Chemistry:Sulfur hexafluoride
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Sulfur hexafluoride
| |||
| Systematic IUPAC name
Hexafluoro-λ6-sulfane[1] | |||
| Other names
Elagas
Esaflon | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| EC Number |
| ||
| 2752 | |||
| KEGG | |||
| MeSH | Sulfur+hexafluoride | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1080 | ||
| |||
| |||
| Properties | |||
| SF 6 | |||
| Molar mass | 146.05 g·mol−1 | ||
| Appearance | Colorless gas | ||
| Odor | odorless[2] | ||
| Density | 6.17 g/L | ||
| Melting point | −50.7 °C (−59.3 °F; 222.5 K)[6] (at or above 2,26 bar air pressure - at normal air pressure it sublimes instead) | ||
| Boiling point | −68.25 °C (−90.85 °F; 204.90 K)[7] (sublimes) | ||
| Critical point (T, P) | 45.51±0.1 °C, 3.749±0.01 MPa[3] | ||
| 0.003% (25 °C)[2] | |||
| Solubility | slightly soluble in water, very soluble in ethanol, hexane, benzene | ||
| Vapor pressure | 2.9 MPa (at 21.1 °C) | ||
| −44.0×10−6 cm3/mol | |||
| Thermal conductivity |
| ||
| Viscosity | 15.23 μPa·s[5] | ||
| Structure | |||
| Orthorhombic, oP28 | |||
| Oh | |||
| Orthogonal hexagonal | |||
| Octahedral | |||
| 0 D | |||
| Thermochemistry | |||
Heat capacity (C)
|
0.097 kJ/(mol·K) (constant pressure) | ||
Std molar
entropy (S |
292 J·mol−1·K−1[8] | ||
Std enthalpy of
formation (ΔfH⦵298) |
−1209 kJ·mol−1[8] | ||
| Pharmacology | |||
| 1=ATC code }} | V08DA05 (WHO) | ||
| License data | |||
| Hazards | |||
| Safety data sheet | External MSDS | ||
| GHS pictograms | 
| ||
| GHS Signal word | Warning | ||
| Template:HPhrases | |||
| Template:PPhrases | |||
| NFPA 704 (fire diamond) | |||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 1000 ppm (6000 mg/m3)[2] | ||
REL (Recommended)
|
TWA 1000 ppm (6000 mg/m3)[2] | ||
IDLH (Immediate danger)
|
N.D.[2] | ||
| Related compounds | |||
Related sulfur fluorides
|
Disulfur decafluoride | ||
Related compounds
|
Selenium hexafluoride Sulfuryl fluoride | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
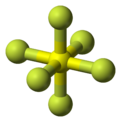
Sulfur hexafluoride or sulphur hexafluoride (British spelling) is an inorganic compound with the formula SF6. It is a colorless, odorless, non-flammable, and non-toxic gas. SF6 has an octahedral geometry, consisting of six fluorine atoms attached to a central sulfur atom. It is a hypervalent molecule. Typical for a nonpolar gas, SF6 is poorly soluble in water but quite soluble in nonpolar organic solvents. It has a density of 6.12 g/L at sea level conditions, considerably higher than the density of air (1.225 g/L). It is generally stored and transported as a liquefied compressed gas.[10]
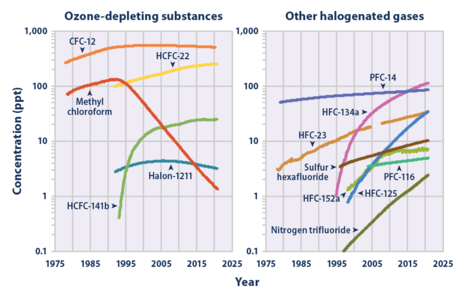
SF6 has 23,500 times greater global warming potential (GWP) than CO
2 as a greenhouse gas (over a 100-year time-frame) but exists in relatively minor concentrations in the atmosphere. Its concentration in Earth's troposphere reached 12.06 parts per trillion (ppt) in February 2025, rising at 0.4 ppt/year.[11] The increase since 1980 is driven in large part by the expanding electric power sector, including fugitive emissions from banks of SF6 gas contained in its medium- and high-voltage switchgear. Uses in magnesium, aluminium, and electronics manufacturing also hastened atmospheric growth.[12]
Synthesis and reactions
Sulfur hexafluoride on Earth exists primarily as a synthetic industrial gas, but has also been found to occur naturally.[13]
SF6 can be prepared from the elements through exposure of S8 to F2. This was the method used by the discoverers Henri Moissan and Paul Lebeau in 1901. Some other sulfur fluorides are cogenerated, but these are removed by heating the mixture to disproportionate any S2F10 (which is highly toxic) and then scrubbing the product with NaOH to destroy remaining SF4.[clarification needed]
Alternatively, using bromine, sulfur hexafluoride can be synthesized from SF4 and CoF3 at lower temperatures (e.g. 100 °C), as follows:[14]
