness for Beetles. University of California Press. p. 31. ISBN 978-0-520-22323-3. https://books.google.com/books?id=ZZ_hfpMo8oAC&pg=PA31.
- ↑ "Photos: Masters of Disguise – Amazing Insect Camouflage". 24 March 2014. http://news.nationalgeographic.com/news/2014/03/140321-insects-fossil-camouflage-mimicry-pictures/.
- ↑ Bedford, Geoffrey O. (1978). "Biology and Ecology of the Phasmatodea". Annual Review of Entomology 23: 125–149. doi:10.1146/annurev.en.23.010178.001013.
- ↑ Ritland, D. B.; Brower, Lincoln P. (1991). "The viceroy butterfly is not a Batesian mimic". Nature 350 (6318): 497–498. doi:10.1038/350497a0. Bibcode: 1991Natur.350..497R. "Viceroys are as unpalatable as monarchs, and significantly more unpalatable than queens from representative Florida populations.".
- ↑ Meyer, A. (2006). "Repeating Patterns of Mimicry". PLOS Biology 4 (10). doi:10.1371/journal.pbio.0040341. PMID 17048984.
- ↑ Kricher, John (1999). "6". A Neotropical Companion. Princeton University Press. pp. 157–158. ISBN 978-0-691-00974-2. https://books.google.com/books?id=Z3pgdvrSmG8C&pg=PA158.
- ↑ Wigglesworth, Vincent Brian. "Insect". Encyclopædia Britannica online. https://www.britannica.com/EBchecked/topic/289001/insect. Retrieved 19 April 2012.
- ↑ "Pollinator Factsheet". United States Forest Service. http://www.fs.fed.us/wildflowers/pollinators/documents/factsheet_pollinator.pdf.
- ↑ Ollerton, Jeff; Alarcón, Ruben; Waser, Nickolas M.; Price, Mary V. et al. (14 February 2009). "A global test of the pollination syndrome hypothesis". Annals of Botany (Oxford University Press) 103 (9): 1471–1480. doi:10.1093/aob/mcp031. PMID 19218577.
- ↑ 147.0 147.1 147.2 Polaszek, Andrew; Vilhemsen, Lars (2023). "Biodiversity of hymenopteran parasitoids". Current Opinion in Insect Science 56. doi:10.1016/j.cois.2023.101026. PMID 36966863. Bibcode: 2023COIS...5601026P.
- ↑ Forbes, Andrew A.; Bagley, Robin K.; Beer, Marc A. et al. (12 July 2018). "Quantifying the unquantifiable: why Hymenoptera, not Coleoptera, is the most speciose animal order". BMC Ecology 18 (1): 21. doi:10.1186/s12898-018-0176-x. PMID 30001194. Bibcode: 2018BMCE...18...21F.
- ↑ Zhang, Qi; Kopylov, Dmitry S.; Rasnitsyn, Alexandr P.; Zheng, Yan; Zhang, Haichun (November 2020). Smith, Andrew. ed. "Burmorussidae, a new family of parasitic wasps (Insecta, Hymenoptera) from mid-Cretaceous Burmese amber". Papers in Palaeontology 6 (4): 593–603. doi:10.1002/spp2.1312. Bibcode: 2020PPal....6..593Z. https://onlinelibrary.wiley.com/doi/10.1002/spp2.1312.
- ↑ Tanaka, S.; Ohsaki, N. (2006). "Behavioral manipulation of host caterpillars by the primary parasitoid wasp Cotesia glomerata (L.) to construct defensive webs against hyperparasitism". Ecological Research 21 (4): 570. doi:10.1007/s11284-006-0153-2. Bibcode: 2006EcoR...21..570T.
- ↑ Poulin, Robert (2011). "The Many Roads to Parasitism: A Tale of Convergence". Advances in Parasitology (Academic Press) 74: 27–28. doi:10.1016/B978-0-12-385897-9.00001-X. ISBN 978-0-12-385897-9. PMID 21295676. https://books.google.com/books?id=9y4AlXka7t0C&pg=PA28.
- ↑ 152.0 152.1 Poulin, Robert; Randhawa, Haseeb S. (February 2015). "Evolution of parasitism along convergent lines: from ecology to genomics". Parasitology 142 (Suppl 1): S6–S15. doi:10.1017/S0031182013001674. PMID 24229807.

- ↑ Gullan, P.J.; Cranston, P.S. (2014). The Insects: An Outline of Entomology, 5th Edition. Wiley. pp. 80–81, 790–. ISBN 978-1-118-84616-2. https://books.google.com/books?id=lF5hBAAAQBAJ&pg=PT790.
