Chemistry:1,1,2-Trichloro-1,2,2-trifluoroethane
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1,1,2-Trichloro-1,2,2-trifluoroethane | |||
| Other names
Arklone P
CFC-113 Freon 113 Frigen 113 TR Freon TF Valclene 1,1,2-trichlorotrifluoroethane | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEMBL | |||
| ChemSpider | |||
PubChem CID
|
|||
| UNII | |||
| |||
| |||
| Properties | |||
| CClF 2CCl 2F | |||
| Molar mass | 187.37 g·mol−1 | ||
| Appearance | Colorless liquid | ||
| Odor | like carbon tetrachloride[1] | ||
| Density | 1.56 g/mL | ||
| Melting point | −35 °C (−31 °F; 238 K) | ||
| Boiling point | 47.7 °C (117.9 °F; 320.8 K) | ||
| 170 mg/L | |||
| Vapor pressure | 285 mmHg (20 °C)[1] | ||
| Thermal conductivity | 0.0729 W m−1 K−1 (300 K)[2] | ||
| Hazards | |||
| Lethal dose or concentration (LD, LC): | |||
LCLo (lowest published)
|
250,000 ppm (mouse, 1.5 hr) 87,000 (rat, 6 hr)[3] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 1000 ppm (7600 mg/m3)[1] | ||
REL (Recommended)
|
TWA 1000 ppm (7600 mg/m3) ST 1250 ppm (9500 mg/m3)[1] | ||
IDLH (Immediate danger)
|
2000 ppm[1] | ||
| Hazards | |||
| Safety data sheet | https://datasheets.scbt.com/sc-251541.pdf | ||
| GHS pictograms |  
| ||
| GHS Signal word | Warning | ||
| NFPA 704 (fire diamond) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
1,1,2-Trichloro-1,2,2-trifluoroethane, also called trichlorotrifluoroethane or CFC-113, is a chlorofluorocarbon. It has the formula Cl
2FC–CClF
2. This colorless, volatile liquid is a versatile solvent.[5]
Production
CFC-113 can be prepared from hexachloroethane and hydrofluoric acid:[6]
This reaction may require catalysts such as antimony, chromium, iron and alumina at high temperatures.[7]
Another synthesis method uses HF on tetrachloroethylene instead.[8]
Atmospheric reactions
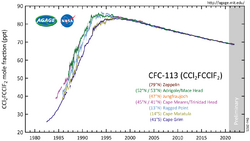
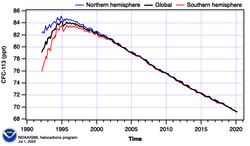
CFC-113 is a very unreactive chlorofluorocarbon. It remains in the atmosphere about 90 years,[9] sufficiently long that it will cycle out of the troposphere and into the stratosphere. In the stratosphere, CFC-113 can be broken up by ultraviolet radiation (where sunlight in the 190-225 nm (UV) range), generating chlorine radicals (Cl•), which initiate degradation of ozone requiring only a few minutes:[10][11]
- CClF
2CCl
2F → C
2F
3Cl
2 + Cl• - Cl• + O
3 → ClO• + O
2
This reaction is followed by:
- ClO• + O → Cl• + O
2
The process regenerates Cl• to destroy more O
3. The Cl• will destroy an average of 100,000 O
3 molecules during its atmospheric lifetime of 1–2 years. In some parts of the world, these reactions have significantly thinned the Earth's natural stratospheric ozone layer that shields the biosphere against solar UV radiation; increased UV levels at the surface can cause skin cancer or even blindness.[12]
Uses
CFC-113 was one of the most heavily produced CFCs. In 1989, an estimated 250,000 tons were produced.[5] It has been used as a cleaning agent for electrical and electronic components.[12] CFC-113’s low flammability and low toxicity made it ideal for use as a cleaner for delicate electrical equipment, fabrics, and metals. It would not harm the product it was cleaning, ignite with a spark or react with other chemicals.[13] It was used as a dry-cleaning solvent, introduced by Du Pont in March 1961 as "Valclene"[14] and was also marketed as the "solvent of the future" by Imperial Chemical Industries in the 1970s under the tradename Arklone. Others from this series were Perklone (Tetrachloroethylene), Triklone (Trichloroethylene), Methoklone (Dichloromethane) and Genklene (1,1,1-Trichloroethane).[15][16]
CFC-113 is one of the three most popular CFCs, along with CFC-11 and CFC-12.[17]
CFC-113 in laboratory analytics has been replaced by other solvents.[18]
Reduction of CFC-113 with zinc gives chlorotrifluoroethylene:[5]
- CFCl
2–CClF
2 + Zn → CClF=CF
2 + ZnCl
2
Dangers
Aside from its immense environmental impacts, Freon 113, like most chlorofluoroalkanes, forms phosgene gas when exposed to a naked flame.[19]
See also
References
- ↑ 1.0 1.1 1.2 1.3 1.4 NIOSH Pocket Guide to Chemical Hazards. "#0632". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0632.html.