There are few chemical reactions for SF6. A main contribution to the inertness of SF6 is the steric hindrance of the sulfur atom, whereas its heavier group 16 counterparts, such as SeF6 are more reactive than SF6 as a result of less steric hindrance.[15] It does not react with molten sodium below its boiling point,[16] but reacts exothermically with lithium. However, alkali metals react with SF6 in liquid ammonia to form the corresponding sulfides and fluorides[17]:
As a result of its inertness, SF6 has an atmospheric lifetime of around 3200 years, and no significant environmental sinks other than the ocean.[18]
Applications
By 2000, the electrical power industry is estimated to use about 80% of the sulfur hexafluoride produced, mostly as a gaseous dielectric medium.[19] Other main uses as of 2015 included a silicon etchant for semiconductor manufacturing, and an inert gas for the casting of magnesium.[20]
Dielectric medium
SF6 is used in the electrical industry as a gaseous dielectric medium for high-voltage sulfur hexafluoride circuit breakers, switchgear, and other electrical equipment, often replacing oil-filled circuit breakers (OCBs) that can contain harmful polychlorinated biphenyls (PCBs). SF6 gas under pressure is used as an insulator in gas insulated switchgear (GIS) because it has a much higher dielectric strength than air or dry nitrogen. The high dielectric strength is a result of the gas's high electronegativity and density. This property makes it possible to significantly reduce the size of electrical gear. This makes GIS more suitable for certain purposes such as indoor placement, as opposed to air-insulated electrical gear, which takes up considerably more room.
Gas-insulated electrical gear is also more resistant to the effects of pollution and climate, as well as being more reliable in long-term operation because of its controlled operating environment. Exposure to an arc chemically breaks down SF6 though most of the decomposition products tend to quickly re-form SF6, a process termed "self-healing".[21] Arcing or corona can produce disulfur decafluoride (S2F10), a highly toxic gas, with toxicity similar to phosgene. S2F10 was considered a potential chemical warfare agent in World War II because it does not produce lacrimation or skin irritation, thus providing little warning of exposure.
SF6 is also commonly encountered as a high voltage dielectric in the high voltage supplies of particle accelerators, such as Van de Graaff generators and Pelletrons and high voltage transmission electron microscopes.
Alternatives to SF6 as a dielectric gas include several fluoroketones.[22][23] Compact GIS technology that combines vacuum switching with clean air insulation has been introduced for a subset of applications up to 420 kV.[24]
Medical use
SF6 is used to provide a tamponade or plug of a retinal hole in retinal detachment repair operations[25] in the form of a gas bubble. It is inert in the vitreous chamber.[26] The bubble initially doubles its volume in 36 hours due to oxygen and nitrogen entering it, before being absorbed in the blood in 10–14 days.[27]
SF6 is used as a contrast agent for ultrasound imaging. Sulfur hexafluoride microbubbles are administered in solution through injection into a peripheral vein. These microbubbles enhance the visibility of blood vessels to ultrasound. This application has been used to examine the vascularity of tumours.[28] It remains visible in the blood for 3 to 8 minutes, and is exhaled by the lungs.[29]
Tracer compound
Sulfur hexafluoride was the tracer gas used in the first roadway air dispersion model calibration; this research program was sponsored by the U.S. Environmental Protection Agency and conducted in Sunnyvale, California on U.S. Highway 101.[30] Gaseous SF6 is used as a tracer gas in short-term experiments of ventilation efficiency in buildings and indoor enclosures, and for determining infiltration rates. Two major factors recommend its use: its concentration can be measured with satisfactory accuracy at very low concentrations, and the Earth's atmosphere has a negligible concentration of SF6.
