Biology:Serine C-palmitoyltransferase
| serine C-palmitoyltransferase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| EC number | 2.3.1.50 | ||||||||
| CAS number | 62213-50-7 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
| Serine palmitoyltransferase | |
|---|---|
 | |
| Identifiers | |
| Symbol | SPT1 |
| PDB | 2JG2 |
| UniProt | Q93UV0 |
| Other data | |
| EC number | 2.3.1.50 |
| serine palmitoyltransferase, long chain base subunit 1 | |
|---|---|
| Identifiers | |
| Symbol | SPTLC1 |
| Alt. symbols | HSN1 |
| NCBI gene | 10558 |
| HGNC | 11277 |
| OMIM | 605712 |
| RefSeq | NM_006415 |
| UniProt | O15269 |
| Other data | |
| EC number | 2.3.1.50 |
| Locus | Chr. 9 q22.31 |
| serine palmitoyltransferase, long chain base subunit 2 | |
|---|---|
| Identifiers | |
| Symbol | SPTLC2 |
| NCBI gene | 9517 |
| HGNC | 11278 |
| OMIM | 605713 |
| RefSeq | NM_004863 |
| UniProt | O15270 |
| Other data | |
| EC number | 2.3.1.50 |
| Locus | Chr. 14 q24.3 |
| serine palmitoyltransferase, long chain base subunit 3 | |
|---|---|
| Identifiers | |
| Symbol | SPTLC3 |
| Alt. symbols | C20orf38, SPTLC2L |
| NCBI gene | 55304 |
| HGNC | 16253 |
| OMIM | 611120 |
| RefSeq | NM_018327 |
| UniProt | Q9NUV7 |
| Other data | |
| EC number | 2.3.1.50 |
| Locus | Chr. 20 p12.1 |
In enzymology, a serine C-palmitoyltransferase (EC 2.3.1.50) is an enzyme that catalyzes the chemical reaction:[2][3]
- palmitoyl-CoA + L-serine CoA + 3-dehydro-D-sphinganine + CO2
Thus, the two substrates of this enzyme are palmitoyl-CoA and L-serine, whereas its 3 products are CoA, 3-dehydro-D-sphinganine, and CO2.[4][5] This reaction is a key step in the biosynthesis of sphingosine which is a precursor of many other sphingolipids.[3]
This enzyme participates in sphingolipid metabolism. It employs one cofactor, pyridoxal phosphate.
Nomenclature
This enzyme belongs to the family of transferases, specifically those acyltransferases transferring groups other than aminoacyl groups. The systematic name of this enzyme class is palmitoyl-CoA:L-serine C-palmitoyltransferase (decarboxylating). Other names in common use include:
- serine palmitoyltransferase,
- SPT, 3-oxosphinganine synthetase, and
- acyl-CoA:serine C-2 acyltransferase decarboxylating.
Structure
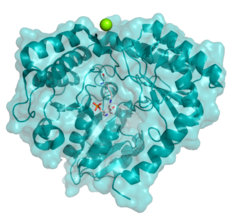
Serine C-palmitoyltransferase is a member of the AOS (a-oxoamine synthase) family of PLP-dependent enzymes, which catalyse the condensation of amino acids and acyl-CoA thioester substrates.[6] The human enzyme is a heterodimer consisting of two monomeric subunits known as long chain base 1 and 2 (LCB1/2) encoded by separate genes.[1] The active site of LCB2 contains lysine and other key catalytic residues that are not present in LCB1, which does not participate in catalysis but is nevertheless required for the synthesis and stability of the enzyme.[7]
As of late 2007, two structures have been solved for this class of enzymes, with PDB accession codes 2JG2 and 2JGT.[1]
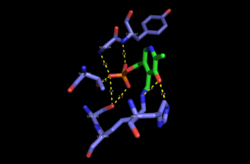
Mechanism
The PLP (pyridoxal 5′-phosphate)-dependent serine C-palmitoyltransferase carries out the first enzymatic step of de novo sphingolipid biosynthesis. The enzyme catalyses a Claisen-like condensation between L-serine and an acyl-CoA thioester (CoASH) substrate (typically C16-palmitoyl) or an acyl-ACP (acyl-carrier protein) thioester substrate, to form 3-ketodihydrosphingosine. Initially PLP cofactor is bound to the active-site lysine via a Schiff base to form the holo-form or internal aldimine of the enzyme. The amine group of L-serine then attacks and displaces the lysine bound to PLP, forming the external aldimine intermediate. Subsequently, deprotonation occurs at the Cα of serine, forming the quinonoid intermediate that attacks the incoming thioester substrate. Following decarboxylation and lysine attack, the product 3-keto-dihydrosphingosine is released and catalytically active PLP is reformed. This condensation reaction forms the sphingoid base or long-chain base found in all subsequent intermediate sphingolipids and complex sphingolipids in the organism.[3]
