Chemistry:Bis(2-chloroethyl)sulfide
| |
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
1-Chloro-2-[(2-chloroethyl)sulfanyl]ethane | |
| Other names
Bis(2-chloroethyl) sulfide
HD Iprit Schwefel-LOST Lost Sulfur mustard Senfgas Yellow cross liquid Yperite Distilled mustard Mustard T- mixture 1,1'-thiobis[2-chloroethane] Dichlorodiethyl sulfide | |
| Identifiers | |
3D model (JSmol)
|
|
| 1733595 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
| 324535 | |
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C4H8Cl2S | |
| Molar mass | 159.07 g·mol−1 |
| Appearance | Colorless if pure. Normally ranges from pale yellow to dark brown. Slight garlic or horseradish type odor.[1] |
| Density | 1.27 g/mL, liquid |
| Melting point | 14.4 °C (57.9 °F; 287.5 K) |
| Boiling point | 217 °C (423 °F; 490 K) begins to decompose at 217 °C (423 °F) and boils at 218 °C (424 °F) |
| 7.6 mg/L at 20°C[2] | |
| Solubility | Alcohols, ethers, hydrocarbons, lipids, THF |
| Hazards | |
| Main hazards | Flammable, toxic, vesicant, carcinogenic, mutagenic |
| Safety data sheet | External MSDS |
| GHS pictograms |  
|
| GHS Signal word | Danger |
| HH300Script error: No such module "Preview warning".Category:GHS errors, HH310Script error: No such module "Preview warning".Category:GHS errors, HH315Script error: No such module "Preview warning".Category:GHS errors, HH319Script error: No such module "Preview warning".Category:GHS errors, HH330Script error: No such module "Preview warning".Category:GHS errors, HH335Script error: No such module "Preview warning".Category:GHS errors | |
| PP260Script error: No such module "Preview warning".Category:GHS errors, PP261Script error: No such module "Preview warning".Category:GHS errors, PP262Script error: No such module "Preview warning".Category:GHS errors, PP264Script error: No such module "Preview warning".Category:GHS errors, PP270Script error: No such module "Preview warning".Category:GHS errors, PP271Script error: No such module "Preview warning".Category:GHS errors, PP280Script error: No such module "Preview warning".Category:GHS errors, PP284Script error: No such module "Preview warning".Category:GHS errors, PP301+P310Script error: No such module "Preview warning".Category:GHS errors, PP302+P350Script error: No such module "Preview warning".Category:GHS errors, PP302+P352Script error: No such module "Preview warning".Category:GHS errors, PP304+P340Script error: No such module "Preview warning".Category:GHS errors, PP305+P351+P338Script error: No such module "Preview warning".Category:GHS errors, PP310Script error: No such module "Preview warning".Category:GHS errors, PP312Script error: No such module "Preview warning".Category:GHS errors, PP320Script error: No such module "Preview warning".Category:GHS errors, PP321Script error: No such module "Preview warning".Category:GHS errors, PP322Script error: No such module "Preview warning".Category:GHS errors, PP330Script error: No such module "Preview warning".Category:GHS errors, PP332+P313Script error: No such module "Preview warning".Category:GHS errors, PP337+P313Script error: No such module "Preview warning".Category:GHS errors, PP361Script error: No such module "Preview warning".Category:GHS errors, PP362Script error: No such module "Preview warning".Category:GHS errors, PP363Script error: No such module "Preview warning".Category:GHS errors, PP403+P233Script error: No such module "Preview warning".Category:GHS errors | |
| NFPA 704 (fire diamond) | |
| Flash point | 105 °C (221 °F; 378 K) |
| Related compounds | |
Related compounds
|
Nitrogen mustard, Bis(chloroethyl) ether, Chloromethyl methyl sulfide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
