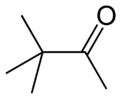
Chemistry:Pinacolone
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
3,3-Dimethylbutan-2-one | |||
| Other names
t-Butyl methyl ketone
1,1,1-Trimethylacetone | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 1209331 | |||
| ChEBI | |||
| ChemSpider | |||
| EC Number |
| ||
| MeSH | Pinacolone | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1224 | ||
| |||
| |||
| Properties | |||
| C6H12O | |||
| Molar mass | 100.161 g·mol−1 | ||
| Appearance | Colorless liquid | ||
| Density | 0.801 g cm−3 | ||
| Melting point | −52[1] °C (−62 °F; 221 K) | ||
| Boiling point | 103 to 106 °C (217 to 223 °F; 376 to 379 K) | ||
| -69.86·10−6 cm3/mol | |||
| Hazards | |||
| Safety data sheet | External MSDS | ||
| GHS pictograms |  
| ||
| GHS Signal word | Danger | ||
| H225, H302, H315, H319, H332, H335, H412 | |||
| P210, P233, P240, P241, P242, P243, P261, P264, P270, P271, P273, P280, P301+312, P302+352, P303+361+353, P304+312, P304+340, P305+351+338, P312, P321, P330, P332+313, P337+313, P362, P370+378 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 5 °C (41 °F; 278 K) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Pinacolone (3,3-dimethyl-2-butanone) is an important ketone in organic chemistry. It is a colorless liquid and has a slight peppermint- or camphor- odor. It is a precursor to triazolylpinacolone in the synthesis of the fungicide triadimefon and in synthesis of the herbicide metribuzin. The molecule is an unsymmetrical ketone. The α-methyl group can participate in condensation reactions. The carbonyl group can undergo the usual reactions (hydrogenation, reductive amination, etc.). It is a Schedule 3 compound under the Chemical Weapons Convention 1993, due to being related to pinacolyl alcohol, which is used in the production of soman.[2] It is also a controlled export in Australia Group member states.[3]
Preparation
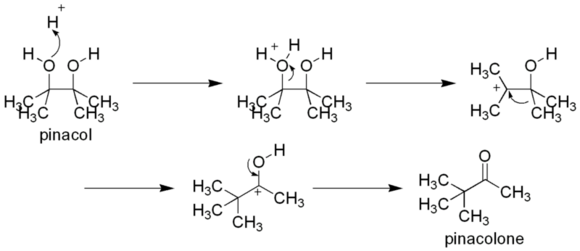
Most famously, at least in the classroom, pinacolone arises by the pinacol rearrangement, which occurs by protonation of pinacol (2,3-dimethylbutane-2,3-diol).[4]
Industrially pinacolone is made by the hydrolysis of 4,4,5-trimethyl-1,3-dioxane, which is the product of isoprene and formaldehyde via the Prins reaction. It also is generated by ketonization of pivalic acid and acetic acid or acetone over metal oxide catalysts. 3-Methylbutanal is a starting material for 2,3-dimethyl-2-butene, which in turn is converted to pinacolone. Pinacolone can also be produced from 2-methy-2-butanol when reacted with C5 alcohols.[5]
Drug Uses
Pinacolone is produced in large amounts for use in fungicides, herbicides, and pesticides.
- retrosynthetic analysis of vibunazole showed that it was derived from pinacolone.
- It is also used to prepare pinacidil, as well as naminidil.
- Stiripentol
- Tribuzone
- Pivaloylacetonitrile is used in the synthesis of Doramapimod.
- Triadimefon
- Diclobutrazole
- Paclobutrazol
- Valconazole
- Diethylstilbestrol pinacolone [18922-13-9].[6]
- Some kind of Bisphenol A derivative also U.S. Patent 4,599,463
- Thiofanox
See also
References
- ↑ "Pinacolone | C6H12O | ChemSpider". http://www.chemspider.com/Chemical-Structure.6176.html?rid=b009474b-a0f1-4132-a843-a9137c137689.
- ↑ Handbook of chemical and biological warfare agents (2nd ed.). CRC Press. 24 August 2007. ISBN 9780849314346.
- ↑ "Export Control List: Chemical Weapons Precursors". australiagroup.net. http://www.australiagroup.net/en/precursors.html.
- ↑ G. A. Hill and E. W. Flosdorf (1941). "Pinacolone". Organic Syntheses. http://www.orgsyn.org/demo.aspx?prep=CV1P0462.; Collective Volume, 1, pp. 462
- ↑ Siegel, H; Eggersdorfer (2012). Ketones. 5. 20. doi:10.1002/14356007.a15_077. ISBN 9783527306732.
- ↑ Oda, T; Sato, Y; Kodama, M; Kaneko, M (July 1993). "Inhibition of DNA topoisomerase I activity by diethylstilbestrol and its analogues.". Biological & Pharmaceutical Bulletin 16 (7): 708–10. doi:10.1248/bpb.16.708. PMID 8401407.
 |