Chemistry:Benfluorex
 | |
| Clinical data | |
|---|---|
| Trade names | Mediator |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
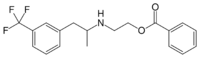
| Formula | C19H20F3NO2 |
| Molar mass | 351.369 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| | |
Benfluorex, sold under the brand name Mediator, is an anorectic and hypolipidemic agent that is structurally related to fenfluramine (a substituted amphetamine). It may improve glycemic control and decrease insulin resistance in people with poorly controlled type-2 diabetes.[1][2]
It was on the market between 1976 and 2009, and is thought to have caused between 500 and 2,000 deaths.[3] It was patented and manufactured by the French pharmaceutical company Servier. However, Servier is suspected of having marketed benfluorex at odds with the drug's medical properties.[4]
On March 29, 2021, a French court fined Servier €2.7m (£2.3m) after finding it guilty of deception and manslaughter.[5]
Drug withdrawn
On 18 December 2009, the European Medicines Agency recommended the withdrawal of all medicines containing benfluorex in the European Union, because their risks, particularly the risk of heart valve disease (fenfluramine-like cardiovascular side effects), are greater than their benefits.[6] Thus Frachon et al. showed a higher rate of unexplained valvular heart disease in people taking benfluorex.[7] Weill et al. looked at over 1 million people with diabetes demonstrating a higher hospitalization rate in benfluorex takers for valvular heart disease.[8] In France, the medication had been marketed by Servier as an adjuvant antidiabetic under the name Mediator. The drug was on the market between 1976 and 2009, and is thought to have caused between 500 and 2,000 deaths.[3] The drug was also used in Spain, Portugal, and Cyprus.
On March 29, 2021, a French court fined Servier €2.7m (£2.3m) after finding it guilty of deception and manslaughter, with Mediator linked to the deaths of up to 2,000 people. The former executive Jean-Philippe Seta was sentenced to a suspended jail sentence of four years. The French medicines agency, accused of failing to act quickly enough on warnings about the drug, was fined €303,000. The pharmaceutical group was acquitted of charges of fraud. [9]
Fenfluramine, a related drug, had been withdrawn from the market in 1997 after reports of heart valve disease,[10][11] pulmonary hypertension, and development of cardiac fibrosis. This side effect is mediated by the metabolite norfenfluramine on 5HT2B receptors of heart valves,[12] leading to a characteristic pattern of heart failure following proliferation of cardiac fibroblasts on the tricuspid valve. Both fenfluramine and benfluorex form norfenfluramine as a metabolite. This side effect led to the withdrawal of fenfluramine as an anorectic drug worldwide, and later to the withdrawal of benfluorex in Europe.
References
- ↑ "Efficacy of benfluorex in combination with sulfonylurea in type 2 diabetic patients: an 18-week, randomized, double-blind study". Diabetes Care 29 (3): 515–20. March 2006. doi:10.2337/diacare.29.03.06.dc05-1439. PMID 16505498.
- ↑ "Addition of benfluorex to biguanide improves glycemic control in obese non-insulin-dependent diabetes: a double-blind study versus placebo". Journal of Diabetes and Its Complications 13 (2): 62–7. 1999. doi:10.1016/S1056-8727(98)00004-X. PMID 10432168.
- ↑ 3.0 3.1 "France braced for diabetic drug scandal report". BBC News. 2011-01-11. https://www.bbc.co.uk/news/world-europe-12155639.
- ↑ "Mediator scandal rocks French medical community". Lancet 377 (9769): 890–2. March 2011. doi:10.1016/s0140-6736(11)60334-6. PMID 21409784.
- ↑ "Mediator drug: French pharmaceutical firm fined over weight loss pill". BBC News. 29 March 2021. https://www.bbc.co.uk/news/world-europe-56562909.
- ↑ "European Medicines Agency recommends withdrawal of benfluorex from the market in European Union". European Medicines Agency. 2009-12-18. http://www.emea.europa.eu/pdfs/human/referral/benfluorex/Benfluorex_81503309en.pdf.
- ↑ Lexchin, J., ed (April 2010). "Benfluorex and unexplained valvular heart disease: a case-control study". PLOS ONE 5 (4): e10128. doi:10.1371/journal.pone.0010128. PMID 20405030. Bibcode: 2010PLoSO...510128F.
- ↑ "Benfluorex and valvular heart disease: a cohort study of a million people with diabetes mellitus". Pharmacoepidemiology and Drug Safety 19 (12): 1256–62. December 2010. doi:10.1002/pds.2044. PMID 20945504.
- ↑ "French pharma firm found guilty over medical scandal in which up to 2,000 died". 29 March 2021. https://www.theguardian.com/world/2021/mar/29/french-pharma-firm-found-guilty-over-medical-scandal-in-which-up-to-2000-died.
- ↑ "Valvular heart disease associated with fenfluramine-phentermine". The New England Journal of Medicine 337 (9): 581–8. August 1997. doi:10.1056/NEJM199708283370901. PMID 9271479.
- ↑ "Appetite suppressants and valvular heart disease". The American Journal of the Medical Sciences 321 (4): 285–91. April 2001. doi:10.1097/00000441-200104000-00008. PMID 11307869.
- ↑ "Evidence for possible involvement of 5-HT(2B) receptors in the cardiac valvulopathy associated with fenfluramine and other serotonergic medications". Circulation 102 (23): 2836–41. December 2000. doi:10.1161/01.cir.102.23.2836. PMID 11104741.
 |