Sulfur hexafluoride was used as a non-toxic test gas in an experiment at St John's Wood tube station in London, United Kingdom on 25 March 2007.[31] The gas was released throughout the station, and monitored as it drifted around. The purpose of the experiment, which had been announced earlier in March by the Secretary of State for Transport Douglas Alexander, was to investigate how toxic gas might spread throughout London Underground stations and buildings during a terrorist attack.
Sulfur hexafluoride is also routinely used as a tracer gas in laboratory fume hood containment testing. The gas is used in the final stage of ASHRAE 110 fume hood qualification. A plume of gas is generated inside of the fume hood and a battery of tests are performed while a gas analyzer arranged outside of the hood samples for SF6 to verify the containment properties of the fume hood.
It has been used successfully as a transient tracer in oceanography to study diapycnal mixing and air-sea gas exchange.[32] The concentration of sulfur hexafluoride in seawater (typically on the order of femtomoles per kilogram[33]) has been classified by the international oceanography community as a "level one" measurement, denoting the highest priority data for observing ocean changes.[34]
Other uses
- The magnesium industry uses SF6 as an inert "cover gas" to prevent oxidation during casting,[35] and other processes including smelting.[36] Once the largest user, consumption has declined greatly with capture and recycling.[12]
- Insulated glazing windows have used it as a filler to improve their thermal and acoustic insulation performance.[37][38]
- SF6 plasma is used in the semiconductor industry as an etchant in processes such as deep reactive-ion etching. A small fraction of the SF6 breaks down in the plasma into sulfur and fluorine, with the fluorine ions performing a chemical reaction with silicon.[39]
- Tires filled with it take longer to deflate from diffusion through rubber due to the larger molecule size.[37]
- Nike likewise used it to obtain a patent and to fill the cushion bags in all of their "Air"-branded shoes from 1992 to 2006.[40] 277 tons were used during the peak in 1997.[37]
- The United States Navy's Mark 50 torpedo closed Rankine-cycle propulsion system is powered by sulfur hexafluoride in an exothermic reaction with solid lithium.[41]
- Waveguides in high-power microwave systems are pressurized with it. The gas electrically insulates the waveguide, preventing internal arcing.
- Electrostatic loudspeakers have used it because of its high dielectric strength and high molecular weight.[42]
- Disulfur decafluoride, a chemical weapon, is produced with it as a feedstock.
- For entertainment purposes, when breathed, SF6 causes the voice to become significantly deeper, due to its density being so much higher than air. This phenomenon is related to the more well-known effect of breathing low-density helium, which causes someone's voice to become much higher. Both of these effects should only be attempted with caution as these gases displace oxygen that the lungs are attempting to extract from the air. Sulfur hexafluoride is also mildly anesthetic.[43][44]
- For science demonstrations / magic as "invisible water" since a light foil boat can be floated in a tank, as will an air-filled balloon.
- It is used for benchmark and calibration measurements in Associative and Dissociative Electron Attachment (DEA) experiments[45][46]
Greenhouse gas
-
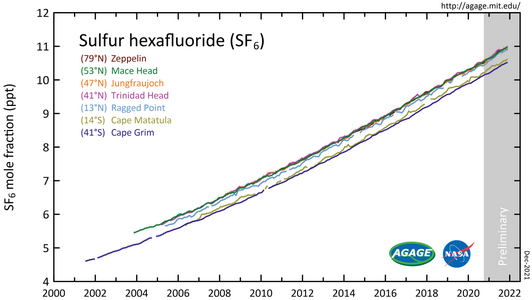
Sulfur hexafluoride (SF6) measured by the Advanced Global Atmospheric Gases Experiment (AGAGE) in the lower atmosphere (troposphere) at stations around the world. Abundances are given as pollution free monthly mean mole fractions in parts-per-trillion.
-
Abundance and growth rate of SF6 in Earth's troposphere (1978-2018).[12]
-
Atmospheric concentration of SF6 vs. similar man-made gases (right graph). Note the log scale.