- ↑ Labandeira, Conrad C. (May 1998). "Early History of Arthropod and Vascular Plant Associations". Annual Review of Earth and Planetary Sciences 26: 329–377. doi:10.1146/annurev.earth.26.1.329. Bibcode: 1998AREPS..26..329L.
- ↑ Wilson, C. G.; Swincer, D. E.; Walden, K. J. (1982). "The introduction of Trioxys complanatus Quilis (Hymenoptera: Aphidiidae), an internal parasite of the Spotted Alfalfa Aphid, into South Australia". Australian Journal of Entomology 21 (1): 13–27. doi:10.1111/j.1440-6055.1982.tb01758.x.
- ↑ Bale, J. S.; van Lenteren, J. C.; Bigler, F. (27 February 2008). "Biological control and sustainable food production". Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences 363 (1492): 761–776. doi:10.1098/rstb.2007.2182. PMID 17827110.
- ↑ Davidson, E. (2006). Big Fleas Have Little Fleas: How Discoveries of Invertebrate Diseases Are Advancing Modern Science. Tucson, Arizona: University of Arizona Press. ISBN 978-0-8165-2544-7. https://archive.org/details/bigfleashavelitt0000davi.
- ↑ Colborn, T.; vom Saal, F. S.; Soto, A. M. (October 1993). "Developmental effects of endocrine-disrupting chemicals in wildlife and humans". Environmental Health Perspectives 101 (5): 378–384. doi:10.2307/3431890. PMID 8080506.
- ↑ Nakamaru, M.; Iwasab, Y.; Nakanishic, J. (October 2003). "Extinction risk to bird populations caused by DDT exposure". Chemosphere 53 (4): 377–387. doi:10.1016/S0045-6535(03)00010-9. PMID 12946395. Bibcode: 2003Chmsp..53..377N.
- ↑ Yu, Hong-SongExpression error: Unrecognized word "et". (2011). "Evidence of selection at melanin synthesis pathway loci during silkworm domestication". Molecular Biology and Evolution 28 (6): 1785–99. doi:10.1093/molbev/msr002. PMID 21212153.
- ↑ Normile, Dennis (2009). "Sequencing 40 Silkworm Genomes Unravels History of Cultivation". Science 325 (5944): 1058–1059. doi:10.1126/science.325_1058a. PMID 19713499. Bibcode: 2009Sci...325.1058N.
- ↑ Holldobler, Wilson (1994). Journey to the ants: a story of scientific exploration. Cambridge, Massachusetts: Belknap Press. pp. 196–199. ISBN 978-0-674-48525-9. https://archive.org/details/journeytoantss00holl/page/196.
- ↑ Jordan, Alex; Patch, Harland M.; Grozinger, Christina M.; Khanna, Vikas (26 January 2021). "Economic Dependence and Vulnerability of United States Agricultural Sector on Insect-Mediated Pollination Service". Environmental Science & Technology 55 (4): 2243–2253. doi:10.1021/acs.est.0c04786. PMID 33496588. Bibcode: 2021EnST...55.2243J. https://pubs.acs.org/doi/pdf/10.1021/acs.est.0c04786.
- ↑ Crane, E. (1990). "Honey from honeybees and other insects". Ethology Ecology & Evolution 3 (sup1): 100–105. doi:10.1080/03949370.1991.10721919.
- ↑ Sanford, M.T.; Dietz, A. (1976). "The fine structure of the wax gland of the honey bee (Apis mellifera L.).". Apidologie 7 (3): 197–207. doi:10.1051/apido:19760301.
- ↑ "Wax Rendering". Bee Culture. 23 March 2016. https://www.beeculture.com/wax-rendering/.
- ↑ "How Shellac Is Manufactured". The Mail (Adelaide, SA : 1912 – 1954). 18 December 1937. http://nla.gov.au/nla.news-article55073762.
- ↑ Bezzina, Neville. "Silk Production Process". Sense of Nature. Sense of Nature Research. http://www.senature.com/sensemagazine/research-technologies/silk-production-process-go-behind-the-scenes-1701.html.
- ↑ Dams, M.; Dams, L. (21 July 1977). "Spanish Rock Art Depicting Honey Gathering During the Mesolithic". Nature 268 (5617): 228–230. doi:10.1038/268228a0. Bibcode: 1977Natur.268..228D.
- ↑ Roffet-Salque, Mélanie (14 June 2016). "Widespread exploitation of the honeybee by early Neolithic farmers". Nature 534 (7607): 226–227. doi:10.1038/nature18451. PMID 26560301.
- ↑ Vainker, Shelagh (2004). Chinese Silk: A Cultural History. Rutgers University Press. p. 20. ISBN 0-8135-3446-1.
- ↑ Christian, David (2000). "Silk Roads or Steppe Roads? The Silk Roads in World History". Journal of World History 2 (1): 1. doi:10.1353/jwh.2000.0004.
