Chemistry:Tofogliflozin
 | |
| Clinical data | |
|---|---|
| Other names | CSG452 |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
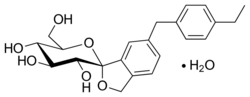
| Formula | C22H28O7 |
| Molar mass | 404.459 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Tofogliflozin (INN,[1]: 88 USAN, codenamed CSG452) is an experimental drug for the treatment of diabetes mellitus and is being developed by Chugai Pharma in collaboration with Kowa and Sanofi.[2] It is an inhibitor of subtype 2 sodium-glucose transport protein (SGLT2), which is responsible for at least 90% of the glucose reabsorption in the kidney. As of September 2012[update], the drug is in Phase III clinical trials.[3][4][needs update]
Chemistry
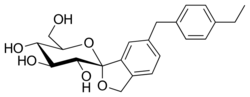
The active moiety or anhydrous form (ChemSpider ID: 28530778, CHEMBL2110731) has the chemical formula C22H26O6 and a molecular mass of 386.44 g/mol.
The United States Adopted Name tofogliflozin applies to the monohydrate, which is the form used as a drug.[5] The International Nonproprietary Name tofogliflozin applies to the anhydrous compound[1] and the drug form is referred to as tofogliflozin hydrate.
See also
References
- ↑ 1.0 1.1 "International Nonproprietary Names for Pharmaceutical Substances (INN). Recommended International Nonproprietary Names: List 65". World Health Organization. https://www.who.int/entity/medicines/publications/druginformation/innlists/RL65.pdf.
- ↑ Chugai Pharmaceutical: Development Pipeline
- ↑ "Selective SGLT2 inhibition by tofogliflozin reduces renal glucose reabsorption under hyperglycemic but not under hypo- or euglycemic conditions in rats". American Journal of Physiology. Endocrinology and Metabolism 304 (4): E414-23. February 2013. doi:10.1152/ajpendo.00545.2012. PMID 23249697.
- ↑ "Discovery of tofogliflozin, a novel C-arylglucoside with an O-spiroketal ring system, as a highly selective sodium glucose cotransporter 2 (SGLT2) inhibitor for the treatment of type 2 diabetes". Journal of Medicinal Chemistry 55 (17): 7828–40. September 2012. doi:10.1021/jm300884k. PMID 22889351.
- ↑ Statement on a nonproprietary name adopted by the USAN council: Tofogliflozin.
 |