According to the Intergovernmental Panel on Climate Change, SF6 is the most potent greenhouse gas. Its global warming potential of 23,900 times that of CO2 when compared over a 100-year period.[47] Sulfur hexafluoride is inert in the troposphere and stratosphere and is extremely long-lived, with an estimated atmospheric lifetime of 800–3,200 years.[48]
Measurements of SF6 show that its global average mixing ratio has increased from a steady base of about 54 parts per quadrillion[13] prior to industrialization, to over 12 parts per trillion (ppt) as of February 2025, and is increasing by about 0.4 ppt (3.5%) per year.[11][49] Average global SF6 concentrations increased by about 7% per year during the 1980s and 1990s, mostly as the result of its use in magnesium production, and by electrical utilities and electronics manufacturers. Given the small amounts of SF6 released compared to carbon dioxide, its overall individual contribution to global warming is estimated to be less than 0.2%,[50] however the collective contribution of it and similar man-made halogenated gases has reached about 10% as of 2020.[51] Alternatives are being tested.[52][53]
In Europe, SF6 falls under the F-Gas directive which bans or controls its use for several applications.[54] Since 1 January 2006, SF6 is banned as a tracer gas and in all applications except high-voltage switchgear.[55] It was reported in 2013 that a three-year effort by the United States Department of Energy to identify and fix leaks at its laboratories in the United States such as the Princeton Plasma Physics Laboratory, where the gas is used as a high voltage insulator, had been productive, cutting annual leaks by 1,030 kilograms (2,280 pounds). This was done by comparing purchases with inventory, assuming the difference was leaked, then locating and fixing the leaks.[56]
Physiological effects and precautions
Sulfur hexafluoride is a nontoxic gas, but by displacing oxygen in the lungs, it also carries the risk of asphyxia if too much is inhaled.[57] Since it is more dense than air, a substantial quantity of gas, when released, will settle in low-lying areas and present a significant risk of asphyxiation if the area is entered. That is particularly relevant to its use as an insulator in electrical equipment since workers may be in trenches or pits below equipment containing SF6.[58]
File:Sulfur Hexafluoride (SF6) Surprise.webm As with all gases, the density of SF6 affects the resonance frequencies of the vocal tract, thus changing drastically the vocal sound qualities, or timbre, of those who inhale it. It does not affect the vibrations of the vocal folds. The density of sulfur hexafluoride is relatively high at room temperature and pressure due to the gas's large molar mass. Unlike helium, which has a molar mass of about 4 g/mol and pitches the voice up, SF6 has a molar mass of about 146 g/mol, and the speed of sound through the gas is about 134 m/s at room temperature, pitching the voice down. For comparison, the molar mass of air, which is about 80% nitrogen and 20% oxygen, is approximately 30 g/mol which leads to a speed of sound of 343 m/s.[59]
Sulfur hexafluoride has an anesthetic potency slightly lower than nitrous oxide;[60] it is classified as a mild anesthetic.[61]
See also
- Selenium hexafluoride
- Tellurium hexafluoride
- Uranium hexafluoride
- Hypervalent molecule
- Halocarbon—another group of major greenhouse gases
- Trifluoromethylsulfur pentafluoride, a similar gas
References
- ↑ "Sulfur Hexafluoride - PubChem Public Chemical Database". PubChem. National Center for Biotechnology Information. https://pubchem.ncbi.nlm.nih.gov/compound/17358.
- ↑ 2.0 2.1 2.2 2.3 2.4 NIOSH Pocket Guide to Chemical Hazards. "#0576". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0576.html.
- ↑ Horstmann, Sven; Fischer, Kai; Gmehling, Jürgen (2002). "Measurement and calculation of critical points for binary and ternary mixtures". AIChE Journal 48 (10): 2350–2356. doi:10.1002/aic.690481024. ISSN 0001-1541. Bibcode: 2002AIChE..48.2350H.
- ↑ Assael, M. J.; Koini, I. A.; Antoniadis, K. D.; Huber, M. L.; Abdulagatov, I. M.; Perkins, R. A. (2012). "Reference Correlation of the Thermal Conductivity of Sulfur Hexafluoride from the Triple Point to 1000 K and up to 150 MPa". Journal of Physical and Chemical Reference Data 41 (2): 023104–023104–9. doi:10.1063/1.4708620. ISSN 0047-2689. Bibcode: 2012JPCRD..41b3104A.