Isoforms
A variety of different serine C-palmitoyltransferase isoforms exist across different species. Unlike in eukaryotes, where the enzyme is heterodimeric and membrane bound, bacterial enzymes are homodimers and cytoplasmic. Studies of the isoform of the enzyme found in the Gram-negative bacterium Sphingomonas paucimobilis were the first to elucidate the structure of the enzyme, revealing that PLP cofactor is held in place by several active site residues including Lys265 and His159.[8] Specifically, the S. paucimobilis isoform features an active-site arginine residue (Arg378) that plays a key role in stabilizing the carboxy moiety of the PLP-L-serine external aldimine intermediate. Similar arginine residues in enzyme homologues (Arg370, Arg390) play analogous roles.[9] Other homologues, such as in Sphingobacterium multivorum, feature the carboxy moiety bound to serine and methionine residues via water in place of arginine.[10] Certain enzyme homologues, such as in S. multivorum as well as Bdellovibrio stolpii, are found to be associated with the inner cell membrane, thus resembling the eukaryotic enzymes.[11] The B. stolpii homologue also features substrate inhibition by palmitoyl-CoA, a feature shared by the yeast and mammalian homologues.[12][13][14]
Clinical significance
HSAN1 (hereditary sensory and autonomic neuropathy type 1) is a genetic disorder caused by mutations in either one of SPTLC1 or SPTLC2, genes encoding the two heterodimeric subunits of the eukaryotic serine C-palmitoyltransferase enzyme.[15][16][17] These mutations have been shown to alter active site specificity, specifically by enhancing the ability of the enzyme to condense L-alanine with the palmitoyl-CoA substrate.[18] This is consistent with elevated levels of deoxysphingoid bases formed by the condensation of alanine with palmitoyl-CoA observed in HSAN1 patients.[19]
Species distribution
Serine C-palmitoyltransferase is expressed in a large number of species from bacteria to humans. The bacterial enzyme is a water-soluble homodimer[2] whereas in eukaryotes the enzyme is a heterodimer which is anchored to the endoplasmic reticulum.[3] Humans and other mammals express three paralogous subunits SPTLC1, SPTLC2, and SPTLC3. It was originally proposed that the functional human enzyme is a heterodimer between a SPTLC1 subunit and a second subunit which is either SPTLC2 or SPTLC3.[20] However more recent data suggest that the enzyme may exist as a larger complex, possibly an octamer, comprising all three subunits.[21]
References
- ↑ 1.0 1.1 1.2 "The structure of serine palmitoyltransferase; gateway to sphingolipid biosynthesis". Journal of Molecular Biology 370 (5): 870–86. July 2007. doi:10.1016/j.jmb.2007.04.086. PMID 17559874.
- ↑ 2.0 2.1 "Bacterial serine palmitoyltransferase: a water-soluble homodimeric prototype of the eukaryotic enzyme". Biochimica et Biophysica Acta (BBA) - Proteins and Proteomics 1647 (1–2): 116–20. April 2003. doi:10.1016/S1570-9639(03)00074-8. PMID 12686119.
- ↑ 3.0 3.1 3.2 3.3 "Serine palmitoyltransferase, a key enzyme of sphingolipid metabolism". Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids 1632 (1–3): 16–30. June 2003. doi:10.1016/S1388-1981(03)00059-3. PMID 12782147.
- ↑ "Biosynthesis of sphingolipid bases. 3. Isolation and characterization of ketonic intermediates in the synthesis of sphingosine and dihydrosphingosine by cell-free extracts of Hansenula ciferri". The Journal of Biological Chemistry 244 (2): 491–6. January 1969. doi:10.1016/S0021-9258(18)94455-8. PMID 4388074. http://www.jbc.org/cgi/content/abstract/244/2/491.
- ↑ "Biosynthesis of dihydrosphingosine in vitro". Hoppe-Seyler's Zeitschrift für Physiologische Chemie 349 (5): 664–70. May 1968. doi:10.1515/bchm2.1968.349.1.664. PMID 4386961.