- ↑ Touloukian, Y.S., Liley, P.E., and Saxena, S.C. Thermophysical properties of matter - the TPRC data series. Volume 3. Thermal conductivity - nonmetallic liquids and gases. Data book. 1970.
- ↑ "1,1,2-Trichloro-1,2,2-trifluoroethane". Immediately Dangerous to Life and Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/idlh/76131.html.
- ↑ Safety Data Sheet fishersci.com
- ↑ 5.0 5.1 5.2 Siegemund, Günter; Schwertfeger, Werner; Feiring, Andrew; Smart, Bruce; Behr, Fred; Vogel, Herward; McKusick, Blaine (2002). "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a11_349.
- ↑ Social and Economic Implications of Controlling the Use of Chlorofluorocarbons in the EEC pitt.edu
- ↑ Kirk-Othmer Encyclopedia of Chemical Technology. 4th ed. Volumes 1: New York, NY. John Wiley and Sons, 1991-Present., p. V11 507 (1994)
- ↑ Robert D. Ashford: Ashford's Dictionary of Industrial Chemicals, p. 1131. 2nd Edition. Wavelength Publications, 2001
- ↑ "Global Change 2: Climate Change". University of Michigan. January 4, 2006. http://www.globalchange.umich.edu/globalchange2/current/lectures/climate_change/the_science.htm.
- ↑ Molina, Mario J. (1996). "Role of chlorine in the stratospheric chemistry". Pure and Applied Chemistry 68 (9): 1749–1756. doi:10.1351/pac199668091749.
- ↑ "Guides | SEDAC". http://www.ciesin.org/TG?OZ/prodcfcs.html.
- ↑ 12.0 12.1 "Chlorofluorocarbons". Columbia Encyclopedia. 2008. http://www.encyclopedia.com/doc/1E1-chlorofl.html. Retrieved 2008-05-28.
- ↑ "Guides | SEDAC". https://sedac.ciesin.columbia.edu/guides?OZ/prodcfcs.html.
- ↑ Coin-Op 1961-04: Vol 2 Iss 4 P. 61
- ↑ Industrial Finishing and Surface Coatings. (1973). UK: Wheatland journals, Limited.
- ↑ Morrison, R. D., Murphy, B. L. (2013). Chlorinated Solvents: A Forensic Evaluation. Royal Society of Chemistry
- ↑ Zumdahl, Steven (1995). Chemical Principles. Lexington: D. C. Heath. ISBN 978-0-669-39321-7. https://archive.org/details/chemicalprincip100zumd.
- ↑ "Use of Ozone Depleting Substances in Laboratories. TemaNord 516/2003". http://www.norden.org/pub/ebook/2003-516.pdf.
- ↑ "False Alarms: The Legacy of Phosgene Gas". https://hvacrschool.com/phosgene-gas/.
 |