- ↑ Assael, M. J.; Kalyva, A. E.; Monogenidou, S. A.; Huber, M. L.; Perkins, R. A.; Friend, D. G.; May, E. F. (2018). "Reference Values and Reference Correlations for the Thermal Conductivity and Viscosity of Fluids". Journal of Physical and Chemical Reference Data 47 (2): 021501. doi:10.1063/1.5036625. ISSN 0047-2689. PMID 30996494. Bibcode: 2018JPCRD..47b1501A.
- ↑ https://encyclopedia.airliquide.com/sulfur-hexafluoride#properties
- ↑ https://encyclopedia.airliquide.com/sulfur-hexafluoride#properties
- ↑ 8.0 8.1 Zumdahl, Steven S. (2009). Chemical Principles 6th Ed. Houghton Mifflin Company. p. A23. ISBN 978-0-618-94690-7.
- ↑ GHS: Record of Schwefelhexafluorid in the GESTIS Substance Database of the Institute for Occupational Safety and Health, accessed on 2021-12-13.
- ↑ Niemeyer, Lutz (1998), Christophorou, Loucas G.; Olthoff, James K., eds., "SF6 Recycling in Electric Power Equipment" (in en), Gaseous Dielectrics VIII (Boston, MA: Springer US): pp. 431–442, doi:10.1007/978-1-4615-4899-7_58, ISBN 978-1-4615-4899-7
- ↑ 11.0 11.1 "Trends in Atmospheric Sulpher Hexaflouride". US National Oceanic and Atmospheric Administration. https://www.esrl.noaa.gov/gmd/ccgg/trends_sf6/.
- ↑ 12.0 12.1 12.2 Simmonds, P. G., Rigby, M., Manning, A. J., Park, S., Stanley, K. M., McCulloch, A., Henne, S., Graziosi, F., Maione, M., and 19 others (2020) "The increasing atmospheric burden of the greenhouse gas sulfur hexafluoride (SF6)". Atmos. Chem. Phys., 20: 7271–7290. doi:10.5194/acp-20-7271-2020. 50px Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
- ↑ 13.0 13.1 Busenberg, E. and Plummer, N. (2000). "Dating young groundwater with sulfur hexafluoride: Natural and anthropogenic sources of sulfur hexafluoride". Water Resources Research (American Geophysical Union) 36 (10): 3011–3030. doi:10.1029/2000WR900151. Bibcode: 2000WRR....36.3011B. https://pubs.er.usgs.gov/publication/70022588#:~:text=Sulfur%20hexafluoride%20(SF6)%20is,during%20the%20past%2040%20years.
- ↑ Winter, R. W.; Pugh, J. R.; Cook, P. W. (January 9–14, 2011). "SF5Cl, SF4 and SF6: Their Bromine−facilitated Production & a New Preparation Method for SF5Br".
- ↑ Duward Shriver; Peter Atkins (2010). Inorganic Chemistry. W. H. Freeman. p. 409. ISBN 978-1-4292-5255-3.
- ↑ Raj, Gurdeep (2010). Advanced Inorganic Chemistry: Volume II (12th ed.). GOEL Publishing House. p. 160. https://books.google.com/books?id=qxRhA3MZg6AC. Extract of page 160
- ↑ Holger Lars Deubner, Florian Kraus: The Decomposition Products of Sulfur Hexafluoride (SF6) with Metals Dissolved in Liquid Ammonia. In: Inorganics. Band 5, Nr. 4, 13. Oktober 2017, S. 68, doi:10.3390/inorganics5040068 (mdpi.com).
- ↑ Stöven, T.; Tanhua, T.; Hoppema, M.; Bullister, J. L. (2015-09-18). "Perspectives of transient tracer applications and limiting cases" (in en). Ocean Science 11 (5): 699–718. doi:10.5194/os-11-699-2015. ISSN 1812-0792. Bibcode: 2015OcSci..11..699S. https://os.copernicus.org/articles/11/699/2015/.
- ↑ Constantine T. Dervos; Panayota Vassilou (2000). "Sulfur Hexafluoride: Global Environmental Effects and Toxic Byproduct Formation". Journal of the Air & Waste Management Association (Taylor and Francis) 50 (1): 137–141. doi:10.1080/10473289.2000.10463996. PMID 10680375. Bibcode: 2000JAWMA..50..137D.