- ↑ Gullan & Cranston 2005, p. 328–348, 400.
- ↑ "Biocontrol Network – Beneficial Insects". Biocontrol Network. http://www.biconet.com/biocontrol.html.
- ↑ Davidson, R. H.; Lyon, William F. (1979). Insect Pests of Farm, Garden, and Orchard. John Wiley & Sons. p. 38. ISBN 978-0-471-86314-4.
- ↑ Dossey, Aaron T. (December 2010). "Insects and their chemical weaponry: New potential for drug discovery". Natural Product Reports 27 (12): 1737–1757. doi:10.1039/c005319h. PMID 20957283.
- ↑ Sherman, Ronald A.; Pechter, Edward A. (1987). "Maggot therapy: a review of the therapeutic applications of fly larvae in human medicine, especially for treating osteomyelitis". Medical and Veterinary Entomology 2 (3): 225–230. doi:10.1111/j.1365-2915.1988.tb00188.x. PMID 2980178.
- ↑ Briggs, John C (October 2017). "Emergence of a sixth mass extinction?". Biological Journal of the Linnean Society 122 (2): 243–248. doi:10.1093/biolinnean/blx063.
- ↑ Owens, Avalon C. S.; Lewis, Sara M. (November 2018). "The impact of artificial light at night on nocturnal insects: A review and synthesis". Ecology and Evolution 8 (22): 11337–11358. doi:10.1002/ece3.4557. PMID 30519447. Bibcode: 2018EcoEv...811337O.
- ↑ Tscharntke, Teja; Klein, Alexandra M.; Kruess, Andreas; Steffan-Dewenter, Ingolf; Thies, Carsten (August 2005). "Landscape perspectives on agricultural intensification and biodiversity and ecosystem service management". Ecology Letters 8 (8): 857–874. doi:10.1111/j.1461-0248.2005.00782.x. Bibcode: 2005EcolL...8..857T.
- ↑ Insect-plant interactions in a crop protection perspective. Academic Press. 19 January 2017. pp. 313–320. ISBN 978-0-12-803324-1.
- ↑ Braak, Nora; Neve, Rebecca; Jones, Andrew K.; Gibbs, Melanie; Breuker, Casper J. (November 2018). "The effects of insecticides on butterflies – A review". Environmental Pollution 242 (Pt A): 507–518. doi:10.1016/j.envpol.2018.06.100. PMID 30005263. Bibcode: 2018EPoll.242..507B. https://radar.brookes.ac.uk/radar/items/fddc88d3-25f2-4274-9321-2f9b3b72047a/1.
- ↑ Wagner, David L.; Van Driesche, Roy G. (January 2010). "Threats Posed to Rare or Endangered Insects by Invasions of Nonnative Species". Annual Review of Entomology 55 (1): 547–568. doi:10.1146/annurev-ento-112408-085516. PMID 19743915.
- ↑ Wilson, E. O.. "Threats to Global Diversity". http://www.globalchange.umich.edu/globalchange2/current/lectures/biodiversity/biodiversity.html.
- ↑ Sánchez-Bayo, Francisco; Wyckhuys, Kris A.G. (April 2019). "Worldwide decline of the entomofauna: A review of its drivers". Biological Conservation 232: 8–27. doi:10.1016/j.biocon.2019.01.020. Bibcode: 2019BCons.232....8S.
- ↑ Saunders, Manu (16 February 2019). "Insectageddon is a great story. But what are the facts?". https://ecologyisnotadirtyword.com/2019/02/16/insectageddon-is-a-great-story-but-what-are-the-facts/.
- ↑ van Klink, Roel (24 April 2020), "Meta-analysis reveals declines in terrestrial but increases in freshwater insect abundances", Science 368 (6489): 417–420, doi:10.1126/science.aax9931, PMID 32327596, Bibcode: 2020Sci...368..417V
- ↑ McGrath, Matt (23 April 2020). "'Insect apocalypse' more complex than thought". BBC News. https://www.bbc.com/news/science-environment-52399373.
- ↑ Pierce, B. A. (2006). Genetics: A Conceptual Approach (2nd ed.). New York: W.H. Freeman and Company. p. 87. ISBN 978-0-7167-8881-2. https://archive.org/details/geneticsconceptu0000unse/page/87.
- ↑ Adams, M. D.; Celniker, S. E.; Holt, R. A.; Evans, C. A.; Gocayne, J. D. et al. (24 March 2000). "The genome sequence of Drosophila melanogaster". Science 287 (5461): 2185–2195. doi:10.1126/science.287.5461.2185. PMID 10731132. Bibcode: 2000Sci...287.2185..