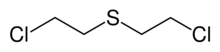
Bis(2-chloroethyl)sulfide is the organosulfur compound with the formula (ClCH
2CH
2)
2S. It is a prominent member of a family of cytotoxic and blister agents known as mustard agents. Sometimes referred to as mustard gas, the term is technically incorrect: bis(2-chloroethyl)sulfide is a liquid at room temperature. In warfare it was dispersed in the form of a fine mist of liquid droplets.[4][5]
Synthesis
Bis(2-chloroethyl)sulfide has been prepared in a variety of ways. In the Depretz method, sulfur dichloride is treated with ethylene:
- SCl
2 + 2 C
2H
4 → (ClC
2H
4)
2S
In the Levinstein process, disulfur dichloride is used instead:[6][failed verification][7]
- S
2Cl
2 + 2 C
2H
4 → (ClC
2H
4)
2S + 1⁄8 S8
In the Meyer method, thiodiglycol is produced from chloroethanol and potassium sulfide, and the resulting diol is then treated with phosphorus trichloride:[8][9]
- 3 (HOC
2H
4)
2S + 2 PCl
3 → 3 (ClC
2H
4)
2S + 2 P(OH)
3
In the Meyer–Clarke method, concentrated hydrochloric acid (HCl) is used instead of PCl3:
- (HOC
2H
4)
2S + 2 HCl → (ClC
2H
4)
2S + 2 H
2O
Thionyl chloride and phosgene, the latter of which is also a choking agent, have also been used as chlorinating agents. These compounds have the added advantage in that if they are used in excess, they remain as impurities in the finished product and can therefore produce additional mechanisms of toxicity.
Reactions
The idealized combustion of mustard gas in oxygen produces hydrochloric acid and sulfuric acid, in addition to carbon dioxide and water:
- (ClC
2H
4)
2S + 7 O
2 → 4 CO
2 + 2 H
2O + 2 HCl + H
2SO
4
Bis(2-chloroethyl)sulfide reacts with sodium hydroxide, giving divinyl sulfide:
- (ClC
2H
4)
2S + 2 NaOH → (CH
2=CH)
2S + 2 H
2O + 2 NaCl
Sodium ethoxide acts similarly.
Safety
Upon skin contact or inhalation, bis(2-chloroethyl)sulfide is a nonspecific toxin. It is a strong alkylating agent, which affects DNA, RNA, and proteins.[10]
See also
References
- ↑ FM 3–8 Chemical Reference handbook, US Army, 1967
- ↑ Mustard agents: description, physical and chemical properties, mechanism of action, symptoms, antidotes and methods of treatment. Organisation for the Prohibition of Chemical Weapons. Accessed June 8, 2010.
- ↑ "Pubchem". https://pubchem.ncbi.nlm.nih.gov/compound/10461.
- ↑ "Sulfur Mustard: Blister Agent" (in en). https://www.cdc.gov/niosh/ershdb/emergencyresponsecard_29750008.html.
- ↑ Ghabili, Kamyar; Agutter, Paul S.; Ghanei, Mostafa; Ansarin, Khalil; Panahi, Yunes; Shoja, Mohammadali M. (2011). "Sulfur mustard toxicity: History, chemistry, pharmacokinetics, and pharmacodynamics". Critical Reviews in Toxicology 41 (5): 384–403. doi:10.3109/10408444.2010.541224. PMID 21329486.
- ↑ Stewart, Charles D. (2006). Weapons of mass casualties and terrorism response handbook. Boston: Jones and Bartlett. p. 47. ISBN 0-7637-2425-4. https://books.google.com/books?id=7ZnXZfwWwgcC&q=levinstein+inventor+mustard&pg=PA46.
- ↑ "Chemical Weapons Production and Storage". Federation of American Scientists. https://fas.org/programs/ssp/bio/chemweapons/production.html.
- ↑ E. M. Faber and G. E. Miller (1932). "β-Thiodiglycol". Organic Syntheses 12: 68. doi:10.15227/orgsyn.012.0068.
- ↑ Institute of Medicine (1993). Chapter 5: Chemistry of Sulfur Mustard and Lewisite. The National Academies Press. ISBN 0-309-04832-X. http://books.nap.edu/openbook.php?record_id=2058&page=71.
- ↑ Kehe, Kai; Szinicz, Ladislaus (2005). "Medical aspects of sulphur mustard poisoning". Toxicology 214 (3): 198–209. doi:10.1016/j.tox.2005.06.014. PMID 16084004.
 |