- ↑ Deborah Ottinger; Mollie Averyt; Deborah Harris (2015). "US consumption and supplies of sulphur hexafluoride reported under the greenhouse gas reporting program". Journal of Integrative Environmental Sciences (Taylor and Francis) 12 (sup1): 5–16. doi:10.1080/1943815X.2015.1092452.
- ↑ Jakob, Fredi; Perjanik, Nicholas, Sulfur Hexafluoride, A Unique Dielectric, Analytical ChemTech International, Inc., http://www.weidmann-solutions.cn/zhenduan/sulfur_hexaflouride.pdf
- ↑ "Archived copy". http://cired.net/publications/cired2015/papers/CIRED2015_0587_final.pdf.
- ↑ Kieffel, Yannick; Biquez, Francois (1 June 2015). "SF6 alternative development for high voltage switchgears". 2015 IEEE Electrical Insulation Conference (EIC). pp. 379–383. doi:10.1109/ICACACT.2014.7223577. ISBN 978-1-4799-7352-1.
- ↑ "Sustainable switchgear technology for a CO2 neutral future". Siemens Energy. 2020-08-31. https://www.siemens-energy.com/global/en/offerings/power-transmission/innovation/blue-high-voltage-products.html.
- ↑ Daniel A. Brinton; C. P. Wilkinson (2009). Retinal detachment: principles and practice. Oxford University Press. p. 183. ISBN 978-0-19-971621-0.
- ↑ Gholam A. Peyman, M.D., Stephen A. Meffert, M.D., Mandi D. Conway (2007). Vitreoretinal Surgical Techniques. Informa Healthcare. p. 157. ISBN 978-1-84184-626-2.
- ↑ Hilton, G. F.; Das, T.; Majji, A. B.; Jalali, S. (1996). "Pneumatic retinopexy: Principles and practice". Indian Journal of Ophthalmology 44 (3): 131–143. PMID 9018990.
- ↑ "Dynamic contrast-enhanced ultrasonography (DCE-US) with quantification of tumor perfusion: a new diagnostic tool to evaluate the early effects of antiangiogenic treatment". Eur Radiol 17 (Suppl. 6): F89–F98. December 2007. doi:10.1007/s10406-007-0233-6. PMID 18376462.
- ↑ "SonoVue, INN-sulphur hexafluoride - Annex I - Summary of Product Characteristics". https://www.ema.europa.eu/documents/product-information/sonovue-epar-product-information_en.pdf.
- ↑ C Michael Hogan (September 10, 2011). "Air pollution line source". Encyclopedia of Earth. http://www.eoearth.org/article/Air_pollution_line_source?topic=49506.
- ↑ "'Poison gas' test on Underground". BBC News. 25 March 2007. https://news.bbc.co.uk/2/hi/uk_news/england/london/6492501.stm.
- ↑ Fine, Rana A. (2010-12-15). "Observations of CFCs and SF6 as Ocean Tracers". Annual Review of Marine Science 3 (1): 173–195. doi:10.1146/annurev.marine.010908.163933. ISSN 1941-1405. PMID 21329203. https://www.annualreviews.org/doi/10.1146/annurev.marine.010908.163933.
- ↑ J.L. Bullister and T. Tanhua, 2010. "Sampling and measurement of chlorofluorocarbons and sulfur hexafluoride in seawater". IOCCP Report No. 14. http://www.go-ship.org/HydroMan.html
- ↑ "GO-SHIP Data Requirements". http://www.go-ship.org/DatReq.html.
- ↑ Scott C. Bartos (February 2002). "Update on EPA's manesium industry partnership for climate protection". US Environmental Protection Agency. http://www.epa.gov/magnesium-sf6/documents/tms_paper.pdf.
- ↑ Ayres, John (2000). "Canadian Perspective on SF6 Management from Magnesium Industry". Environment Canada. https://www.epa.gov/sites/default/files/2016-02/documents/conf00_ayres_paper.pdf.
- ↑ 37.0 37.1 37.2 J. Harnisch and W. Schwarz (2003-02-04). "Final report on the costs and the impact on emissions of potential regulatory framework for reducing emissions of hydrofluorocarbons, perfluorocarbons and sulphur hexafluoride". Ecofys GmbH. http://www.oekorecherche.de/sites/default/files/publikationen/ecofys_oekorecherchestudy.pdf.