- ↑ Ceurstemont, Sandrine (6 July 2013). "Inevitable insectivores? Not so fast". New Scientist 219 (2924): 35. doi:10.1016/S0262-4079(13)61691-7. https://www.sciencedirect.com/science/article/abs/pii/S0262407913616917. Retrieved 3 December 2021.
- ↑ "Insects could be the key to meeting food needs of growing global population". The Guardian. 31 July 2010. http://www.theguardian.com/environment/2010/aug/01/insects-food-emissions.
- ↑ Ramos-Elorduy, Julieta; Menzel, Peter (1998). Creepy crawly cuisine: the gourmet guide to edible insects. Inner Traditions / Bear & Company. p. 44. ISBN 978-0-89281-747-4. https://books.google.com/books?id=Q7f1LkFz11gC. Retrieved 23 April 2014.
- ↑ 194.0 194.1 "Insects as Food for Humans". https://www.si.edu/spotlight/buginfo/insects-food.
- ↑ Gullan & Cranston 2005, pp. 10–13.
- ↑ Michels, John (1880). John Michels. ed. Science. 1. New York: American Association for the Advance of Science. p. 69. https://books.google.com/books?id=aDkLAAAAYAAJ&pg=PA69.
- ↑ Maierbrugger, Arno (14 May 2013). "UN: Insects are 'food of the future' (video)". Inside Investor. http://investvine.com/un-bugs-are-food-of-the-future-video/.
- ↑ Verheyen, Geert; Ooms, Tom; Vogels, Liesbeth; Vreysen, Steven; Bovy, Ann; Van Miert, Sabine; Meersman, Filip (1 May 2018). "Insects as an Alternative Source for the Production of Fats for Cosmetics". Journal of Cosmetic Science 69 (3): 187–202. PMID 30052193. https://www.researchgate.net/publication/326671736.
- ↑ "From Pest to Pot: Can Insects Feed the World?". 15 August 2016. https://www.nationalgeographic.com/culture/article/sponsor-content-from-pest-pot-can-insects-feed-the-world.
- ↑ "How AgriProtein makes chicken food from maggots". Wired UK. https://www.wired.co.uk/article/food-of-larvae. Retrieved 13 January 2022.
- ↑ Bugs. "Bugs – das Wirbellosenmagazin". https://www.ms-verlag.de/magazine/bugs.
- ↑ Gullan & Cranston 2005, p. 9.
Sources
- Gullan, P. J.; Cranston, P. S. (2005). The Insects: An Outline of Entomology (3rd ed.). Oxford: Blackwell Publishing. ISBN 978-1-4051-1113-3. https://archive.org/details/isbn_9781405111133.
- Gullan, P. J.; Cranston, P. S. (2014). The Insects: An Outline of Entomology (5th ed.). Oxford: Wiley Blackwell. ISBN 978-1-118-84616-2. https://books.google.com/books?id=lF5hBAAAQBAJ.
- Nation, James L. (2001). Insect Physiology and Biochemistry (1st ed.). CRC Press. ISBN 978-0-8493-1181-9. https://books.google.com/books?id=l3v2tOvz1uQC.
- Resh, Vincent H.; Carde, Ring T. (2009). Encyclopedia of Insects (2 ed.). Academic Press. ISBN 978-0-12-374144-8.
- Schowalter, Timothy Duane (2006). Insect Ecology: An Ecosystem Epproach (2nd (illustrated) ed.). Academic Press. ISBN 978-0-12-088772-9. https://books.google.com/books?id=3PD6R-AEvwEC. Retrieved 27 October 2015.
External links
- The Insects — an Outline of Entomology (in both English and Russian)
- Insect species and observations on iNaturalist
- Overview of Orders of Insects
- A Safrinet Manual for Entomology and Arachnology SPC
- Tree of Life Project – Insecta, Insecta Movies
- Fossil Insect Database : Holotypes at the International Palaeoentological Society
- UF Book of Insect Records
- InsectImages.org 24,000 high resolution insect photographs
Wikidata ☰ Q1390 entry
 |









![The giant dragonfly-like insect Meganeura monyi grew to wingspans of 75 cm (2 ft 6 in) in the late Carboniferous, around 300 million years ago.[39]](/wiki/images/thumb/8/81/Meganeura.png/322px-Meganeura.png)




![A parasitoid wasp ovipositing into an aphid[155]](/wiki/images/thumb/0/04/CSIRO_ScienceImage_2357_Spotted_alfalfa_aphid_being_attacked_by_parasitic_wasp.jpg/180px-CSIRO_ScienceImage_2357_Spotted_alfalfa_aphid_being_attacked_by_parasitic_wasp.jpg)