- ↑ "Pyridoxal phosphate enzymes: mechanistic, structural, and evolutionary considerations". Annual Review of Biochemistry 73: 383–415. 2004. doi:10.1146/annurev.biochem.73.011303.074021. PMID 15189147.
- ↑ "The topology of the Lcb1p subunit of yeast serine palmitoyltransferase". The Journal of Biological Chemistry 279 (51): 53707–16. December 2004. doi:10.1074/jbc.M410014200. PMID 15485854.
- ↑ "Multifunctional role of His159in the catalytic reaction of serine palmitoyltransferase". The Journal of Biological Chemistry 284 (23): 15487–95. June 2009. doi:10.1074/jbc.M808916200. PMID 19346561.
- ↑ "Role of a conserved arginine residue during catalysis in serine palmitoyltransferase". FEBS Letters 585 (12): 1729–34. June 2011. doi:10.1016/j.febslet.2011.04.013. PMID 21514297. https://www.pure.ed.ac.uk/ws/files/11045134/Role_of_a_conserved_arginine_residue_during_catalysis_in_serine_palmitoyltransferase.pdf.
- ↑ "Structural insights into the enzymatic mechanism of serine palmitoyltransferase from Sphingobacterium multivorum". Journal of Biochemistry 146 (4): 549–62. October 2009. doi:10.1093/jb/mvp100. PMID 19564159.
- ↑ "Molecular characterization of membrane-associated soluble serine palmitoyltransferases from Sphingobacterium multivorum and Bdellovibrio stolpii". Journal of Bacteriology 189 (15): 5749–61. August 2007. doi:10.1128/JB.00194-07. PMID 17557831.
- ↑ "Tsc3p is an 80-amino acid protein associated with serine palmitoyltransferase and required for optimal enzyme activity". The Journal of Biological Chemistry 275 (11): 7597–603. March 2000. doi:10.1074/jbc.275.11.7597. PMID 10713067.
- ↑ "Purification of the serine palmitoyltransferase complex responsible for sphingoid base synthesis by using affinity peptide chromatography techniques". The Journal of Biological Chemistry 275 (12): 8409–15. March 2000. doi:10.1074/jbc.275.12.8409. PMID 10722674.
- ↑ "Relevance of platinum-sensitivity status in relapsed/refractory extensive-stage small-cell lung cancer in the modern era: a patient-level analysis of southwest oncology group trials". Journal of Thoracic Oncology 10 (1): 110–5. January 2015. doi:10.1097/JTO.0000000000000385. PMID 25490004.
- ↑ "SPTLC1 is mutated in hereditary sensory neuropathy, type 1". Nature Genetics 27 (3): 261–2. March 2001. doi:10.1038/85817. PMID 11242106.
- ↑ "Mutations in the yeast LCB1 and LCB2 genes, including those corresponding to the hereditary sensory neuropathy type I mutations, dominantly inactivate serine palmitoyltransferase". The Journal of Biological Chemistry 277 (12): 10194–200. March 2002. doi:10.1074/jbc.M107873200. PMID 11781309.
- ↑ "Mutations in the SPTLC2 subunit of serine palmitoyltransferase cause hereditary sensory and autonomic neuropathy type I". American Journal of Human Genetics 87 (4): 513–22. October 2010. doi:10.1016/j.ajhg.2010.09.010. PMID 20920666.
- ↑ "A disease-causing mutation in the active site of serine palmitoyltransferase causes catalytic promiscuity". The Journal of Biological Chemistry 285 (30): 22846–52. July 2010. doi:10.1074/jbc.M110.122259. PMID 20504773.
- ↑ "Hereditary sensory neuropathy type 1 is caused by the accumulation of two neurotoxic sphingolipids". The Journal of Biological Chemistry 285 (15): 11178–87. April 2010. doi:10.1074/jbc.M109.092973. PMID 20097765.
- ↑ "Cloning and initial characterization of a new subunit for mammalian serine-palmitoyltransferase". The Journal of Biological Chemistry 281 (49): 37275–81. December 2006. doi:10.1074/jbc.M608066200. PMID 17023427.
- ↑ "Is the mammalian serine palmitoyltransferase a high-molecular-mass complex?". The Biochemical Journal 405 (1): 157–64. July 2007. doi:10.1042/BJ20070025. PMID 17331073.
 |