- ↑ Hopkins, Carl (2007). Sound insulation - Google Books. Elsevier / Butterworth-Heinemann. pp. 504–506. ISBN 978-0-7506-6526-1. https://books.google.com/books?id=bqUxCGJd0owC&q=hexafluoride+insulating+glass&pg=PA504.
- ↑ "Dry Etching of Silicon Materials in SF6 Based Plasmas". Journal of the Electrochemical Society. September 1987. http://www.eng.auburn.edu/~tzengy/Publications/Plasma%20Lab%20Publications/Dry%20etching%20of%20silicon%20materials%20in%20SF6%20based%20plasma.pdf.
- ↑ Stanley Holmes (September 24, 2006). "Nike Goes For The Green". Bloomberg Business Week Magazine. http://www.businessweek.com/stories/2006-09-24/nike-goes-for-the-green.
- ↑ Hughes, T.G.; Smith, R.B.; Kiely, D.H. (1983). "Stored Chemical Energy Propulsion System for Underwater Applications". Journal of Energy 7 (2): 128–133. doi:10.2514/3.62644. Bibcode: 1983JEner...7..128H.
- ↑ Dick Olsher (October 26, 2009). "Advances in loudspeaker technology - A 50 year prospective". The Absolute Sound. http://www.theabsolutesound.com/articles/advances-in-loudspeaker-technology-a-50-year-perspective-tas-196/.
- ↑ Edmond I Eger MD (September 10, 1968). "Anesthetic Potencies of Sulfur Hexafluoride, Carbon Tetrafluoride, Chloroform and Ethrane in Dogs: Correlation with the Hydrate and Lipid Theories of Anesthetic Action". Anesthesiology: The Journal of the American Society of Anesthesiologists (Anesthesiology - The Journal of the American Society of Anesthesiologists, Inc) 30 (2): 127–134. http://anesthesiology.pubs.asahq.org/article.aspx?articleid=1964594.
- ↑ WTOL (2015-01-27). Sound Like Darth Vader with Sulfur Hexafluoride. YouTube. Imagination Station.
- ↑ Braun, M; Marienfeld, S; Ruf, M-W; Hotop, H (2009-05-26). "High-resolution electron attachment to the molecules CCl4and SF6over extended energy ranges with the (EX)LPA method" (in en). Journal of Physics B: Atomic, Molecular and Optical Physics 42 (12). doi:10.1088/0953-4075/42/12/125202. ISSN 0953-4075. Bibcode: 2009JPhB...42l5202B. https://iopscience.iop.org/article/10.1088/0953-4075/42/12/125202.
- ↑ Fenzlaff, Marita; Gerhard, Rolf; Illenberger, Eugen (1988-01-01). "Associative and dissociative electron attachment by SF6 and SF5Cl". The Journal of Chemical Physics 88 (1): 149–155. doi:10.1063/1.454646. ISSN 0021-9606. Bibcode: 1988JChPh..88..149F. https://aip.scitation.org/doi/10.1063/1.454646.
- ↑ "2.10.2 Direct Global Warming Potentials". Intergovernmental Panel on Climate Change. 2007. http://www.ipcc.ch/publications_and_data/ar4/wg1/en/ch2s2-10-2.html.
- ↑ A. R. Ravishankara, S. Solomon, A. A. Turnipseed, R. F. Warren; Solomon; Turnipseed; Warren (8 January 1993). "Atmospheric Lifetimes of Long-Lived Halogenated Species". Science 259 (5092): 194–199. doi:10.1126/science.259.5092.194. PMID 17790983. Bibcode: 1993Sci...259..194R. https://www.science.org/doi/abs/10.1126/science.259.5092.194. Retrieved 22 February 2013.
- ↑ "Sulfur hexafluoride (SF6) data from hourly in situ samples analyzed on a gas chromatograph located at Cape Matatulu (SMO)". aftp.cmdl.noaa.gov. July 7, 2020. ftp://aftp.cmdl.noaa.gov/data/hats/sf6/insituGCs/CATS/monthly/smo_SF6_MM.dat. (To view documents see Help:FTP)
- ↑ "SF6 Sulfur Hexafluoride". PowerPlantCCS Blog. 19 March 2011. http://powerplantccs.com/blog/2011/03/sf6-sulfur-hexafluoride.html.
- ↑ Butler J. and Montzka S. (2020). "The NOAA Annual Greenhouse Gas Index (AGGI)". NOAA Global Monitoring Laboratory/Earth System Research Laboratories. https://www.esrl.noaa.gov/gmd/aggi/aggi.html.
- ↑ "g3, the SF6-free solution in practice | Think Grid". 18 February 2019. https://think-grid.org/g3-sf6-free-solution-practice.
- ↑ Mohamed Rabie; Christian M. Franck (2018). "Assessment of Eco-friendly Gases for Electrical Insulation to Replace the Most Potent Industrial Greenhouse Gas SF6". Environmental Science & Technology (American Chemical Society) 52 (2): 369–380. doi:10.1021/acs.est.7b03465. PMID 29236468. Bibcode: 2018EnST...52..369R.
- ↑ David Nikel (2020-01-15). "Sulfur hexafluoride: The truths and myths of this greenhouse gas". phys.org. https://phys.org/news/2020-01-sulfur-hexafluoride-truths-myths-greenhouse.html.
- ↑ "Climate: MEPs give F-gas bill a 'green boost'". www.euractiv.com (EurActiv.com). 13 October 2005. http://www.euractiv.com/sustainability/climate-meps-give-gas-bill-green-boost/article-145749.
- ↑ Michael Wines (June 13, 2013). "Department of Energy's Crusade Against Leaks of a Potent Greenhouse Gas Yields Results". The New York Times. https://www.nytimes.com/2013/06/14/us/department-of-energys-crusade-against-leaks-of-a-potent-greenhouse-gas-yields-results.html.
- ↑ "Sulfur Hexafluoride". Hazardous Substances Data Bank. U.S. National Library of Medicine. http://toxnet.nlm.nih.gov/cgi-bin/sis/search/a?dbs+hsdb:@term+@DOCNO+825.
- ↑ "Guide to the safe use of SF6 in gas". UNIPEDE/EURELECTRIC. http://www.eurelectric.org/Download/Download.aspx?DocumentID=2136.
- ↑ "Physics in Speech". University of New South Wales. http://www.animations.physics.unsw.edu.au/jw/speech.html.
- ↑ Adriani, John (1962). The Chemistry and Physics of Anesthesia (2nd ed.). Illinois: Thomas Books. p. 319. ISBN 978-0-398-00011-0.
- ↑ Weaver, Raymond H.; Virtue, Robert W. (1 November 1952). "The mild anesthetic properties of sulfur hexafluoride" (in en). Anesthesiology 13 (6): 605–607. doi:10.1097/00000542-195211000-00006. PMID 12986223. https://anesthesiology.pubs.asahq.org/article.aspx?articleid=1971324.
Further reading
- "Sulfur hexafluoride". Air Liquide Gas Encyclopedia. http://encyclopedia.airliquide.com/Encyclopedia.asp?GasID=34.
- Christophorou, Loucas G., ed (1991). Gaseous Dielectrics VI. Plenum Press. ISBN 978-0-306-43894-3.
- Holleman, A. F.; Wiberg, E. (2001). Inorganic Chemistry. San Diego: Academic Press. ISBN 0-12-352651-5.
- Khalifa, Mohammad (1990). High-Voltage Engineering: Theory and Practice. New York: Marcel Dekker. ISBN 978-0-8247-8128-6. OCLC 20595838.
- Maller, V. N.; Naidu, M. S. (1981). Advantages in High Voltage Insulation and Arc Interruption in SF6 and Vacuum. Oxford; New York: Pergamon Press. ISBN 978-0-08-024726-7. OCLC 7866855.
- SF6 Reduction Partnership for Electric Power Systems
- Matt McGrath (September 13, 2019). "Climate change: Electrical industry's 'dirty secret' boosts warming". BBC News. https://www.bbc.com/news/science-environment-49567197.
External links
- Fluoride and compounds fact sheet— National Pollutant Inventory
- High GWP Gases and Climate Change from the U.S. EPA website
- International Conference on SF6 and the Environment (related archive)
- CDC - NIOSH Pocket Guide to Chemical Hazards
 |



![Abundance and growth rate of SF6 in Earth's troposphere (1978-2018).[12]](/wiki/images/thumb/7/7b/AGAGE_sulfur_hexafluroride_growth.png/404px-AGAGE_sulfur_hexafluroride_growth.png)